INTRODUCTION
Allogenic blood transfusion (ABT) is a life-saving procedure in many clinical situations. However, ABT can result in unpredictable clinical consequences and risks despite technical improvements to reduce such problems [1]. A study evaluated transfusion management procedures in elective orthopedic surgeries, particularly hip and knee arthoplasties, and identified the benefits of autologous blood transfusions over ABT [2]. Several recent reports have indicated that perioperative erythropoietin in conjunction with intravenous iron sucrose supplementation is an effective alternative to ABT [3,4]. However, there are no guidelines for its dosage and safety limits. Preoperative autologous blood donation with high dose erythropoietin, although clinically optimal, is quite costly [5] Further, individual perioperative blood loss is unpredictable and therefore concerns regarding the cost-effectiveness and the safety of autologous blood donations exist. We present a case in which we managed unexpected and severe bleeding after a total hip replacement arthroplasty (THR) without a blood transfusion. We used a combination of high dose intravenous iron sucrose and moderate to low doses of erythropoietin as previously described [3,4,6,7]. Postoperative management was used exclusively in this case.
CASE REPORT
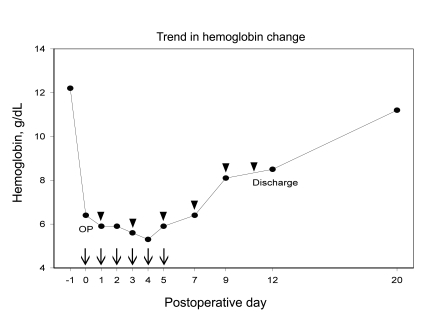
An 80-year-old woman presented to our clinic with intractable left hip pain and magnetic resonance imaging (MRI) revealed avascular necrosis of the femoral head. A total hip arthroplasty was recommended. The patient refused to donate blood for an autologous blood transfusion as a result of her religious beliefs. Her past medical history was unremarkable. Physical examination was within normal limits except for left hip tenderness. Preoperative hemoglobin was 12.2 g/dL. Other laboratory results including complete blood count, aspartate aminotransferase, alanine transaminase, blood urea nitrogen, creatinine, prothrombin time and activated partial thromboplastin time were all within normal range. A chest radiograph showed left pleural adhesions and regional parenchymal fibrotic changes, which were most likely sequelae from prior pulmonary tuberculosis. Arterial blood gas measurements and pulmonary function test results were within normal limits. Electrocardiogram , 2-D echocardiography, and carotid Doppler studies revealed no significant findings except for aging changes in the aortic valve and relaxation abnormalities. Despite her age, elective total hip arthroplasty was scheduled because of her good general health, lack of anemia, and lack of risk factors contributing to ischemic complications. Aprotinin was administered perioperatively from the procedure induction to 6 hours postoperatively until there was no significant drainage from the surgical site. However, massive blood loss put the patient in a hypovolemic and hypotensive status postoperatively despite efforts to minimize bleeding. Postoperative hemoglobin decreased to 6.4 g/dL from the preoperative measurement of 12.2 g/dL. The patient refused an allogenic blood transfusion and therefore intravenous iron sucrose (Venoferum®, Choongwoe Co., Seoul, Korea) administration was initiated. A dose of 100 mg of iron sucrose diluted in 100 mL of normal saline was administered intravenously over 1 hour twice per day up to 700 mg of cumulative dosage from the day of surgery to the third postoperative day. Additionally, 2,000 IU of recombinant erythropoietin (Recormon®, Choongwoe Co.) was injected every other day with a daily supply of iron sucrose vitamin B12, vitamin C, and folic acid. Dalteparin was administered at a dose of 5,000 IU per day to prevent deep vein thrombosis. Hemoglobin level decreased to 5.3 g/dL on the fourth postoperative day (Table 1). Further injection of 200 mg of iron sucrose in 200 mL of normal saline was administered twice per day on the fourth and fifth postoperative days. A total of 1,500 mg of iron sucrose was administered intravenously for 6 consecutive days. The patient complained of mild chest discomfort and dyspnea after the last dose of iron sucrose, and this was most likely a side effect of intravenous iron sucrose which resolved with a 50 mg hydrocortisone injection. On the seventh postoperative day, the patient had a hemoglobin of 6.4 g/dL on and was able to walk with assistance. There were no significant complications or residual side effects of high dose intravenous iron sucrose or erythropoietin therapy. The final dose of erythropoietin was administered on the twelfth postoperative day with a hemoglobin of 8.5 g/dL (Fig. 1), a corrected reticulocyte count of 2.64%, a transferrin saturation of 30%, and a ferritin level of 906 ng/mL. Total dose of erythropoietin was 12,000 IU which was equivalent to 40 IU/kg for a single dose and 240 IU/kg for the cumulative dose. The patient was discharged in good condition with oral doses of vitamin B12, vitamin C and folic acid. The patient presented for follow-up on the twentieth postoperative day with a hemoglobin of 11.2 g/dL and no clinical complications.
DISCUSSION
Studies have demonstrated the safety of intravenous iron sucrose over intravenous iron sucrose dextran [8]. The dextran molecule, which acts as a carbohydrate antigen, causes severe transfusion reactions and even fatalities. This limits the use of iron sucrose dextran in the clinical setting [9]. Recent reports have suggested the role of perioperative intravenous iron sucrose in enhancing the safety of allogenic blood transfusions [3,4]. Intravenous iron sucrose doses did not exceed 400 mg perioperatively with single injection doses of erythropoietin as high as 40,000 IU in previous studies. We reported relatively high doses of parenteral iron sucrose and low doses of erythropoietin in the management of an anemic patient who refused an allogenic blood transfusion. The typical dose of parenteral iron sucrose can be as high as 200 mg without significant side effects in surgical or dialysis patients [3,4,10] and these patients received recombinant erythropoietin at single doses of 300 IU/kg or cumulative doses of 40,000 IU/wk [11]. Clinical conditions such as inflammation result in inadequate erythropoietin production and function. However, additional reservoirs of erythropoietin in the patient with normal renal function are stimulated in hypoxic conditions. Therefore, conditions which result in relative erythropoietin deficiency might benefit from the administration of low dose erythropoietin. This low dose erythropoietin could correct anemia and prove to be cost-effective with an adequate supplement of available iron sucrose in the acute clinical setting. Erythropoietin administration is more costly than parenteral iron sucrose among alternatives for ABT.
The inflammatory status of the postoperative patient affects iron sucrose metabolism and is mediated by several cytokines. The cytokine hepcidin, which is important in familial hemochromatosis, plays a chief role in the impairment of iron sucrose utilization in the clinical setting [12]. Hepcidin is a synthesized by hepatocytes and stimulated by inflammation or iron sucrose overload. It is a negative regulator for both iron sucrose absorption in the gut and iron sucrose release from macrophages [13] in its role in anemia of chronic illness. Physiologic reduction of serum iron sucrose could serve as a protective strategy against bacteria since limitation of available iron sucrose creates an inhospitable environment in bacterial infection. However, hepcidin limits iron sucrose utilization and enhances iron sequestration, and therefore baseline exogenous iron supplementation would not result in adequate levels of available iron for bone marrow erythropoiesis, particularly if stimulated by exogenous erythropoietin. Further, both decreased gut absorption of iron and sequestration of iron mediated by hepcidin suggest a particular benefit of parenteral iron as compared with oral iron supplementation.
Erythropoietin accelerates the erythropoiesis in its role as an anti-apoptotic cytokine and is involved in protection against ischemic insults including strokes or renal injuries. Erythropoietin receptors are located in the bone marrow, heart, brain, and kidney [14], which explains the nonhematologic effects of erythropoietin. Its protective effects in trauma patients is documented in a large randomized study [15]. However, the routine administration of erythropoietin in critical patients is still controversial [16].
There may be a role for erythropoietin in the management of the hypovolemic and ischemic patients because of its protective and anti-ischemic activity [17]. Erythropoietin could serve as a bridge to maintain hematocrit from the time gap between blood loss and hemoglobin increase. Although it is not indicated in general critical cases, it may have a role as an adjunct therapy in the management of the anemic patient to reduce the rate of ABT.
Folic acid and vitamin B12 are essential in rapid erythropoiesis. Vitamin C has an active role in the release of sequestrated iron from reticuloendothelial cells and therefore facilitates the erythropoietin response. Favorable effects of parenteral iron treatment combined with vitamin C administration also suggest that vitamin C contributes to decreased oxidative stress as an antioxidant, may reduce iron-overload toxicity, and may decrease the total body iron requirement [18]. We believe that the administration of vitamin C in our patient, even at a minimal dosage, contributed to accelerated hematopoiesis.
Aprotinin was used in this case study instead of a perioperative blood salvage system. Aprotinin is a polypeptide extracted from bovine lung tisse and has anti-fibrinolytic and anti-kallikrein activity. There are several reports revealing the efficacy and safety of aprotinin in orthopedic surgeries [19]. However, its association with thrombosis and renal failure limits its use in clinical practice.
There are several alternatives to ABT in clinical case management. Preoperative autologous blood donation, perioperative blood salvage systems, and intraoperative hemodilution all serve to reduce the ABT rate in major orthopedic surgeries [20]. However, the patient in this case report refused all blood products and, as a result, all of the aforementioned procedures. Preoperative erythropoietin with intravenous iron sucrose without autologous blood donation would have been the primary option in this case.
In conclusion, aggressive high dose intravenous iron sucrose with low dose erythropoietin was successful in our postoperative management of a severely anemic patient.




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





