 |
 |
| Korean J Intern Med > Volume 28(6); 2013 > Article |
|
Abstract
Background/Aims
This study was performed to compare the mucosal findings after esophagogastroduodenoscopy in two groups before and after the use of alendronate only and following administration of the enteric-coated alendronate (5 mg) and calcitriol (0.5 µg) combined drug (Maxmarvil, Yuyu Co.).
Methods
The study population consisted of 33 postmenopausal healthy female volunteers, aged 50 to 70 years (mean age, 58 ± 5) without gastrointestinal symptoms and with normal baseline endoscopic findings. Esophagogastroduodenoscopy was performed at baseline and was repeated 2 weeks later after daily intake of Maxmarvil (n = 17 subjects) or alendronate only (n = 16 subjects). Mucosal injury scores were reported by an endoscopist after 2 weeks of treatment with each medication schedule.
Results
Esophageal mucosal injuries developed in two of 16 subjects in the alendronate only group and 0 of 17 in the Maxmarvil group. Gastric mucosal injuries developed in eight subjects in the alendronate group and four subjects in the Maxmarvil group; this difference was statistically significant.
Conclusions
The mucosal damage scores for the alendronate group (total score 24) were significantly higher than those for the Maxmarvil group (total score 9) in the esophagus and stomach. Therefore, this study suggested that enteric-coated Maxmarvil is less harmful to gastrointestinal mucosa than alendronate, and may improve the tolerability of osteoporosis medication in clinical practice.
Osteoporosis is a chronic disease that typically requires long-term therapy to increase patients' bone mineral density (BMD) and to prevent the occurrence of fractures [1].
Alendronate is a bisphosphonate (BP) that has been shown to increase BMD in the treatment of osteoporosis in postmenopausal females [2,3]. One of the most common adverse events associated with the use of alendronate is upper gastrointestinal (GI) discomfort. Esophageal and gastric irritation in patients receiving aminobisphosphonate drugs is well recognized [4,5]. The rates of GI adverse events and the discontinuation of alendronate in clinical practice are much higher than those reported in randomized, double-blinded, clinical trials [6-13].
Ettinger et al. [13] reported that discontinuation of alendronate therapy occurs in about 30% of patients within the first 6 months of treatment, which is most often due to GI complaints. Moreover, efforts should be made to minimize the risk of adverse GI events and to improve the tolerability of alendronate medication, because patients with osteoporosis who were in good long-term medication compliance were shown to have substantially lower risk of fracture [14,15].
BPs are usually dissolved in the stomach and form crystals with gastric acid that can cause irritation of mucosa of the esophagus and stomach [16]. The combination of alendronate (5 mg) and calcitriol (1,25(OH)2D3, 0.5 µg) (Maxmarvil, Yuyu Co., Seoul, Korea) has a unique characteristic that allows it to circumvent this negative GI reaction, in that the drug, a pH 5.5 sensitive enteric-coated tablet, passes through the stomach and is absorbed in the intestine.
Shiraki et al. [17] reported that a half dose (5 mg) of alendronate was useful in the treatment of osteoporosis in the Japanese population, and that it had less adverse events. According to the data from a Korean clinical trial reported by Rhee et al. [18], Maxmarvil was effective in increasing lumbar BMD and was presumed to be safe. Therefore, we hypothesized that Maxmarvil has lesser effects on the GI mucosa than conventional alendronate medication. To test our hypothesis, we compared the endoscopic findings between the two groups before and after the use of both alendronate only and Maxmarvil.
Initially, 107 postmenopausal females aged 50 to 70 years old were enrolled in the study. The subjects were selected according to the following inclusion criteria: no menstruation for more than 12 months with high follicle stimulating hormone (FSH) level (> 30 IU/L) and the presence of more than one osteoporotic vertebral fracture (defined as having vertebral fracture more than grade 2 by Genant semiquantitative method [19], except those with any fracture at L2 to L4) or those having BMD at L1 to L4 with T score < -2.5. Exclusion criteria included a history of peptic ulcer, gastroesophageal reflux disease, esophagitis, gastritis, GI surgery, and current or recent treatment with any H2-receptor antagonist, proton pump inhibitor, sucralfate, misoprostol, cisapride, metoclopramide, aspirin, and nonsteroidal anti-inflammatory drugs for 2 weeks prior to entry. In addition, patients with secondary osteoporosis related to glucocorticoid usage, hyperthyroidism, hyperparathyroidism, or Paget disease were excluded. Patients with serious diseases, such as renal impairment (serum Cr > 1.5 mg/dL) or hepatic dysfunction (aspartate aminotransferase, alanine aminotransferase > 2-fold the normal range) and receiving estrogen, progesterone, calcitonin, fluoride, BP, or calcitriol within 12 weeks prior to entry were excluded. Patients with conditions that contraindicated the use of BP, such as those who were unable to sit down or stand for more than 30 minutes, or those who had hypersensitivity to the drug, were also excluded.
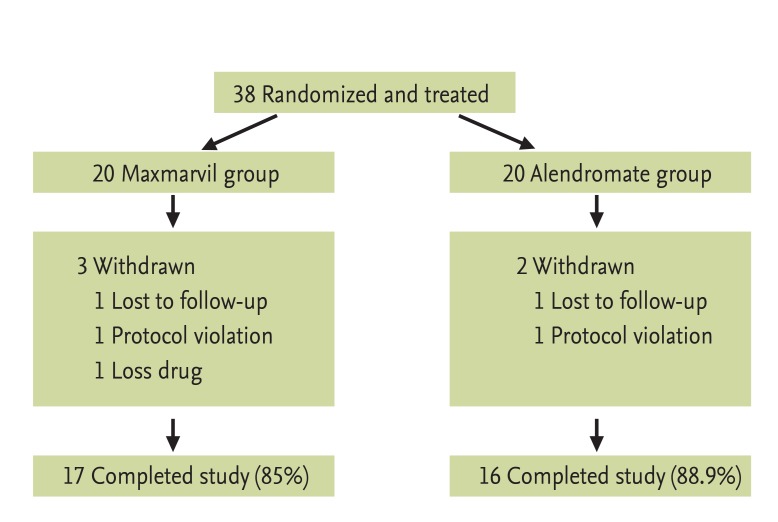
This was a singlecenter, open label, randomized, head to head clinical study. Baseline esophagogastroduodenoscopy (EGD) was performed in each subject that satisfied the criteria for participation in the study. Thirty-eight subjects with normal endoscopic findings at baseline were randomized to receive the study drug for 14 days. One group of patients received tablets of Maxmarvil (n = 20) and the other group received tablets of alendronate (n = 18) in the early morning with a glass of plain water after an overnight fast. After medication, they were instructed to remain upright for at least 30 minutes before the first food intake of the day. During the study, five of 38 subjects (Maxmarvil group, three and alendronate group, two) were withdrawn due to loss of follow-up and protocol violation. The remaining 33 subjects (Maxmarvil group, 17 and alendronate group, 16) completed the study (Fig. 1).
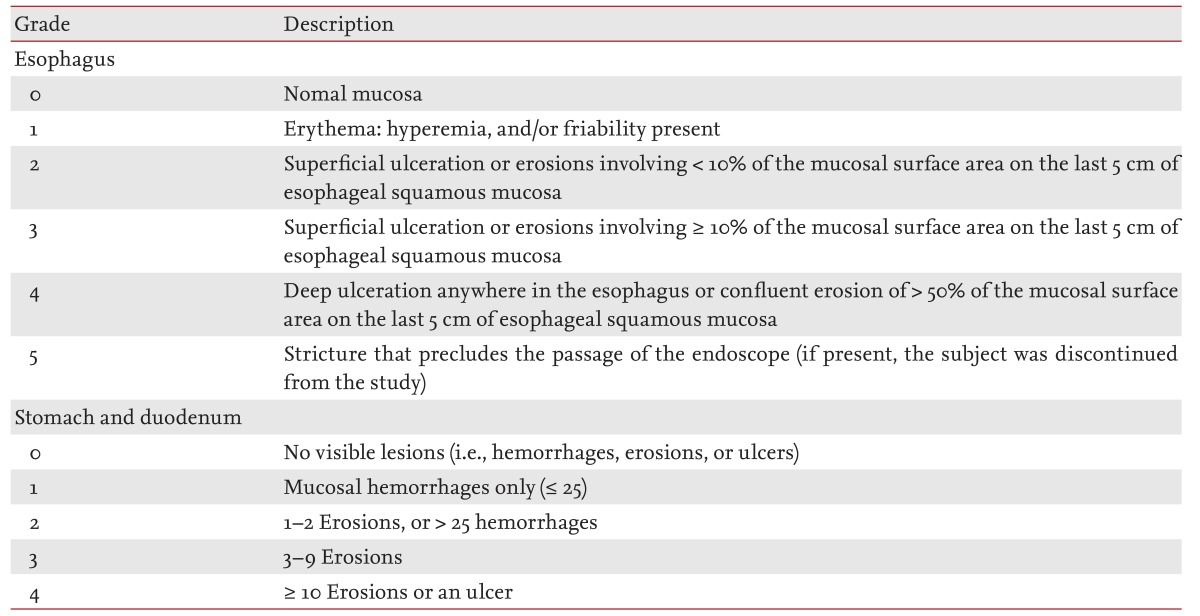
All endoscopic examinations were performed at baseline and on day 15 by the same gastroenterologist, who remained blinded to the treatment information. Esophageal mucosa were assessed using the Hetzel-Dent grading/scoring system, and gastric and duodenal mucosa were assessed using the Lanza grading system (Table 1) [8,20].
Height (cm) and weight (kg) were measured in duplicate and the results were averaged. Body mass index (BMI) was calculated by dividing the weight (kg) by the square of height (m2).
BMD measurements were performed using a central dual-energy X-ray absorptiometer (QDR 4500A, Hologic, Waltham, MA, USA). Vertebral fractures seen on spinal X-rays were diagnosed using a visual semiquantitative assessment [19].
Written informed consent was obtained from each participant, and the study protocol was designed in accordance with the ethical guidelines of the 1975 Declaration of Helsinki, as reflected in a prior approval by the institution's Human Research Committee.
Statistical analysis was performed using SPSS version 11.0 for Windows (SPSS Inc., Chicago, IL, USA). All results are presented as the mean ± SD. The mean values were compared between the two treatment groups with Student t test. Fisher exact test was used to analyze the difference in mucosal damage grading score between the two groups. In all analyses, p < 0.05 was taken to indicate statistical significance.
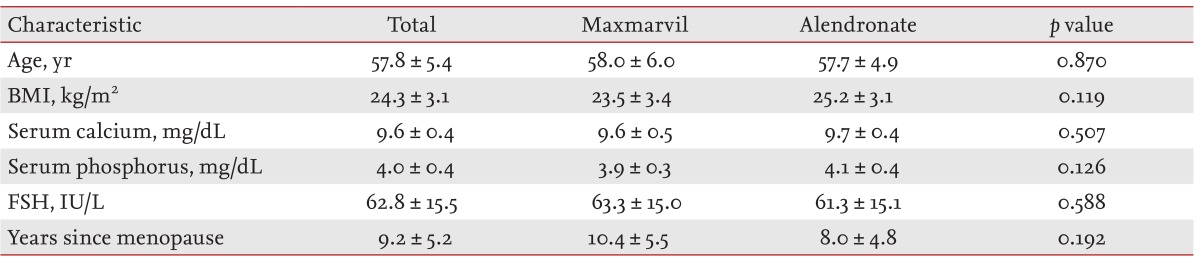
The general characteristics of the study subjects are presented in Table 2. Thirty-three subjects who fulfilled the eligibility criteria were enrolled in the study, and completed treatment without violation of the study protocol. The mean age was 58 years, and the mean BMI was 24.3 kg/m2. The mean number of years since menopause was 9.2 years. The age, BMI, and number of years since menopause were not significantly different between the two groups (p = 0.870, p = 0.119, and p = 0.192, respectively). In addition, there were no statistically significant differences in the mean serum calcium, phosphorus, or FSH levels between the two groups (p = 0.507, p = 0.126, and p = 0.588).
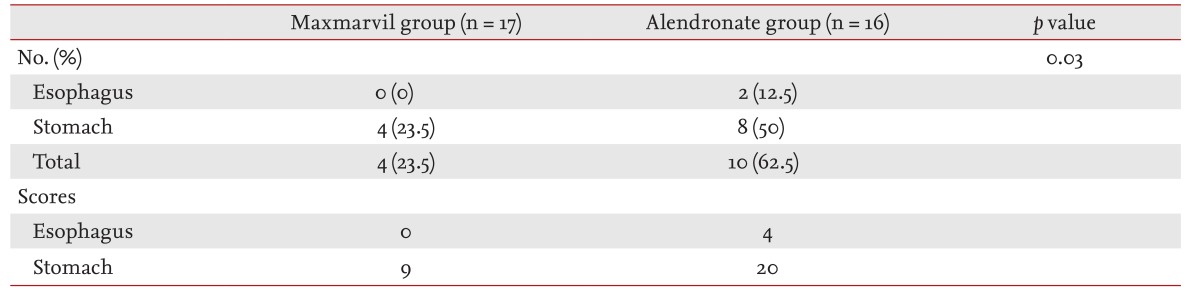
Esophageal and gastric mucosal injury were significantly different between the two groups (p = 0.03) (Table 3). Esophageal mucosal injuries developed in two subjects in the alendronate only treatment group (mucosal damage score, 4), while no esophageal mucosal injuries were found in the Maxmarvil group. Gastric mucosal injuries developed in eight of 16 subjects with alendronate treatment (mucosal damage score, 20) and in four of 17 subjects with Maxmarvil treatment (mucosal damage score, 9); these differences were statistically significant.
In this open label, randomized, head to head clinical study, we found that the mucosal damage scores in the esophagus and stomach of the Maxmarvil (enteric-coated combination of alendronate 5 mg and calcitriol 0.5 mg) group were significantly lower than those of the alendronate group, suggesting that enteric-coated Maxmarvil may be less harmful to the GI mucosa.
Most previous studies of the relationships between upper GI irritability and alendronate were in the form of placebo-controlled trials. Based on extensive clinical trials, patients treated with alendronate at doses of 5 to 10 mg appeared to show little GI irritability compared to those given a placebo [21,22]. However, GI adverse events and the discontinuation of alendronate are more common in real clinical practice than reported in clinical trials [9-13,23].
Several endoscopic studies have argued that alendronate is toxic to the upper GI tract. Graham and Malaty [24] reported that 10 mg of alendronate administered for 14 days caused visible gastric mucosal injury in 47% of patients, and that three of 15 volunteers developed gastric ulcers. In contrast, Lanza et al. [8] reported that daily administration of 10 mg of alendronate had no toxic effect on the gastric mucosa. There are a number of reasons why the literature might present conflicting data concerning the adverse GI effects of alendronate, and there may also be some discordant results between patients in clinical trials and those in general clinical practice due to several possible factors. One of the more subtle factors is that general clinical patients may not be highly motivated because they did not volunteer for medication. Another factor is that general physicians may not provide adequate education, encouragement, and useful information that are usually supported in clinical trials by a specialized researcher.
Maxmarvil, a combination of alendronate (5 mg) and calcitriol (1,25(OH)2D3, 0.5 µg), is prepared as enteric-coated tablets. Therefore, this drug, a pH 5.5 sensitive enteric-coated tablet, passes the stomach and is dissolved in the intestine and then absorbed. Enteric-coated preparations can prevent BPs from dissolving in the stomach, and hence from irritating the mucosa of the esophagus and stomach through crystallization of a gastric acid-drug mixture [16]. We postulated that enteric-coated Maxmarvil may be less harmful to the upper GI mucosa than alendronate.
To our knowledge, this is the first study to evaluate and compare the GI mucosal adverse events using endoscopy after medication. Approximately 50% of patients discontinued daily BP therapy within 1 year, which negatively impacts treatment outcomes. Almost three quarters (73%) of patients who stopped alendronate therapy had experienced a side effect, and a little less than half (46%) were experiencing heartburn, nausea, and stomachache [25].
The general dosage of alendronate is 10 mg daily. However, lower dosages of alendronate, such as 5 mg daily, showed similar increases in BMD in postmenopausal Korean and Japan women with osteoporosis, as that seen in Caucasian women treated with 10 mg daily of alendronate [17,18]. There were only a few cases of GI discomfort associated with Maxmarvil. A study in Japan showed that even a daily dose of 2.5 mg of alendronate was as effective as a 10 mg daily dose of alendronate in increasing lumbar BMD over 36 weeks of treatment [26]. Adverse events may negatively impact medication compliance, and may therefore increase the risk of fracture.
Alendronate-induced small intestinal damage has not been studied in detail. Haderslev et al. [27] examined whether an alendronate daily dose of 10 mg is effective and tolerable in Crohn disease. They reported that alendronate was generally well tolerated and did not adversely affect the clinical course of the disease. Although alendronate has not been associated with intestinal mucosal irritation, enteric-coated Maxmarvil is mostly absorbed in the intestine, and therefore further research is needed to establish the intestinal safety and tolerability of the drug.
Our study had several limitations, of which the main limitation was the small sample size. The inclusion criteria for the study population was very strict, and hence many participants (64.5%) were excluded due to abnormal findings on baseline EGD. Although the sample size was calculated to detect a statistically significant difference between the groups, the statistical power of our study would have been higher if the sample size had been larger. In fact, the number of subjects enrolled in other endoscopy studies has usually been 20 to 32 [28,29]. More than half (64.5%) of the population were excluded due to abnormal findings on baseline EGD. An additional limitation was that the subjects in our study were all postmenopausal Korean females. Hence, further studies are warranted with larger study populations of various ages and ethnic groups. A weekly dosing regimen of alendronate is currently preferred, as it has the potential to provide greater convenience and enhance compliance. Lanza et al. [30] conducted a randomized, double blind, placebo-controlled endoscopy study to confirm the upper GI safety and tolerability of 70 mg of oral alendronate once weekly, and reported that it was not associated with any increased endoscopic lesions in the upper GI tract compared to a placebo control. Therefore, a comparison between daily Maxmarvil (alendronate 5 mg and calcitriol 0.5 µg) and weekly alendronate (70 mg) once weekly on GI adverse events and efficacy will be interesting in current clinical practice.
In conclusion, we found that the mucosal damage scores in the esophagus and stomach for the Maxmarvil (enteric-coated combined alendronate 5 mg and calcitriol 0.5 µg) group were significantly lower than those for the alendronate only group. This study suggested that enteric-coated Maxmarvil is less harmful toward the GI mucosa than alendronate and improves the tolerability of medication in clinical practice. However, our data are preliminary, and the study requires more statistical power and insight into the pathogenic mechanisms of GI protection by enteric-coated alendronate with calcitriol.
1. The mucosal damage scores in the esophagus and stomach for the Maxmarvil (enteric-coated combined alendronate 5 mg and calcitriol 0.5 µg) group were significantly lower than those for the alendronate only group.
2. Enteric-coated Maxmarvil is less harmful toward the gastrointestinal mucosa than alendronate and may improve the tolerability of medication in clinical practice.
References
1. NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy, March 7-29, 2000: highlights of the conference. South Med J 2001;94:569–573PMID : 11440324.


2. Liberman UA, Weiss SR, Broll J, et al. Effect of oral alendronate on bone mineral density and the incidence of fractures in postmenopausal osteoporosis: the Alendronate Phase III Osteoporosis Treatment Study Group. N Engl J Med 1995;333:1437–1443PMID : 7477143.


3. Pols HA, Felsenberg D, Hanley DA, et al. Multinational, placebo-controlled, randomized trial of the effects of alendronate on bone density and fracture risk in postmenopausal women with low bone mass: results of the FOSIT study: Fosamax International Trial Study Group. Osteoporos Int 1999;9:461–468PMID : 10550467.


4. Lufkin EG, Argueta R, Whitaker MD, et al. Pamidronate: an unrecognized problem in gastrointestinal tolerability. Osteoporos Int 1994;4:320–322PMID : 7696825.


5. Recker RR, Lewiecki EM, Miller PD, Reiffel J. Safety of bisphosphonates in the treatment of osteoporosis. Am J Med 2009;122(2 Suppl):S22–S32PMID : 19187809.


6. Bauer DC, Black D, Ensrud K, et al. Upper gastrointestinal tract safety profile of alendronate: the fracture intervention trial. Arch Intern Med 2000;160:517–525PMID : 10695692.


7. Tucci JR, Tonino RP, Emkey RD, Peverly CA, Kher U, Santora AC 2nd. Effect of three years of oral alendronate treatment in postmenopausal women with osteoporosis. Am J Med 1996;101:488–501PMID : 8948272.


8. Lanza F, Rack MF, Simon TJ, Lombardi A, Reyes R, Suryawanshi S. Effects of alendronate on gastric and duodenal mucosa. Am J Gastroenterol 1998;93:753–757PMID : 9625122.


9. de Groen PC, Lubbe DF, Hirsch LJ, et al. Esophagitis associated with the use of alendronate. N Engl J Med 1996;335:1016–1021PMID : 8793925.


10. Levine J, Nelson D. Esophageal stricture associated with alendronate therapy. Am J Med 1997;102:489–491PMID : 9217649.


11. Kelly R, Taggart H. Incidence of gastrointestinal side effects due to alendronate is high in clinical practice. BMJ 1997;315:1235. PMID : 9393243.



12. Ribeiro A, DeVault KR, Wolfe JT 3rd, Stark ME. Alendronate-associated esophagitis: endoscopic and pathologic features. Gastrointest Endosc 1998;47:525–528PMID : 9647380.


13. Ettinger B, Pressman A, Schein J. Clinic visits and hospital admissions for care of acid-related upper gastrointestinal disorders in women using alendronate for osteoporosis. Am J Manag Care 1998;4:1377–1382PMID : 10338731.

14. Caro JJ, Ishak KJ, Huybrechts KF, Raggio G, Naujoks C. The impact of compliance with osteoporosis therapy on fracture rates in actual practice. Osteoporos Int 2004;15:1003–1008PMID : 15167989.


15. Siris ES, Harris ST, Rosen CJ, et al. Adherence to bisphosphonate therapy and fracture rates in osteoporotic women: relationship to vertebral and nonvertebral fractures from 2 US claims databases. Mayo Clin Proc 2006;81:1013–1022PMID : 16901023.


16. Ezra A, Golomb G. Administration routes and delivery systems of bisphosphonates for the treatment of bone resorption. Adv Drug Deliv Rev 2000;42:175–195PMID : 10963835.


17. Shiraki M, Kushida K, Fukunaga M, et al. A double-masked multicenter comparative study between alendronate and alfacalcidol in Japanese patients with osteoporosis: the Alendronate Phase III Osteoporosis Treatment Research Group. Osteoporos Int 1999;10:183–192PMID : 10525709.


18. Rhee Y, Kang M, Min Y, et al. Effects of a combined alendronate and calcitriol agent (Maxmarvil) on bone metabolism in Korean postmenopausal women: a multicenter, double-blind, randomized, placebo-controlled study. Osteoporos Int 2006;17:1801–1807PMID : 17019520.


19. Genant HK, Wu CY, van Kuijk C, Nevitt MC. Vertebral fracture assessment using a semiquantitative technique. J Bone Miner Res 1993;8:1137–1148PMID : 8237484.


20. Hetzel DJ, Dent J, Reed WD, et al. Healing and relapse of severe peptic esophagitis after treatment with omeprazole. Gastroenterology 1988;95:903–912PMID : 3044912.


21. Black DM, Cummings SR, Karpf DB, et al. Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures: Fracture Intervention Trial Research Group. Lancet 1996;348:1535–1541PMID : 8950879.


22. Hosking D, Chilvers CE, Christiansen C, et al. Prevention of bone loss with alendronate in postmenopausal women under 60 years of age: Early Postmenopausal Intervention Cohort Study Group. N Engl J Med 1998;338:485–492PMID : 9443925.


23. Maconi G, Bianchi Porro G. Multiple ulcerative esophagitis caused by alendronate. Am J Gastroenterol 1995;90:1889–1890PMID : 7572919.

24. Graham DY, Malaty HM. Drug-induced gastric ulcers are caused by more than just NSAIDs: alendronate gastric ulcers. Gastroenterology 1998;114(Suppl 1):A138.

25. Tosteson AN, Grove MR, Hammond CS, et al. Early discontinuation of treatment for osteoporosis. Am J Med 2003;115:209–216PMID : 12947959.


26. Shiraki M, Kushida K, Fukunaga M, et al. A placebo-controlled, single-blind study to determine the appropriate alendronate dosage in postmenopausal Japanese patients with osteoporosis: the Alendronate Research Group. Endocr J 1998;45:191–201PMID : 9700472.


27. Haderslev KV, Tjellesen L, Sorensen HA, Staun M. Alendronate increases lumbar spine bone mineral density in patients with Crohn's disease. Gastroenterology 2000;119:639–646PMID : 10982756.


28. Lowe CE, Depew WT, Vanner SJ, Paterson WG, Meddings JB. Upper gastrointestinal toxicity of alendronate. Am J Gastroenterol 2000;95:634–640PMID : 10710050.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



