 |
 |
| Korean J Intern Med > Volume 29(2); 2014 > Article |
|
Abstract
Hepatocellular carcinoma (HCC) is one of the most common cancers worldwide. The only curative treatment modalities for HCC are surgery, percutaneous ablation, and liver transplantation. Unfortunately, the majority of patients have unresectable disease at diagnosis. Therefore, effective treatment options are needed for patients with advanced HCC. The current standard treatment for patients with advanced HCC, according to the Barcelona Clinic Liver Cancer staging system, is the multikinase inhibitor sorafenib. Other alternative therapies are required, due to the limited treatment response to, and tolerance of, this molecular target agent. Clinical trials of hepatic artery infusion chemotherapy, radioembolization, and multimodal treatments have shown favorable results in advanced HCC patients. This article introduces new treatment modalities for advanced HCC and discusses future therapeutic possibilities.
Hepatocellular carcinoma (HCC) is the sixth most common cancer and third leading cause of cancer-related death worldwide, and its incidence is increasing [1]. Despite the widespread use of surveillance programs in at-risk populations, more than half of HCC cases are diagnosed late, and curative therapies such as surgical resection, transplantation, or radiofrequency ablation are possible in fewer than 30% of the patients. Most HCC patients have unresectable disease at diagnosis [2,3]. The prognosis of patients with HCC remains poor, and life expectancy is difficult to predict. Although patients with advanced HCC have a median survival of less than 1 year, the performance status is heterogeneous and the tumor burden ranges from single nodules associated with limited portal vein thrombosis (PVT) amenable to curative attempts to multiple intrahepatic metastasis associated with extrahepatic spread.
Sorafenib, the multityrosine kinase inhibitor, improves the overall survival (OS) and has become the current standard of care in advanced HCC [4,5]. Sorafenib has shifted the paradigm of systemic therapy for HCC to molecular-targeted therapies. However, alternative therapies are required due to the low response rates, high cost-benefit ratio, and unsuitability of molecular agents in clinical practice.
Hepatic arterial infusion chemotherapy (HAIC) has been used in advanced HCC in Asia with a view to improving the therapeutic indexes. HAIC delivers drugs directly to the tumor bed, where they have a greater first-pass effect; in addition, the systemic side effects are potentially minimized [6,7].
Yttrium-90 (90Y) radioembolization is a novel transarterial approach to radiation therapy for liver cancer that has achieved efficacious tumor control in advanced patients with PVT [8]. A study of transarterial chemoembolization (TACE) combined with sorafenib or radiation therapy reported favorable efficacy and safety for advanced HCC treatment [9,10].
As more therapies become available for patients with advanced HCC, the treatment decisions become increasingly complex. An individualized treatment strategy for advanced HCC is necessary. It is important to know about recent treatment modalities. Therefore, this article introduces recent treatments for advanced HCC.
Sorafenib is the only molecular target agent approved for the treatment of patients with advanced HCC (extrahepatic metastasis or vascular invasion). This multikinase inhibitor showed an OS benefit via its antiangiogenic and antiproliferative effects on C- and B-Raf serine/threonine kinases (comprising the Raf/MEKL/ERK pathway), vascular endothelial growth factor receptor (VEGFR) and platelet-derived growth factor receptor (PDGFR) tyrosine kinases, and Flt-4 and c-Kit [11,12].
In the first randomized controlled trial (Sorafenib HCC Assessment Randomized Protocol, SHARP), which was conducted in Europe and the United States [4], patients with advanced HCC who had not received previous systemic therapy were assigned randomly to placebo or sorafenib 400 mg twice a day. The median OS was 10.7 months for the sorafenib group versus 7.9 months for the control group (hazard ratio [HR, sorafenib/placebo], 0.69; 95% confidence interval [CI], 0.55 to 0.88). The time to progression (TTP) on sorafenib and placebo also differed significantly and was 5.5 and 2.8 months, respectively (HR [sorafenib/placebo], 0.58; 95% CI, 0.45 to 0.74). A second trial was designed as a bridging study to evaluate the overall efficacy and safety of sorafenib in the Asia-Pacific population [5]. The median OS for the sorafenib group was 6.5 months vs. 4.2 months for the placebo (HR [sorafenib/placebo], 0.68; 95% CI, 0.50 to 0.93). Patients in the sorafenib group had a longer median TTP (2.8 months vs. 1.4 months; p < 0.001; HR, 0.57; 95% CI, 0.42 to 0.79).
The most common drug-related adverse events of sorafenib include diarrhea, fatigue, hand-foot skin reaction (HFSR), and rash. These events occurred in 20% to 40% of patients, and most were grade 1 or 2. The most common causes of treatment interruption or dose reduction were HFSR, rash, and diarrhea [4,5,13]. In the Korean population, HFSR was the most common reason for treatment interruption. Sorafenib might be less well tolerated by Asian patients, as compared to Western patients. The grade 3 adverse rate was 8% in SHARP compared with 11% in the Asian trial [14,15]. HFSR occurred in 21% of the patients in the SHARP trial; the rate was 45% in the Asian phase III sorafenib trial.
Hepatic artery infusion chemotherapy has been used to treat advanced HCC patients with vascular invasion, multiple intrahepatic lesions, or both. This treatment is theoretically more effective against HCC than systemic chemotherapy because hepatic arterial infusion of chemotherapeutic agents enables the delivery of high doses of drugs directly to the hypervascular HCC. In addition, the first-pass effect in the hepatic parenchyma results in lower systemic drug levels after HAIC than after systemic administration. HAIC reduces the drug toxicity and side effects. In Asia, especially Japan and Korea, HAIC has been applied in advanced HCC with a view to improving the therapeutic indexes.
Angiography of the celiac trunk and superior mesenteric artery via the femoral artery is used to evaluate the hepatic artery vascularization and portal vein patency. HAIC is contraindicated if there is any uncorrectable delivery of anticancer drugs to the gastrointestinal tract or reversed blood flow out of the liver. If necessary, nontarget vessels are embolized to minimize the flow of chemotherapeutic agents into both uninvolved liver parenchyma and extrahepatic tissues. After finding the artery feeding the HCC, the tip of the catheter is inserted into the proper hepatic or common hepatic artery. The proximal end of the catheter is connected to the injection port, which is implanted in a subcutaneous pocket in the right iliac fossa.
In clinical practice, TACE has long been used as palliative therapy for unresectable HCC. However, high-dose hepatic artery infusion chemotherapy has showed favorable outcomes in patients with intractable, advanced HCC. Kim et al. [16] compared TACE and HAIC in advanced HCC and found that the objective response rate was better with HAIC (16.7% vs. 0%; p=0.030). The OS was longer with HAIC (median survival 193 days vs. 119 days; p=0.026) and there were no serious adverse effects in the HAIC group.
Various chemotherapeutic agents have been tried individually or in combination for HAIC, with the most commonly used being the thymidylate synthase inhibitor 5-fluorouracil (5-FU) and cisplatin, a DNA cross-linking compound [17,18]. The mechanism of 5-FU might involve antitumor effects via cell-cycle arrest and the induction of apoptosis. Cisplatin amplified the effect of 5-FU and is regarded as a biochemical modulator of 5-FU [19,20]. Cisplatin not only inhibits DNA synthesis directly but it also interrupts methionine transport into tumor cells, resulting in a reduction in the intracellular methionine level. Simultaneously, cisplatin increases the levels of intracellular reduced folic acid (5,10-CH2THF, THF), which is necessary for tight binding of FdUMP, a major active metabolite of 5-FU, to its target enzyme, thymidylate synthetase [21,22]. While various combinations of 5-FU and cisplatin have been investigated for use in HAIC [23,24], few studies have assessed these regimens in patients with advanced HCC.
In a recent prospective multicenter study in Korea, Woo et al. [25] compared high-dose (5-FU, 500 mg/m2 on days 1 to 3 and cisplatin, 60 mg/m2 on day 2) and low-dose (5-FU, 170 mg/m2 and cisplatin, 7 mg/m2 on days 1 to 5) HAIC. The objective response rate was significantly better in the high-dose group (16.7% vs. 0%; p=0.024). The median time to disease progression (145 days vs. 90 days; p=0.095) and OS (193 days vs. 153 days; p=0.108) were prolonged slightly in the high-dose group. HAIC showed more therapeutic benefit with the high-dose regimen.
In addition, Kim et al. [22] reported the long-term clinical outcome of high-dose HAIC. During a median of 9.5 months, the overall disease control rate was 62.3%. The median progression-free survival (PFS) and OS were 6.0 and 9.5 months, respectively. These results are similar to the reported efficacy of sorafenib.
However, the limitations of the HAIC studies are the small sample sizes and lack of large randomized trials. It will be necessary to compare sorafenib and HAIC in advanced HCC. Randomized trials comparing HAIC and sorafenib are ongoing and might validate the overall clinical advantages of HAIC.
Traditionally, HCC has been regarded as a radioresistant tumor due to the limited ability to deliver lethal doses using external beam techniques. Radioembolization is a new concept in radiation therapy for HCC. Microspheres containing 90Y are injected through the hepatic artery, become trapped at the precapillary level, and selectively emit higher-dose radiation to the HCC. 90Y is a pure beta-emitter, and generates high-energy radiation with a short half-life (2.67 days), and a short tissue penetration (mean, 2.5 mm; maximum, 11 mm) [8]. This mechanism limits the exposure of the surrounding normal liver parenchyma, thereby permitting higher dose delivery than with external radiation.
Before a radioembolization procedure, all patients should undergo a 99mTc-labeled macroaggregated albumin test to see whether the pulmonary shunt is less than 20%. If the pulmonary shunt exceeds 20%, systemic distribution of the radioactive isotope with resulting toxicity is anticipated. Any uncorrectable delivery of 90Y to the gastrointestinal tract, reversed blood flow out of the liver, or complete PVT are contraindications of radioembolization, and the presence of these is evaluated together with the shunt study by angiography before treatment [26]. If necessary, nontarget vessels are embolized to minimize the flow of 90Y microspheres into both uninvolved liver parenchyma and extrahepatic tissues. Estimation of the radiation dose actually delivered to the tumor tissue is complicated. The basic premise for 90Y microsphere dosimetry is to ensure that the liver parenchyma exposure does not exceed 70 Gy, while at least 120 Gy must accumulate within the tumor to deliver a dose-dependent tumoricidal effect [27]. Accurate assessment of the target tumor and liver volumes is critical because they affect the absorbed radiation dose estimates directly. The desired 90Y activity is calculated using a partition model or body surface area model [28]. Infusion of the microspheres into one or both lobes can generally be accomplished in a single session, unless the patient has impaired liver function, in which case two procedures separated by 30 to 45 days might be required [29]. Unlike TACE, the response to radioembolization needs to be assessed approximately 3 months postprocedure due to the delayed radiation effect on the tumor [30].
Kulik et al. [31] evaluated a phase II study on the safety and clinical benefit of radioembolization in a larger cohort of patients with unresectable HCC complicated by PVT. The partial response (PR) rate was 42.2%. Stable disease (SD) was seen in 34.7%. The median survival of patients with branch PVT after 90Y was 261 days, while that of patients with main PVT was 148 days. In an Italian study, the objective response was 34.3%, whereas the disease control rate was 74.3% [32]. During a median follow-up of 36 months, the median TTP was 11 months with no significant difference between PVT versus no PVT (7 months vs. 13 months). The median OS was 15 months with a non-significant trend in favor of non-PVT versus PVT patients (18 months vs. 13 months). The tumor response was significantly correlated with the absorbed dose in target lesions and a threshold of 500 Gy predicted the response. 90Y radioembolization was effective in advanced HCC, particularly in the case of PVT.
In a recent prospective multicenter study in Korea, Kim et al. [33] reported the efficacy and safety of radioembolization in 40 HCC patients. The median follow-up was 29 months. At 3 months, a complete response (CR), PR, and SD were seen in 4 patients (10.0%), 19 patients (47.5%), and 15 patients (37.5%), respectively. At 3 months, the response rate was 57.5% (23/40) and the disease control rate was 95% (38/40), while the respective values at 6 months were 63.9% (23/36) and 83.3% (30/36). The median TTP was 18 months. The most common adverse event was mild to moderate abdominal pain, and all of the complications were manageable.
Approximately 20% of the patients develop liver-related toxicity and 3% suffer treatment-related death [34]. No randomized controlled trial has compared the efficacy of 90Y radioembolization with sorafenib in advanced HCC patients, although a randomized trial comparing 90Y radioembolization and sorafenib is ongoing.
The Barcelona Clinic Liver Cancer staging and treatment strategy does not include multimodal treatments involving the use of combination therapies, although they are often used in clinical practice with good effect [35].
The potential efficacy of sorafenib combined with locoregional treatment is an interesting issue in patients with advanced HCC. There is a rationale for combined treatment. TACE induces ischemia and has a chemotherapeutic effect, resulting in local and systemic increases in vascular endothelial growth factor which can increase tumor angiogenesis and aggravation [36,37]. Theoretically, sorafenib can counteracted this upregulation. In a retrospective study, Choi et al. [9] compared the treatment efficacy of sorafenib combined with TACE with sorafenib alone in advanced HCC. In the propensity score-matched cohort, the median TTP in the combined group was significantly longer than that in the monotherapy group (2.7 months vs. 2.1 months, respectively; p=0.011), while the median OS was not (9.1 months vs. 6.7 months; p=0.21). TACE plus sorafenib was superior to sorafenib alone with respect to TTP in patients with advanced HCC, although the survival benefit was not significant.
Combined TACE and radiation therapy is an alternative treatment option in patients with locally advanced HCC with PVT [38,39]. In the past, radiotherapy has not often been used to treat HCC due to the relatively low tolerance of the liver to radiotherapy [40,41]. However, recent technological developments have enabled more successful treatment of HCC by delivering a substantial dose of radiation to the tumor and avoiding damage to surrounding organs. This has made it possible to spare the volume of uninvolved liver from radiation therapy [42]. Yoon et al. [10] evaluated the long-term clinical outcomes of 412 HCC patients with PVT treated with TACE and radiation therapy. The radiotherapy volume included the PVT, with a 2- to 3-cm margin to cover the adjacent HCC. Intrahepatic primary HCC was managed by TACE before or after radiotherapy. Main or bilateral PVT was observed in 200 patients (48.5%). The median radiation dose was 40 Gy (range, 21 to 60 Gy) delivered in 2- to 5-Gy fractions. They found that 3.6% of the patients achieved a CR and 24.3% of the patients achieved a PR. The median patient survival was 10.6 months. Forty-one patients (10.0%) showed grade 3 to 4 hepatic toxicity during or 3 months after completion of radiotherapy. Grades 2 to 3 gastroduodenal complications were observed in 15 patients (3.6%).
These results suggest that combination treatment is feasible and tolerable. However, it remains unclear whether multimodal treatments have additive or synergistic efficacy benefits versus sorafenib alone.
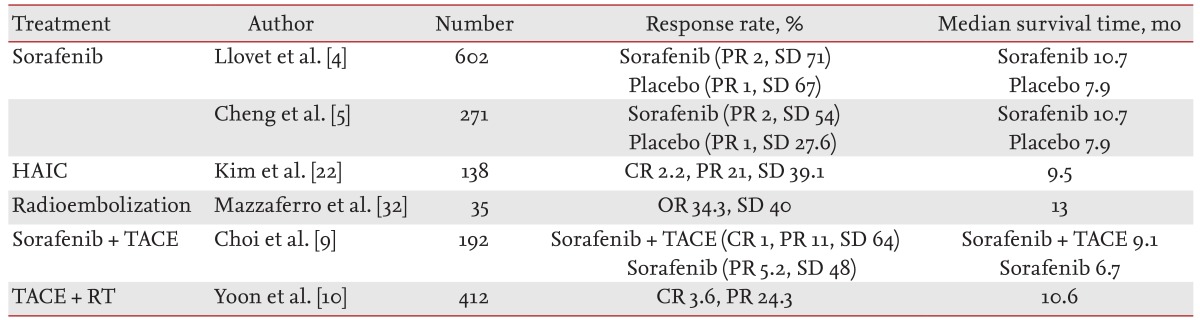
Sorafenib molecular-targeted therapy has become the standard treatment in advanced HCC, shifting the paradigm of systemic therapy for HCC to molecular-targeted therapies. Nevertheless, alternative therapies are required due to the low response rates and unsuitability of molecular agents. Clinical trials of HAIC, radioembolization, and multimodal treatments have shown favorable efficacy and tolerance in advanced HCC patients. Table 1 summarizes the clinical trials of treatments for advanced HCC [4,5,9,10,22,32].
The studies of novel therapies and multimodal treatments are limited by small sample sizes and a lack of large randomized trials. It will be necessary to compare sorafenib with other modalities in advanced HCC. Randomized trials comparing sorafenib should improve results and various treatment modalities might be a feasible, promising approach in the treatment of advanced HCC.
Acknowledgments
Supported by a National R&D Program Grant for Cancer Control from the Ministry of Health, Welfare, and Family Affairs, Republic of Korea (R0620390-1).
References
1. Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin 2005;55:74ŌĆō108PMID : 15761078.


2. Llovet JM, Burroughs A, Bruix J. Hepatocellular carcinoma. Lancet 2003;362:1907ŌĆō1917PMID : 14667750.


3. Bruix J, Sherman M, Practice Guidelines Committee, American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma. Hepatology 2005;42:1208ŌĆō1236PMID : 16250051.


4. Llovet JM, Ricci S, Mazzaferro V, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med 2008;359:378ŌĆō390PMID : 18650514.


5. Cheng AL, Guan Z, Chen Z, et al. Efficacy and safety of sorafenib in patients with advanced hepatocellular carcinoma according to baseline status: subset analyses of the phase III Sorafenib Asia-Pacific trial. Eur J Cancer 2012;48:1452ŌĆō1465PMID : 22240282.


6. Cheng AL, Kang YK, Chen Z, et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol 2009;10:25ŌĆō34PMID : 19095497.


7. Ganeshan A, Upponi S, Hon LQ, Warakaulle D, Uberoi R. Hepatic arterial infusion of chemotherapy: the role of diagnostic and interventional radiology. Ann Oncol 2008;19:847ŌĆō851PMID : 18029972.


8. Sangro B, Inarrairaegui M, Bilbao JI. Radioembolization for hepatocellular carcinoma. J Hepatol 2012;56:464ŌĆō473PMID : 21816126.


9. Choi GH, Shim JH, Kim MJ, et al. Sorafenib alone versus sorafenib combined with transarterial chemoembolization for advanced-stage hepatocellular carcinoma: results of propensity score analyses. Radiology 2013;269:603ŌĆō611PMID : 23864102.


10. Yoon SM, Lim YS, Won HJ, et al. Radiotherapy plus transarterial chemoembolization for hepatocellular carcinoma invading the portal vein: long-term patient outcomes. Int J Radiat Oncol Biol Phys 2012;82:2004ŌĆō2011PMID : 21621346.


11. Kudo M. Signaling pathway and molecular-targeted therapy for hepatocellular carcinoma. Dig Dis 2011;29:289ŌĆō302PMID : 21829020.


12. Wilhelm SM, Adnane L, Newell P, Villanueva A, Llovet JM, Lynch M. Preclinical overview of sorafenib, a multikinase inhibitor that targets both Raf and VEGF and PDGF receptor tyrosine kinase signaling. Mol Cancer Ther 2008;7:3129ŌĆō3140PMID : 18852116.


13. Omata M, Lesmana LA, Tateishi R, et al. Asian Pacific Association for the Study of the Liver consensus recommendations on hepatocellular carcinoma. Hepatol Int 2010;4:439ŌĆō474PMID : 20827404.



14. Lee HC. Systemic chemotherapy of hepatocellular carcinoma: Korean experience. Oncology 2008;75(Suppl 1):114ŌĆō118PMID : 19092280.


15. Furuse J, Ishii H, Nakachi K, Suzuki E, Shimizu S, Nakajima K. Phase I study of sorafenib in Japanese patients with hepatocellular carcinoma. Cancer Sci 2008;99:159ŌĆō165PMID : 17953709.


16. Kim HY, Kim JD, Bae SH, et al. A comparative study of high-dose hepatic arterial infusion chemotherapy and transarterial chemoembolization using doxorubicin for intractable, advanced hepatocellular carcinoma. Korean J Hepatol 2010;16:355ŌĆō361PMID : 21415578.



17. Ishikawa T, Imai M, Kamimura H, et al. Improved survival for hepatocellular carcinoma with portal vein tumor thrombosis treated by intra-arterial chemotherapy combining etoposide, carboplatin, epirubicin and pharmacokinetic modulating chemotherapy by 5-FU and enteric-coated tegafur/uracil: a pilot study. World J Gastroenterol 2007;13:5465ŌĆō5470PMID : 17907289.



18. Yoshikawa M, Ono N, Yodono H, Ichida T, Nakamura H. Phase II study of hepatic arterial infusion of a fine-powder formulation of cisplatin for advanced hepatocellular carcinoma. Hepatol Res 2008;38:474ŌĆō483PMID : 18430093.


19. Nagano H. Treatment of advanced hepatocellular carcinoma: intraarterial infusion chemotherapy combined with interferon. Oncology 2010;78(Suppl 1):142ŌĆō147PMID : 20616597.


20. Yamasaki T, Sakaida I. Hepatic arterial infusion chemotherapy for advanced hepatocellular carcinoma and future treatments for the poor responders. Hepatol Res 2012;42:340ŌĆō348PMID : 22151009.


21. Nishiyama M, Yamamoto W, Park JS, et al. Low-dose cisplatin and 5-fluorouracil in combination can repress increased gene expression of cellular resistance determinants to themselves. Clin Cancer Res 1999;5:2620ŌĆō2628PMID : 10499641.

22. Kim BK, Park JY, Choi HJ, et al. Long-term clinical outcomes of hepatic arterial infusion chemotherapy with cisplatin with or without 5-fluorouracil in locally advanced hepatocellular carcinoma. J Cancer Res Clin Oncol 2011;137:659ŌĆō667PMID : 20552225.


23. Itamoto T, Nakahara H, Tashiro H, et al. Hepatic arterial infusion of 5-fluorouracil and cisplatin for unresectable or recurrent hepatocellular carcinoma with tumor thrombus of the portal vein. J Surg Oncol 2002;80:143ŌĆō148PMID : 12115797.


24. Murata K, Shiraki K, Kawakita T, et al. Low-dose chemotherapy of cisplatin and 5-fluorouracil or doxorubicin via implanted fusion port for unresectable hepatocellular carcinoma. Anticancer Res 2003;23(2C):1719ŌĆō1722PMID : 12820447.

25. Woo HY, Bae SH, Park JY, et al. A randomized comparative study of high-dose and low-dose hepatic arterial infusion chemotherapy for intractable, advanced hepatocellular carcinoma. Cancer Chemother Pharmacol 2010;65:373ŌĆō382PMID : 19763572.


26. Strigari L, Sciuto R, Rea S, et al. Efficacy and toxicity related to treatment of hepatocellular carcinoma with 90Y-SIR spheres: radiobiologic considerations. J Nucl Med 2010;51:1377ŌĆō1385PMID : 20720056.


27. Ho S, Lau WY, Leung TW, Chan M, Johnson PJ, Li AK. Clinical evaluation of the partition model for estimating radiation doses from yttrium-90 microspheres in the treatment of hepatic cancer. Eur J Nucl Med 1997;24:293ŌĆō298PMID : 9143467.



28. Kao YH, Tan EH, Ng CE, Goh SW. Clinical implications of the body surface area method versus partition model dosimetry for yttrium-90 radioembolization using resin microspheres: a technical review. Ann Nucl Med 2011;25:455ŌĆō461PMID : 21643807.


29. Kennedy A, Nag S, Salem R, et al. Recommendations for radioembolization of hepatic malignancies using yttrium-90 microsphere brachytherapy: a consensus panel report from the radioembolization brachytherapy oncology consortium. Int J Radiat Oncol Biol Phys 2007;68:13ŌĆō23PMID : 17448867.


30. Wang SC, Bester L, Burnes JP, et al. Clinical care and technical recommendations for 90yttrium microsphere treatment of liver cancer. J Med Imaging Radiat Oncol 2010;54:178ŌĆō187PMID : 20598004.


31. Kulik LM, Carr BI, Mulcahy MF, et al. Safety and efficacy of 90Y radiotherapy for hepatocellular carcinoma with and without portal vein thrombosis. Hepatology 2008;47:71ŌĆō81PMID : 18027884.


32. Mazzaferro V, Sposito C, Bhoori S, et al. Yttrium-90 radioembolization for intermediate-advanced hepatocellular carcinoma: a phase 2 study. Hepatology 2013;57:1826ŌĆō1837PMID : 22911442.


33. Kim DY, Park BJ, Kim YH, et al. Radioembolization with Yttrium-90 resin microspheres in hepatocellular carcinoma: a multicenter prospective study. Am J Clin Oncol 2013;9. 21. [Epub]. http://dx.doi.org/10.1097/COC.0b013e3182a78dba.

34. Salem R, Lewandowski RJ, Mulcahy MF, et al. Radioembolization for hepatocellular carcinoma using Yttrium-90 microspheres: a comprehensive report of long-term outcomes. Gastroenterology 2010;138:52ŌĆō64PMID : 19766639.


35. Cabibbo G, Latteri F, Antonucci M, Craxi A. Multimodal approaches to the treatment of hepatocellular carcinoma. Nat Clin Pract Gastroenterol Hepatol 2009;6:159ŌĆō169PMID : 19190599.


36. Liao XF, Yi JL, Li XR, Deng W, Yang ZF, Tian G. Angiogenesis in rabbit hepatic tumor after transcatheter arterial embolization. World J Gastroenterol 2004;10:1885ŌĆō1889PMID : 15222029.



37. Shim JH, Park JW, Kim JH, et al. Association between increment of serum VEGF level and prognosis after transcatheter arterial chemoembolization in hepatocellular carcinoma patients. Cancer Sci 2008;99:2037ŌĆō2044PMID : 19016764.


38. Seong J, Park HC, Han KH, et al. Clinical results of 3-dimensional conformal radiotherapy combined with transarterial chemoembolization for hepatocellular carcinoma in the cirrhotic patients. Hepatol Res 2003;27:30ŌĆō35PMID : 12957204.


39. Park MS, Kim SU, Park JY, et al. Combination treatment of localized concurrent chemoradiation therapy and transarterial chemoembolization in locally advanced hepatocellular carcinoma with intrahepatic metastasis. Cancer Chemother Pharmacol 2013;71:165ŌĆō173PMID : 23079897.


40. Ingold JA, Reed GB, Kaplan HS, Bagshaw MA. Radiation hepatitis. Am J Roentgenol Radium Ther Nucl Med 1965;93:200ŌĆō208.

-
METRICS

- Related articles
-
Intussusception caused by small bowel metastasis from hepatocellular carcinoma2024 January;39(1)
Dermatomyositis associated with hepatitis B virus-related hepatocellular carcinoma2014 March;29(2)
Molecular targeted therapy for advanced gastric cancer2013 March;28(2)
Practical Treatments for Constipation in Korea2012 September;27(3)
Application of Helical Tomotherapy for Two Cases of Advanced Hepatocellular Carcinoma2011 June;26(2)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print


