 |
 |
| Korean J Intern Med > Volume 30(4); 2015 > Article |
|
Abstract
Background/Aims
Although pyogenic liver abscesses (PLAs) can be successfully treated, the visual prognosis of patients with endogenous endophthalmitis (EE) associated with a PLA is poor. Early diagnosis and prompt intervention may salvage useful vision. Therefore, we investigated risk factors for EE in patients with PLA, to facilitate early diagnosis.
Methods
Data from 626 patients diagnosed with PLA between January 2004 and July 2013 were analyzed retrospectively. Patients were divided into two groups: those with liver abscess-associated endogenous endophthalmitis (LAEE) and non-LAEE.
Results
The prevalence of EE in PLA patients was 1.92%. The mean age for all patients (373 males, 59.6%) was 62.8 years. Upon multivariate logistic regression, a liver abscess or another systemic infection (odds ratio [OR], 5.52; p = 0.005), an abscess in the right superior segment (OR, 5.26; p = 0.035), and Klebsiella pneumoniae infection (OR, 3.68; p = 0.039), were risk factors for LAEE. The final visual outcomes of patients with LAEE included no light perception in seven, hand motion only in three, and decreased visual acuity in two. Vitrectomy and early intravitreal injections of antibiotics improved visual acuity and preserved useful vision.
A pyogenic liver abscess (PLA) is a serious life-threatening condition, with a mortality rate of 6% to 14% [12]. With recent advances in diagnostic tools and treatment modalities, most cases are currently diagnosed at early stages and effectively treated. However, severe complications including endophthalmitis, metastatic central nervous system infections, psoas abscesses, splenic abscesses, abscess rupture, and septic pulmonary emboli may develop in some patients [3456].
Endogenous endophthalmitis (EE) is the most common and serious septic complication of PLA [78]; in a previous study, the reported incidence was 0.84% during 1 year of PLA follow-up [9]. EE is initially associated with subacute visual impairment, leading to loss of vision (blindness) despite aggressive treatment [8]. Factors suggested to be prognostic of visual outcomes in EE patients include initial visual acuity, early diagnosis, and vitrectomy [10]. Therefore, failure to make a timely diagnosis and to intervene at an early stage pose substantial risks to affected patients.
Little information on risk factors for EE in PLA patients is available. Diabetes mellitus (DM) and Klebsiella pneumoniae infection have been reported to be independent risk factors [8]. However, other studies have found that DM may not independently predict EE [911], and culture of K. pneumoniae may require a relatively long period of time. Therefore, we investigated risk factors for EE in PLA patients, to facilitate rapid diagnosis.
A total of 698 patients diagnosed with PLA were admitted to the Division of Gastroenterology of the Chonnam National University Hospital in Gwangju between January 2004 and July 2013; 72 patients were excluded because laboratory data were incomplete at the end of their follow-up periods, or PLA was difficult to distinguish from metastatic liver cancer, cholangiocarcinoma, or cystadenoma. Demographic information, laboratory data, imaging results, the treatment modalities chosen, microbiological results, and visual outcomes were retrospectively collected from medical records.
PLA was diagnosed using sonography, computed tomography, or both modalities. Radiographic reports were retrospectively reviewed to determine the location and number of abscesses. The anatomy of the affected liver segment was described using the Couinaud classification [12]. Multiple abscesses were diagnosed when more than one abscess was evident in either the same or different lobes. Systemic infection was defined as PLA with a sequela of sepsis or septic embolization to the spleen, kidney, brain, and/or lung. A systemic infection was diagnosed using laboratory and radiological data, and via culture of fluid from an involved site.
If EE was suspected, an ophthalmologic consultation was scheduled on the request of either a physician or a patient. The thorough ophthalmologic examination included assessment of visual acuity, slit-lamp biomicroscopy, measurement of intraocular pressure, indirect ophthalmoscopy, and ultrasonography if needed.
Blood and abscess cavity fluid were subjected to culture of aerobic and anaerobic organisms using standard techniques. All identified organisms were assumed to play etiological roles.
The primary therapeutic modality was either percutaneous drainage or open surgery. If neither of these procedures was performed, management featured (appropriate) antibiotic therapy only. Intravenous and intravitreal antibiotics were given on suspicion of EE. Pars plans vitrectomy and/or evisceration were performed at the discretion of treating ophthalmologists.
The Institutional Review Board of Chonnam National University Medical School approved the present study (approval no. CNUH-2014-030).
The liver abscess-associated endogenous endophthalmitis (LAEE) and non-LAEE groups were compared. Continuous variables (e.g., age) are expressed as means ┬▒ standard deviations. Appropriate statistical tests (Student t test, Pearson chi-square test, or the Mann-Whitney U test) were used to compare the baseline characteristics of patients in the two groups. Factors that were significant upon univariate logistic regression were entered into stepwise multivariate logistic regression to identify independently significant EE predictors. Statistical significance was accepted when the p < 0.05, or equivalently, when the 95% confidence interval (CI) of the odds ratio (OR) excluded unity. Statistical analyses were performed using SPSS version 20.0 (IBM Co., Armonk, NY, USA).
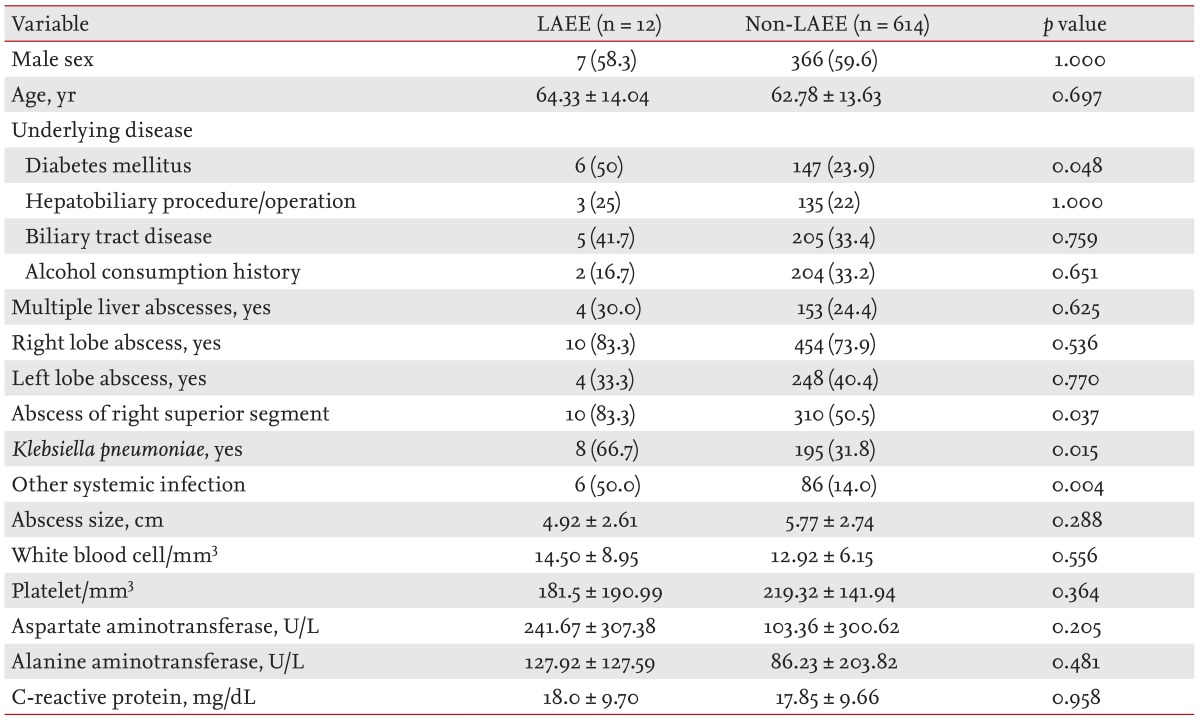
The characteristics of the 626 PLA patients (373 males, 59.6%; 253 females, 40.4%) are shown in Table 1. The mean patient age was 62.8 ┬▒ 13.6 years. Underlying conditions included DM in 153 patients (24.4%), a history of alcohol consumption in 206 (32.9%), biliary tract disease in 210 (33.5%), a previous hepatobiliary procedure/operation in 138 (22%), and another systemic infection in 92 (14.7%). These latter infections were sepsis (n = 51, 8%); and infections of the lung (n = 33, 5.2%), kidney (n = 12, 1.9%), brain (n = 2, 0.3%), abdominal cavity (n = 4, 0.6%), and spine (n = 1, 0.2%).
Of the 92 patients, eight had multiple metastatic infections (five at two metastatic sites and three at three such sites). Overall, 464 patients (74.1%) had right lobe liver abscesses, 252 had (40.3%) left lobe abscesses, and 157 (25.1%) had multiple liver abscesses. Of the 626 patients, 247 (39.5%) underwent percutaneous drainage, five (0.8%) underwent surgical drainage, and 374 (59.7%) received antibiotics only.
Of the 626 patients, 12 (1.92%) were diagnosed with LAEE. The proportions of patients with a systemic infection (p = 0.004), K. pneumoniae infection (p = 0.015), an abscess in the right superior segment (p = 0.037), and DM (p = 0.048) were all significantly higher in the LAEE group.
The most common initial symptoms were blurred vision and ocular pain (Table 2). Ocular symptoms developed prior to the diagnosis of liver abscesses in eight patients (66.7%). Initial visual problems were chemosis only in one patient, ocular pain in two, blurred vision in nine, and light perception only in one. The interval between EE diagnosis and intravitreal antibiotic injection/vitrectomy was less than 24 hours. Appropriate systemic and intravitreal antibiotics were prescribed for nine patients; the antibiotics included ceftazidime 2.25 mg/0.1 mL and vancomycin 1 mg/0.1 mL (Fig. 1). Pars plana vitrectomy was performed in three patients. Enucleation or evisceration was performed to relieve pain or infection in four. The final visual outcomes were no light perception in seven patients, hand motion only in three, and decreased visual acuity in two. Early intravitreal antibiotic injection or vitrectomy preserved useful vision. The details are shown in Table 2.
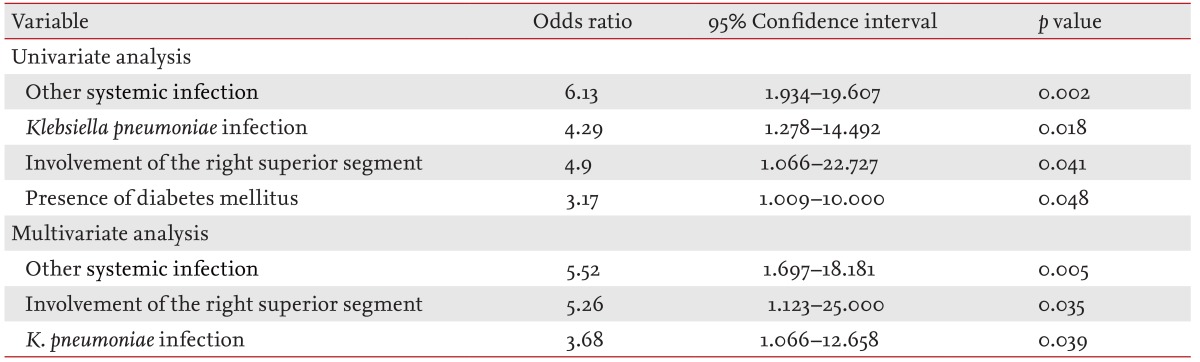
Univariate logistic regression showed that the presence of another systemic infection (OR, 6.13; p = 0.002), K. pneumoniae infection (OR, 4.29; p = 0.018), an abscess in the right superior segment (OR, 4.9; p = 0.041), and underlying DM (OR, 3.17; p = 0.048) were significant risk factors for EE in PLA patients.
Multivariate logistic regression confirmed that the presence of another systemic infection (OR, 5.52; p = 0.005), an abscess in the right superior segment (OR, 5.26; p = 0.035), and K. pneumoniae infection (OR, 3.68; p = 0.039) were significant risk factors for LAEE in PLA patients (Table 3).
The culture rate from abscess pockets of the non-LAEE group was 66.4% (146/220) and that of the LAEE group was 66.7% (6/9). The blood culture rate in the non-LAEE group was 30.4% (175/575) and that in the LAEE group was 27.3% (3/11).
K. pneumoniae, Streptococcus species, and Enterococcus species were the most common organisms in pus of both groups. Bacteremia was evident in 174 patients (27.8%). K. pneumoniae and Escherichia coli were the most commonly isolated organisms from both groups. The culture rate of K. pneumoniae from abscess pockets was 55.5% (121/218) and that from blood was 21.7% (125/575).
An important novel finding of the present study is that the presence of another systemic infection, an abscess in the right superior segment, and K. pneumoniae infection were all significantly associated with LAEE, facilitating early prediction of EE development and allowing the visual outcomes of patients with PLA to be partly preserved.
The prevalence of EE in PLA patients of the present study was 1.92%, comparable to the values of previous studies (0.84% to 6.9%) [69]. One large population-based study that explored the epidemiological association between PLA and EE found that about 0.84% of PLA patients developed EE during a 1-year follow-up period [9]. EE is generally uncommon, but is the most frequent serious septic complication of PLA [7], and the outcome is usually dismal. Despite early diagnosis of EE in PLA patients, visual outcomes are generally poor. However, useful vision can be preserved in patients treated with intravitreal antibiotic injections or vitrectomy. The alternative is legal blindness.
Consistent with the results of previous studies [1314], K. pneumoniae was the bacterium most commonly isolated from pus and blood. Fifty percent of patients with LAEE were infected with K. pneumoniae; endogenous K. pneumoniae endophthalmitis is the most common metastatic infection from a K. pneumoniae liver abscess [15], which has emerged as the most common pathogen of liver abscesses in Taiwan [413]. KLAs are becoming more common in other countries of southeast Asia, including Korea, Singapore, Japan, and Thailand, and they constitute an emerging infectious disease in the United States and elsewhere worldwide [1617]. The frequency of K. pneumoniae culture was lower in the present study than in previous works [18], but it is possible that some patients underwent antibiotic therapy at other facilities prior to transfer to our tertiary referral center.
DM and K. pneumoniae infection have been reported to be independent risk factors for the development of EE in PLA patients [8]. Consistent with the data of other studies, we found that DM and the K. pneumoniae infection rate were significantly higher in the LAEE group. Although DM and K. pneumoniae infection (in particular, infection by serotype K1) have previously been suggested to be risk factors for EE development in PLA patients, the detailed mechanisms remain unclear. The virulence of the K1 strain is enhanced by production of a mucoviscous exopolysaccharide web that exhibits hyper-mucoviscosity, high-level resistance to phagocytosis by macrophages and neutrophils, and resistance to complement deposition [1920]. In addition, diabetes interferes with chemotaxis by polymorphonuclear leukocytes and impairs phagocytosis of K. pneumoniae strains of capsular serotype K1 or K2 in patients with poor glycemic control [21]. However, although we found that K. pneumoniae infection was a risk factor for LAEE, as did previous studies [7], DM was not significantly associated with LAEE in the present study.
This is the first study to show that the presence of another systemic infection and an abscess in the right superior segment are significantly associated with LAEE. Such clinical features may aid in the early diagnosis of LAEE; unfortunately, K. pneumoniae cultures grow rather slowly. Systemic infection is considered characteristic of liver abscesses caused by K. pneumonias of serotype K1, and such abscesses are a well-known risk factor for EE [3]. It is possible that the observed systemic infections are propagated via the inferior vena cava (IVC); the right superior segment is anatomically close to the IVC and the hepatic vein of the right superior segment and the IVC are obtusely angled. Our results thus suggest that the risk of EE development should be considered in PLA patients with other systemic infections and abscesses of the right superior segment. Physicians should be alert to the possible development of EE when a PLA patient with these risk factors complains of ocular symptoms.
Our study had certain limitations. First, we retrospectively analyzed data collected in a single center; therefore, our results may be inapplicable to other patient populations. Second, our sample sizes were small, especially that of the LAEE group, making it difficult to effectively compare outcomes between the two groups. Third, we did not perform K. pneumoniae genotypic analysis (for example, we did not search for the K1 strain). Fourth, the frequency of K. pneumoniae culture in the present study was lower than those of previous studies. Larger prospective studies are required to confirm the translational utility and applicability of our findings.
In conclusion, the prevalence of EE in patients with PLA was 1.92%. PLA patients with other systemic infections, abscesses in the right superior segment, and K. pneumoniae infections require close monitoring and early intervention to treat LAEE. Prompt intervention such as intravitreal antibiotic injection or early vitrectomy may salvage useful vision.
1. The prevalence of endogenous endophthalmitis in patients with pyogenic liver abscesses is 1.92%.
2. The presence of another systemic infection, an abscess in the right superior segment, and Klebsiella pneumoniae infection were significant risk factors for the development of endogenous endophthalmitis in patients with pyogenic liver abscesses.
3. Prompt intervention including intravitreal antibiotic injections or early vitrectomy may salvage useful vision in patients with endogenous endophthalmitis.
Acknowledgments
This study was supported by grants from the Chonnam National University Hospital, Gwangju, Korea.
Conflict of Interest
Conflict of interest: No potential conflict of interest relevant to this article was reported.
References
1. Yu SC, Ho SS, Lau WY, et al. Treatment of pyogenic liver abscess: prospective randomized comparison of catheter drainage and needle aspiration. Hepatology 2004;39:932ŌĆō938PMID : 15057896.


2. Alvarez Perez JA, Gonzalez JJ, Baldonedo RF, et al. Clinical course, treatment, and multivariate analysis of risk factors for pyogenic liver abscess. Am J Surg 2001;181:177ŌĆō186PMID : 11425062.


3. Fang CT, Lai SY, Yi WC, Hsueh PR, Liu KL, Chang SC. Klebsiella pneumonia genotype K1: an emerging pathogen that causes septic ocular or central nervous system complications from pyogenic liver abscess. Clin Infect Dis 2007;45:284ŌĆō293PMID : 17599305.


4. Wang JH, Liu YC, Lee SS, et al. Primary liver abscess due to Klebsiella pneumoniae in Taiwan. Clin Infect Dis 1998;26:1434ŌĆō1438PMID : 9636876.


5. Chung DR, Lee SS, Lee HR, et al. Emerging invasive liver abscess caused by K1 serotype Klebsiella pneumoniae in Korea. J Infect 2007;54:578ŌĆō583PMID : 17175028.


6. Sheu SJ, Kung YH, Wu TT, Chang FP, Horng YH. Risk factors for endogenous endophthalmitis secondary to klebsiella pneumoniae liver abscess: 20-year experience in Southern Taiwan. Retina 2011;31:2026ŌĆō2031PMID : 21499189.


7. Chen YJ, Kuo HK, Wu PC, et al. A 10-year comparison of endogenous endophthalmitis outcomes: an east Asian experience with Klebsiella pneumoniae infection. Retina 2004;24:383ŌĆō390PMID : 15187660.


8. Yang CS, Tsai HY, Sung CS, Lin KH, Lee FL, Hsu WM. Endogenous Klebsiella endophthalmitis associated with pyogenic liver abscess. Ophthalmology 2007;114:876ŌĆō880PMID : 17467526.


9. Hu CC, Ho JD, Lou HY, Keller JJ, Lin HC. A one-year follow-up study on the incidence and risk of endophthalmitis after pyogenic liver abscess. Ophthalmology 2012;119:2358ŌĆō2363PMID : 22817832.


10. Lee S, Um T, Joe SG, et al. Changes in the clinical features and prognostic factors of endogenous endophthalmitis: fifteen years of clinical experience in Korea. Retina 2012;32:977ŌĆō984PMID : 22105504.


11. Sng CC, Jap A, Chan YH, Chee SP. Risk factors for endogenous Klebsiella endophthalmitis in patients with Klebsiella bacteraemia: a case-control study. Br J Ophthalmol 2008;92:673ŌĆō677PMID : 18245273.


12. Smith D, Downey D, Spouge A, Soney S. Sonographic demonstration of Couinaud's liver segments. J Ultrasound Med 1998;17:375ŌĆō381PMID : 9623474.


13. Yang CC, Chen CY, Lin XZ, Chang TT, Shin JS, Lin CY. Pyogenic liver abscess in Taiwan: emphasis on gas-forming liver abscess in diabetics. Am J Gastroenterol 1993;88:1911ŌĆō1915PMID : 8237941.

14. Lau YJ, Hu BS, Wu WL, Lin YH, Chang HY, Shi ZY. Identification of a major cluster of Klebsiella pneumoniae isolates from patients with liver abscess in Taiwan. J Clin Microbiol 2000;38:412ŌĆō414PMID : 10618128.




15. Fung CP, Chang FY, Lee SC, et al. A global emerging disease of Klebsiella pneumoniae liver abscess: is serotype K1 an important factor for complicated endophthalmitis? Gut 2002;50:420ŌĆō424PMID : 11839725.



16. Saccente M. Klebsiella pneumoniae liver abscess, endophthalmitis, and meningitis in a man with newly recognized diabetes mellitus. Clin Infect Dis 1999;29:1570ŌĆō1571PMID : 10585817.


17. Ohmori S, Shiraki K, Ito K, et al. Septic endophthalmitis and meningitis associated with Klebsiella pneumoniae liver abscess. Hepatol Res 2002;22:307ŌĆō312PMID : 11929716.


18. Rahimian J, Wilson T, Oram V, Holzman RS. Pyogenic liver abscess: recent trends in etiology and mortality. Clin Infect Dis 2004;39:1654ŌĆō1659PMID : 15578367.


19. Lin JC, Chang FY, Fung CP, et al. High prevalence of phagocytic-resistant capsular serotypes of Klebsiella pneumoniae in liver abscess. Microbes Infect 2004;6:1191ŌĆō1198PMID : 15488738.


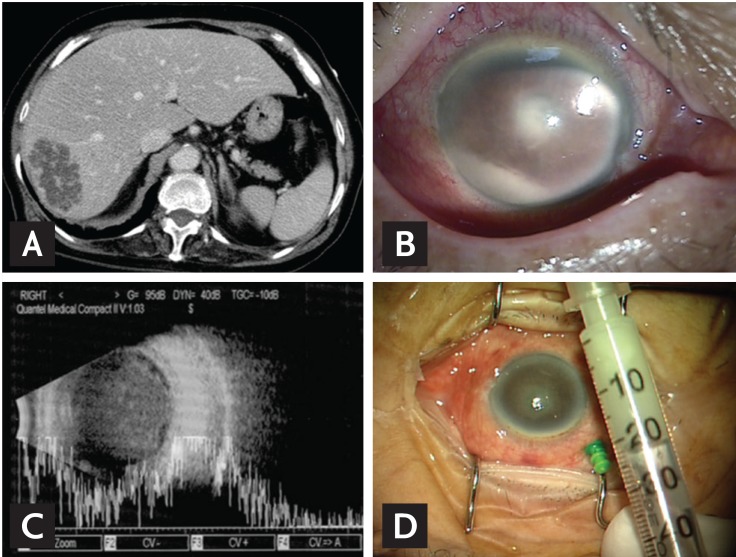
Figure┬Ā1
(A) Abdominal computed tomography revealing a 4.5 ├Ś 6.7 ├Ś 5.8 cm liver abscess in the right hepatic dome of a 73-year-old woman who presented with fever, chills, and blurred vision. (B) Image of an eye with endophthalmitis. Note pus accumulation in the anterior chamber (hypopyon) associated with a cloudy cornea. (C) A B-scan orbital ultrasonogram revealed a dense vitreous solution of high opacity. (D) Intravitreal tapping was performed and antibiotics were injected through a 25-G Trocar cannula.
Table┬Ā1
Clinical characteristics of patients with pyogenic liver abscesses alone, compared to those with both abscesses and liver abscess-associated endogenous endophthalmitis
Table┬Ā2
Management of and visual outcomes in patients with liver abscess-associated endogenous endophthalmitis
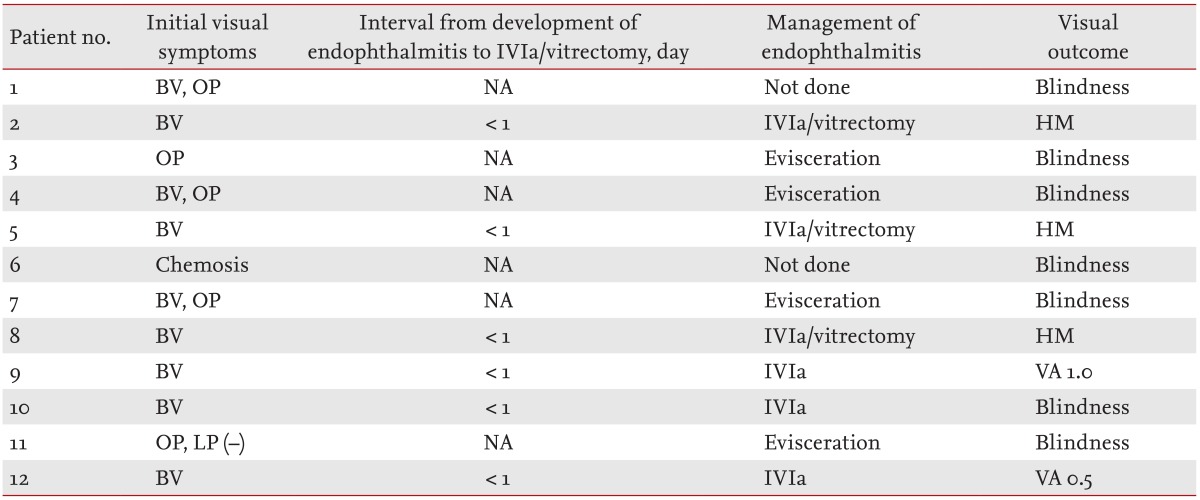
Patient no. 1 refused vitrectomy and evisceration. Patient no. 6 was in no condition to undergo aggressive ophthalmological treatment; the patient was intubated. Thus, these two patients received only antibiotic-containing eyedrops.
BV, blurred vision; OP, ocular pain; NA, not available; IVIa, intravitreal injection of antibiotics; HM, hand motion; VA, visual acuity; LP, light perception.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



