To the Editor,
The rectum can be affected by a variety of malignant and benign diseases: malignant diseases are rectal cancer, carcinoid tumor, and lymphoma, and benign diseases are polyps, solitary rectal ulcer syndrome, and other inflammatory diseases including pseudomembranous colitis, ulcerative colitis (UC), radiation proctitis, and lymphoid follicular proctitis (LFP). LFP is a very rare benign lymphoproliferative disease involving the rectum [1]. Among lymphoproliferative diseases, LFP is the rarest inflammatory disease, which was previously regarded as a form of UC, or can be misdiagnosed as a mucosa-associated lymphoid tissue (MALT) lymphoma [2]. In contrast to UC and lymphoma, its endoscopic and histopathologic findings are different from them: the endoscopic findings of LFP are usually multiple nodular, granular or congested mucosa without ulcerations, and histologic findings are characterized by lymphoid follicular hyperplasia without acute inflammatory infiltration. Here, we report the case of LFP accidently found during screening colonoscopy as an unusual form of a subepithelial tumor (SET)-like lesion resembling a carcinoid tumor.
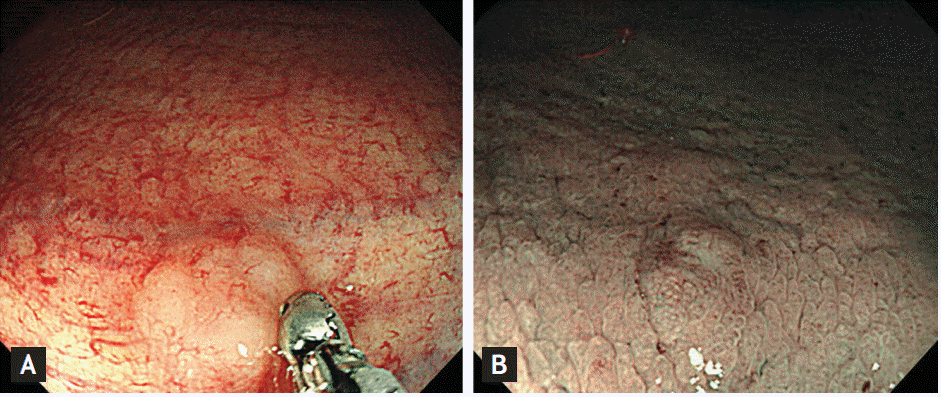
A 66-year-old woman underwent screening colonoscopy in our digestive disease center in Hanyang University Hospital. She was taking statins for dyslipidemia. Three years ago she had undergone gastroscopy and colonoscopy for screening, which was unremarkable except reflux esophagitis and atrophic gastritis. The present colonoscopy incidentally revealed a somewhat pale and whitish SET-like elevated lesion in the rectum. It was a hard, solitary sessile mass of about 5 mm in size at 10 cm from the anal verge (Fig. 1A). The overlying mucosa of the mass appeared normal, but there was neither cushion sign nor rolling sign, resembling a carcinoid tumor involving the submucosa being not ruled out. Also, in narrow band imaging (NBI) mucosal capillary network was arranged in a honeycomb pattern and there were no disorganized structure or increased density of micro-vessels, indicating similar NBI findings to carcinoid tumors (Fig. 1B).
This soliltary SET-like lesion in the rectum was considered a carcinoid tumor at first because of its SET-like appearance with a pale whitish surface, and was decided to be removed by the endoscopist for both diagnosis and treatment. After informed consent, the rectal lesion was completely resected by an endoscopic submucosal dissection technique without complications.
Histopathologic findings of the resected specimen included dense and diffuse lymphoid infiltration and confluent aggregates of large reactive lymphoid follicles in the mucosal and submucosa (Fig. 2A). Together with the lymphoid hyperplasia, reactive proliferation was evident in the germinal center while the mantle zone conserved in the submucosal layer. However, there were no findings of crypt abscess or granuloma. Also, the diffuse follicular lymphoid hyperplasia was immunohistochemically positive for CD20 (Fig. 2B), but negative for Bcl-2, a marker for lymphoma (Fig. 2C) and for synaptophysin, a marker for neuroendocrine tumor (Fig. 2D). Therefore, the final diagnosis was LFP. After establishing the diagnosis and completing the treatment, the patient was placed on a regular follow-up schedule without any symptoms.
A solitary elevated lesion in the rectum should be differentiated from benign lymphoproliferative diseases such as lymphoid polyps of the rectum and also malignant diseases such as carcinoid tumor and lymphoma. On the other hand, LFP should also be differentiated from non-neoplastic diseases such as distal UC, and allergic proctitis. For these differentiations, histopathologic findings are important: the histopathologic findings were prominent lymphoid follicular hyperplasia with reactive germinal center but conserved mantle zone. The histopathologic difference from UC was the absence of any crypt abscess and of neutrophil and eosinophil infiltration [2]. The difference from benign lymphoid polyp was the absence of well-demarcated proliferative lymphoid follicles in the surrounding tissue, and the difference from malignant lymphoma of the MALT type was the absence of malignant lymphoid cells and destructive follicular structure [3]. For histopathologic confirmation it is mandatory to obtain intact follicular structure of lymphoid tissues, but forceps biopsy alone may be insufficient because of small, crushed, and distorted tissues obtained [3]. Therefore, to accurately diagnose and differentiate LFP from other possible diseases involving the rectum, endoscopic resection is required, as performed in our case. On the other hand, endoscopic ultrasonography (EUS) must have been helpful in providing important information on this lesion. However, we performed ESD before EUS for pathologic examination and complete removal. A previous report described that EUS image in LFP showed rectal wall thickening limited to the mucosal and submucosal layers, compared with that of carcinoid tumor showing hypoechoic lesion in the deep mucosal layer.
What causes LFP has not been clearly defined. A previous report suggested a role of vascular or mechanical causes in its development showing diversion colitis (nonspecific inflammation of a dysfunctional segment after diversion of the fecal stream) after surgery due to UC as well as after surgery due to severe hemorrhoids and rectal prolapse [4]. However in our patient, as there was no surgery or vascular or mechanical injury, the cause of her LFP is unclear. Concerning symptoms of LFP, our patient did not have any associated symptoms or signs. According to a study of 20 patients with rectal biopsies showing LFP, its common symptoms are reported to be rectal bleeding and abdominal pain, while tenesmus and mucus discharge were also reported [2]. Most of the symptomatic cases showed multiple lesions with a congested granular appearance at endoscopy, whereas our patient had a small solitary lesion producing no symptoms, probably because there was only one rectal lesion.
Is treatment needed in patients with asymptomatic LFP? In cases with symptoms such as rectal bleeding or abdominal pain, treatment should be directed to alleviating or arresting these symptoms. These symptoms as well as endoscopic findings have been reported to respond to topical anti-inflammatory agents such as mesalamine or sulphasalazine, but systemic mesalamine and steroids have been found to be ineffective [5]. Although the natural course of LFP is unclear, it is possible that LFP presenting with a solitary lesion, as in our case, might progress to multiple nodular lesions and thereby produce symptoms such as rectal bleeding. In terms of treatment modality, alternative treatment modality include cap-assisted endoscopic mucosal resection in which the lesion is removed using a snare after it is suctioned into a cap and ligated by a band, as in rectal carcinoid tumor. Considering a benign nature of the lesion with small size confined to the mucosa and submucosa, this modality could completely remove the lesion.
In conclusion, this case concerns a very rare benign inflammatory disease of the rectum, LFP. It was presented as a nodular, SET-like lesion resembling a rectal carcinoid tumor without any symptoms, and was endoscopically removed for both diagnosis and treatment. Therefore, we show that endoscopic resection of the solitary LFP may be an effective and safe treatment as well as a diagnostic tool for confirmation. However, further studies are needed to establish the natural course of LFP as well as its clinical and endoscopic presentations.




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





