 |
 |
| Korean J Intern Med > Volume 33(1); 2018 > Article |
|
Abstract
Acknowledgments
Figure 1.
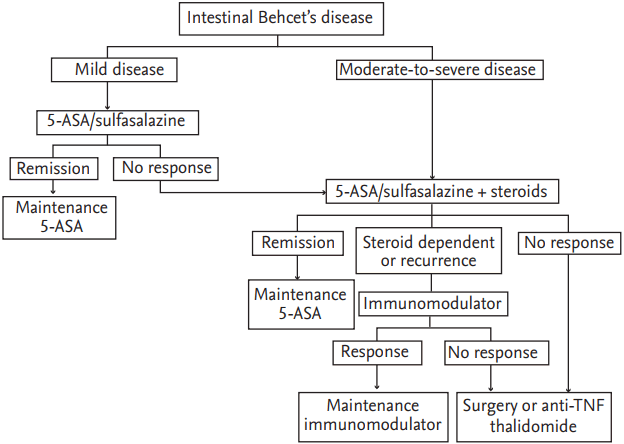
Table 1.
| Drugs | Study | Type of study | No. of patients or articles | Dose of medication | Indication/Outcomes |
|---|---|---|---|---|---|
| 5-ASA | Yoo et al. (1997) [16] | Case report | Total: 31 Sulfasalazine user: 14 | 1 g/day → 2–4 g/day → 1–2 g/day (maintenance) | Symptom improved: 79% |
| Sonta et al. (2000) [34] | Case report | Total: 1 | 1,200 mg/day | Lesion: esophageal ulcers | |
| Positive clinical efficacy | |||||
| Jung et al. (2012) [18] | Retrospective cohort study | Total: 292 | - | Maintaining remission; mild-to-moderate intestinal BD | |
| Clinical remission: 143 | |||||
| Hisamatsu et al. (2014) [13] | Japanese consensus statements | Relevant articles: 15 | 5-ASA: 2.25–3.00 g/day | Indication: maintenance therapy | |
| Sulfasalazine: 3–4 g/day | |||||
| Hatemi et al. (2016) [19] | Retrospective cohort study | Total: 60 | - | Remission: 10 of 16 patients (62.5%) | |
| 5-ASA user: 16 | Disease free duration: 89.3 ± 64.5 months | ||||
| Corticosteroids | Nakase et al. (2001) [20] | Case report | Total: 2 | Case 1: PD 30 mg → 40 mg → no improved: IV 2.5 mg dexamethasone every 2 weeks | Case 1: after IV corticosteroid use, oral, abdominal pain improved & inflammatory parameters: normalized |
| Case 2: PD 40 mg → 25 mg/ day → recur: IV 2.5 mg limethasone every 2 weeks | Case 2: after IV corticosteroid use, abdominal symptoms disappeared & inflammatory parameters: normalized | ||||
| Toda et al. (2002) [21] | Case report | Total: 1 | 1 g/day of IV methylpred- nisolone, 3 days → IV PD 40 mg/day → 20 mg PD IA injection | Disease remission | |
| Yasuo et al. (2003) [22] | Case report | Total: 1 | PD 0.5 mg/kg daily | Lesion: esophageal ulcer | |
| Symptom improved & esophagoscopy after 4 weeks: mucosa healing | |||||
| Park et al. (2010) [23] | Retrospective cohort study | Total: 54 | Median dosage: 0.58 mg/kg (range, 0.39–1.20) | Clinical remission: 25 (46.3%) | |
| 1 Year treatment response: 26 (48.1%) | |||||
| Cumulative probability of surgery: 17.5% at 1 year; 49.1% at 3 years | |||||
| Hisamatsu et al. (2014) [13] | Japanese consensus statements | Relevant articles: 15 | The initial dose of corticosteroids: 0.5–1 mg/kg/day for 1–2 weeks | Indication: induction therapy | |
| Clinical improvement → tapered by 5 mg every week & stopped | Severe symptoms (i.e., abdominal pain, diarrhea, GI bleeding) | ||||
| Immunomodulators | Choi et al. (2000) [24] | Retrospective cohort study | Total: 43 | AZA: 50 mg/day | Re-operation rates: decreased in AZA user group (7% vs. 25% at 2 years; 25% vs. 47% at 5 years) maintenance ther- apy in patients who require surgery |
| Remission: 16 (38%) | |||||
| Matsumura et al. (2010) [28] | Case report | Total: 1 | Oral tacrolimus: 10–12 ng/mL (induction) → 5–10 ng/mL (maintenance) | Refractory case to 5-ASA, steroid, immunomodulators, thalidomide, infliximab | |
| Remission: 33 months after starting tacrolimus | |||||
| Jung et al. (2012) [26] | Retrospective cohort study | Total: 272 | AZA: Initial dose 25 or 50 mg daily → 2.0–2.5 mg/kg (gradually increased every 2–4 weeks) | Indication: maintenance therapy | |
| AZA user: 67 (24.6%) | Remission: 39 (58.2%) of 67 (maintaining medically or surgically induced remission) | ||||
| 6-MP: Initial dose 0.5 → 1.0–1.5 mg/kg (gradually increased) | Clinical relapse: 13 (33.3%) of 39 | ||||
| Relapse risk factors: younger age (< 25 years) at diagnosis, lower hemoglobin level (< 11 g/dL) | |||||
| Hisamatsu et al. (2014) [13] | Japanese consensus statements | Relevant articles: 15 | AZA: Initial dose 25–50 mg/day | Indication: corticosteroid- dependent, corticosteroid- resistant, or anti-TNF-α mAb-resistant | |
| Lee et al. (2015) [25] | Retrospective cohort study | Total: 77 | AZA: 2–2.5 mg/kg/day | Postoperative recurrence rate: lower in patients who received post-operative thiopurines than 5-ASA user (p = 0.050) | |
| Thiopurine user: 27 (35.1%) | 6-MP: 1–1.5 mg/kg/day | ||||
| Park et al. (2015) [27] | Retrospective cohort study | Total: 196 (IBD) | AZA: 2–2.5 mg/kg/day | Cumulative relapse-free survival rate: higher in the leukopenic group than in the non-leukopenic group (p < 0.001) | |
| Intestinal BD: 83 | 6-MP: converted to an equivalent AZA dose by multiplying by 2.08 | ||||
| Hatemi et al. (2016) [19] | Retrospective cohort study | Total: 44 | AZA | Remission and no relapse: 24 (65%) | |
| Mean follow-up duration: 68.6 ± 43.6 months | |||||
| Park et al. (2017) (unpublished data) | Retrospective cohort study | Total: 10 | MTX: various dose (subcutaneously & oral) | Steroid free remission: 3 patients (30%) at 3 months, 4 patients (50%) at 6 months | |
| MTX monotherapy: 4 MTX + ADA: 6 | ADA: 160 mg in week 0, 80 mg in week 2, and 40 mg every other week | CRP levels: significantly decreased at 6 months com- pared with the baseline (p = 0.039). | |||
| Thalidomide | Sayarlioglu et al. (2004) [32] | Case report | Total: 1 | 100 mg/day | Lesion: recurrent intestinal perforation cecum, transverse colon, terminal ileum |
| Remission: 4 months follow-up | |||||
| Yasui et al. (2008) [31] | Case report | Total: 7 | Initial dose: 2 mg/kg/day → if necessary, 3 mg/kg/day (increase) or 1–0.5 mg/kg/ day (decreased) | Inclusion case: conventional therapy failure, severe steroid toxicity | |
| Clinical improvement: all patients | |||||
| Lee et al. (2010) [33] | Case report | Total: 4 | Case 1: 200 mg/day | Inclusion case: refractory to steroid, 5-ASA, immunomodulators | |
| Case 2: 100 mg/day | Clinical improvement: 3 of 4 | ||||
| Case 3: 100 mg/day | |||||
| Case 4: 50 → 100 mg/day | |||||
| IVIG | Beales (1998) [30] | Case report (letters) | Total: 1 | 400 mg/kg/day for 5 days | Lesion: oral, genital ulcer, uveitis, acneiform rash, arthralgia, intestine |
| Previous therapy: cyclosporine, thalidomide maintenance → IV methyl PD, oral mesala- mine add → no effect | |||||
| Clinical improvement: after 30 months | |||||
| Cantarini et al. (2016) [29] | Case report | Total: 4 | Case 3: 400 mg/kg/day for 5 days per month | Lesion: mucocutaneous, ocular, neurological, GI | |
| GI BD: 1 | Previous therapy: prednisone, cyclosporine, AZA, mesalazine | ||||
| Clinical improvement: after 14 months |
BD, Behçet’s disease; 5-ASA, 5-aminosalicylic acid; IVIG, intravenous immunoglobulin; PD, prednisolone; IV, intravenous; IA, intra-arterial; GI, gastrointestinal; AZA, azathioprine; 6-MP, 6-mercaptopurine; TNF-α, tumor necrosis factor α; mAb, monoclonal antibody; IBD, inflammatory bowel disease; MTX, methotrexate; ADA, adalimumab; CRP, C-reactive protein.
Table 2.
| Drugs | Study | Type of study | No. of patients | Dose of medication | Outcomes |
|---|---|---|---|---|---|
| Infliximab | Hassard et al. (2001) [53] | Case report | Total: 1 | 5 mg/kg, at 0, 2, 7, and 23 weeks | Disease activity: CDAI 270 (baseline) → 13 (at 2 weeks) |
| Remission: after 10 weeks | |||||
| Travis et al. (2001) [55] | Case report | Total: 2 | Case 1: 3 mg/kg (dose reduced because of recent sepsis) | Case 1: CRP 72 mg/L → 45 (on steroids) → 70 (sepsis) → 29 (on infliximab) → 12 (12 weeks later) | |
| Case 2: 5 mg/kg | Case 2: CRP 109 mg/L → 25 (on steroids) → 73 (recur) → 53→ < 6 (on infliximab) | ||||
| Kram et al. (2003) [61] | Case report | Total: 1 | 5 mg/kg dose over a 6-week period (3 infusions) | CRP 18.7 → normal (on infliximab) → 22.5 mg/mL (relapse) → normal (on infliximab + MTX) | |
| Lee et al. (2007) [67] | Case report | Total: 1 | 5 mg/kg | CRP: 70 → 6.7 mg/L (on infliximab) CDAI: 183 → 88 (on infliximab) | |
| HBI: 10 → 2 (on infliximab) | |||||
| Byeon et al. (2007) [56] | Case report | Total: 1 | 5 mg/kg | Lesion: unhealed anastomosis site, early recurrent ulcers after a distal ileocecectomy | |
| Remission: on endoscopy at 15 days after the infliximab infusion | |||||
| Ugras et al. (2008) [62] | Case report | Total: 1 | 5 mg/kg at weeks 0, 2, and 6 | Remission: 7 months | |
| Naganuma et al. (2008) [54] | Case report | Total: 6 | 5 mg/kg at 0, 2, 6 weeks (induction) & every 8 weeks (maintain) | Clinical remission: 4 of 6 (induction), 4 of 4 (maintain) | |
| Surgery: 2 of 6 (case 5-ileum ulceration, case 6-ileum ulceration with internal fistula) | |||||
| Maruyama et al. (2012) [63] | Case report | Total: 1 | 5 mg/kg at 0, 2, and 6 weeks (induction) & every 8 weeks (maintain) | Remission: 6 years | |
| Iwata et al. (2011) [44] | Retrospective study | Total: 10 | Infliximab (3 mg/kg at 0, 2, and 6 weeks & every 8 weeks) + MTX (3 mg/kg to 200 mg) | Short-term response: improved clinical symptoms & decreased ESR, CRP within 4 weeks | |
| Long-term response: remain remission at 6, 12, and 24 months | |||||
| Lee et al. (2013) [45] | Retrospective multicenter study | Total: 28 | 5 mg/kg at weeks 0,2, and 6 & every 8 weeks | Moderate to severe disease | |
| Clinical response: 2 weeks (75%), 4 (64.3%), 30 (50%), 54 (39.1%) | |||||
| Clinical remission: 2 (32.1%), 4 (28.6%), 30 (46.2%), 54 (39.1%) | |||||
| Biological response rate: 2 (82.1%), 4 (57.1%), 30 (53.8%), 54 (43.5%) | |||||
| Sustained response: 15 (53.6%) | |||||
| Kinoshita et al. (2013) [64] | Retrospective study | Total: 15 | 5 mg/kg at 0, 2 and 6 weeks & every 8 weeks | Short-term response at week 10: 12 of 15 (80%) | |
| Clinical remission: 8 of 15 (53%) | |||||
| Clinical response: 4 of 15 (27%) | |||||
| Long-term response after 12 months: 7 of 11 (64%) | |||||
| Clinical remission: 3 of 11 (27%) | |||||
| Clinical response: 4 of 11 (36%) | |||||
| After 24 months: 4 of 8 (50%) | |||||
| Clinical remission & response: 3 of 8 (38%) | |||||
| Ideguchi et al. (2014) [65] | Retrospective study | Total: 43 Infliximab user: 7 | - | 1 of 7: remission | |
| 2 of 7: insufficient efficacy | |||||
| 1 of 7: AE, sepsis | |||||
| 2 of 7: unchanged | |||||
| 1 of 7: for concurrent rheumatoid arthritis | |||||
| Hibi et al. (2016) [47] | Prospective, open-label, single-arm phase 3 study | Total: 18 Intestinal BD (11), NBD (3), VBD (4) | 5 mg/kg at weeks 0, 2, 6 (induction) & every 8 weeks (maintain) until week 46 | Complete responder: 61% (11 of 18) | |
| 10 mg/kg (patients who showed inad- equate responses after week 30) | Intestinal BD: improvement in clinical symptoms & decrease in CRP levels after week 2 | ||||
| ADA | De Cassan et al. (2011) [58] | Case report | Total: 2 | Induction dose of 160 mg subcutaneously 80 mg 2 weeks later | Intestinal BD |
| Maintenance: 40 mg every other week | Repeated steroid-dependent flares and failure of maintenance | ||||
| Clinical response, remission: 1-year follow-up | |||||
| Shimizu et al. (2012) [66] | Case report | Total: 1 | 40 mg every other week | Lesion: marginal ulcer after surgery (total gastrectomy) in stomach | |
| Improvement: after 3 months | |||||
| Tanida et al. (2015) [48] | Multicenter, open-label, uncontrolled study | Total: 20 | 160 mg at the start | Intestinal BD | |
| 80 mg at 2 weeks | Symptom relief & endoscopic assessment scores of 1 or lower at week 24 of treatment: 9 of 20 (45%) | ||||
| 40 mg, every other week & 80 mg for inadequate response/flare for 52 weeks | At week 52: 12 patients (60%) | ||||
| Tanida et al. (2016) [57] | Case report | Total: 8 | ADA: 160 mg in week 0, 80 mg in week 2, 40 mg every other week over 52 weeks | Refractory intestinal BD (failed to respond to conventional treatments) | |
| Primary outcome | |||||
| MI: at 10 weeks; 5 of 8 (62.5%); at 52 weeks; 6 of 8 (75%) | |||||
| Improvement of Global GI symptoms to score 0: at 10 weeks; 3 of 8 (37.5%); at 52 weeks; 4 of 8 (50%) | |||||
| Vitale et al. (2017) [59] | Multicenter retrospective observational study | Total: 100 | 40 mg subcutane- ously every 14 days (5 patients: initially treated with 40 mg every 7 days) | GI involvement: 47 of 100 | |
| At 12 weeks: ADA induced clinical efficacy in 81 patients | |||||
| At 24 months: 67 of 100 patients were still on ADA therapy | |||||
| Combination therapy with DMARDs: no significantly superior to monotherapy | |||||
| Inoue et al. (2017) [46] | Open label study following a phase 3 clinical trial | Total: 20 | Initiated at 160 mg; 2 weeks, 80 mg, followed by 40 mg every other week until the study end | Intestinal BD | |
| Study completion: 15 | Incidence of AEs through week 100; 544.4 events/100 person-years | ||||
| MI at weeks 52: 60.0% | |||||
| MI at weeks 100: 40.0% | |||||
| CR at weeks 52: 20.0% | |||||
| CR at weeks 100: 15.0% | |||||
| Etanercept | Ma et al. (2014) [49] | Retrospective comparative study based on observation | Total: 54 | Etanercept, subcutaneously at the dose of 25 mg twice a week | Intestinal BD |
| Conventional therapy: 35 | Primary outcome: 4 criteria for diagnosis of BD | ||||
| Etanercept: 19 | Remission rate, ulcer healing, recovery rate of ESR and CRP: significantly higher in Etanercept group than conventional group. | ||||
| Less adverse reactions in intestinal BD: etanercept group | |||||
| ANA/CAN | Cantarini et al. (2015) [50] | Case report | Total: 9 | ANA, 100 or 150 mg/day | GI involvement BD: 3 of 9 |
| With prednisone (5–25 mg/day) | On refractory to TNF blockers, standardized therapies | ||||
| Response to ANA: 8 of 9 | |||||
| Relapse: 8 of 9 | |||||
| Remain completely under control on ANA monotherapy: 1 patient | |||||
| Emmi et al. (2016) [60] | Retrospective study | Total: 30 | ANA, 100 mg/day, subcutaneously | GI involvement BD: 13% (4 of 30) | |
| ANA: 27 | CAN, 150 mg, subcutaneously every 6–8 weeks | CR at 12 months: 13 of 27 | |||
| CAN: 3 | Maintain therapy: 6 of 13 | ||||
| Maintained the same drug: 1 of 6 | |||||
| Shifted from ANA to CAN: 6 | |||||
| AE: ANA, 4 of 27 (15%); CAN, 0 of 27 | |||||
| Vitale et al. (2014) [51] | Case report | Total: 3 | CAN, 150 mg every 6 weeks | Case 1: GI involve (+), symptom-free, 6-month follow-up | |
| Case 2: GI involve (+), symptom-free, 12-month follow-up | |||||
| Case 3: GI involve (–), symptom-free, 6-month follow-up | |||||
| Tocilizumab | Deroux et al. (2015) [52] | Case report & literature review | Total: 15 | 8 mg/kg every 4 weeks or 2 weeks | Refractory BD |
| Case: 4 | GI involvement: 3 of 4 (case)/1 of 11 (literature) | ||||
| Literature review related case: 11 | In 4 cases: BD activity decreased significantly | ||||
| In literature review (11 previous cases): 8 of 11, improvement; 3 of 11, without efficacy |
BD, Behçet’s disease; TNF, tumor necrosis factor; CDAI, Crohn’s Disease Activity Index; CRP, C-reactive protein; MTX, methotrexate; HBI, Harvey-Bradshaw Index; ESR, erythrocyte sedimentation rate; AE, adverse event; BD, Behçet’s disease; NBD, neurological Behçet’s disease; VBD, vascular Behçet’s disease; ADA, adalimumab; GI, gastrointestinal; DMARD, disease modifying anti-rheumatic drug; MI, marked improvement; CR, complete remission; ANA, anakinra; CAN, canakinumab.
REFERENCES
-
METRICS

- Related articles
-
Korean treatment recommendations for patients with axial spondyloarthritis2024 January;39(1)
Korean treatment recommendations for patients with axial spondyloarthritis2023 September;38(5)
Novel treatments for inflammatory bowel disease2018 January;33(1)
Successful treatment with tacrolimus of refractory adult-onset Still’s disease2014 March;29(2)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print


