Delayed gastric emptying induced by glucagon-like peptide-1 receptor agonists and its implications for perioperative risk during anesthesia
Article information
Abstract
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) are increasingly prescribed for managing type 2 diabetes mellitus and obesity because of their glucose-lowering, weight-reducing, and cardiovascular benefits. However, their potential to delay gastric emptying has raised growing concern in perioperative care, particularly regarding the risk of pulmonary aspiration during general anesthesia. Recent studies have shown that patients treated with GLP-1 RAs may retain considerable gastric contents even after standard preoperative fasting, thereby increasing the likelihood of regurgitation and aspiration during anesthesia induction or emergence. Short-acting GLP-1 RAs cause more pronounced delays in gastric emptying, whereas long-acting agents may exert residual effects depending on dose and treatment duration. Several international anesthesia societies have issued updated guidance to mitigate these risks, yet available evidence remains limited and sometimes inconsistent. This review summarizes the pharmacological mechanisms and clinical implications of GLP-1 RA–induced delayed gastric emptying, evaluates current literature on perioperative aspiration risk, and emphasizes the importance of interdisciplinary collaboration between endocrinologists and anesthesiologists to ensure safe and individualized perioperative management.
INTRODUCTION
The recent development of diabetes medications with diverse mechanisms of action, such as glucagon-like peptide-1 receptor agonists (GLP-1 RAs) and sodium–glucose cotransporter–2 (SGLT–2) inhibitors, has shifted type 2 diabetes management from a glucocentric approach to one centered on complications and comorbidities. In particular, GLP-1 RAs, beyond their potent glucose-lowering effects, promote substantial weight loss by delaying gastric emptying and suppressing appetite through both the peripheral nervous system (PNS) and central nervous system (CNS). This broad pharmacologic profile not only ensures better glycemic control and improved cardiovascular outcomes but also establishes GLP-1 RAs as a leading therapeutic option for obesity management [1,2]. However, the gastrointestinal motility–inhibiting effects of GLP-1 RAs, particularly delayed gastric emptying, have raised concerns regarding pulmonary aspiration during general anesthesia or procedural sedation [3]. These concerns have sparked international debate about the adequacy of current standard preoperative fasting guidelines in the context of the gastrointestinal motor–depressant effects of GLP-1 RAs, leading to the issuance of updated clinical recommendations [4–8]. In Korea, the number of patients with type 2 diabetes and obesity treated with GLP-1 RAs has increased rapidly in recent years; nevertheless, case reports and studies addressing anesthesia-related precautions in this population remain scarce or have not been systematically analyzed. Despite the potentially fatal consequences of pulmonary aspiration during anesthesia or sedation, standardized guidelines have yet to be established, underscoring the need for close collaboration and consensus between endocrinologists and anesthesiologists.
This review comprehensively examines recent literature on the mechanisms of GLP-1 RA–induced delayed gastric emptying and its association with perioperative aspiration pneumonia. It also summarizes current international clinical guidelines and discusses practical considerations for safe and individualized perioperative management.
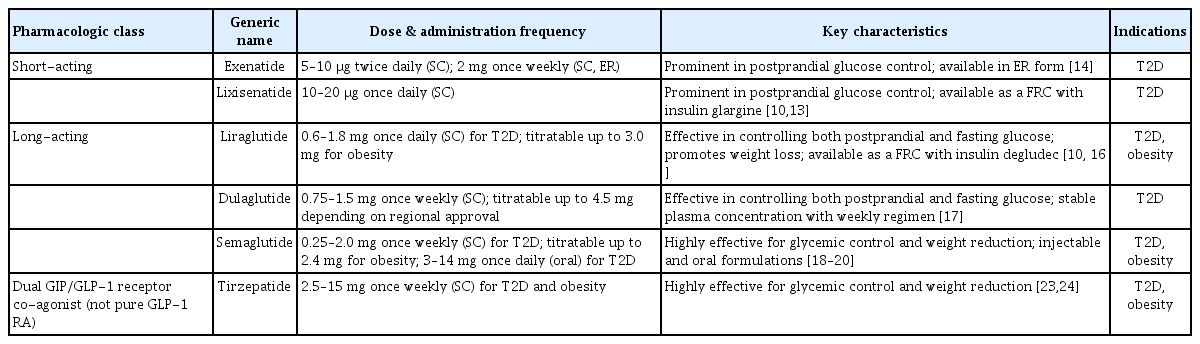
TYPES AND CHARACTERISTICS OF GLP-1 RAS
GLP-1 RAs are classified as either short-acting or long-acting, depending on the plasma drug concentration maintained between consecutive doses. Generally, a drug is considered short-acting if its blood concentration declines before the next dose and long-acting if its pharmacologic effects persist until the subsequent dose. This classification is closely linked to dosing frequency and differences in the degree of delayed gastric emptying [9]. Several extended formulations have also been developed, including fixed-ratio combination (FRC) agents that combine GLP-1 RAs with long-acting insulin or glucose-regulating peptides such as glucose-dependent insulinotropic polypeptide (GIP), which act as GLP-1 co-agonists [10,11] (Table 1).
Short-acting agents include exenatide, administered at 5–10 μg twice daily, and lixisenatide, administered at 10–20 μg once daily [12,13]. Notably, exenatide also has an extended-release formulation developed for 2 mg once weekly, whereas lixisenatide is available as an FRC with insulin glargine, a basal insulin [10,14]. These short-acting agents produce a rapid increase in plasma drug concentration shortly after administration and have a short half-life, allowing effective suppression of postprandial glucose excursions. Their principal mechanism involves a sustained delay in gastric emptying, which slows glucose absorption and attenuates the postprandial glycemic peak [15].
Long-acting agents include liraglutide, dulaglutide, and semaglutide. Liraglutide, the first GLP-1 RA approved by the U.S. Food and Drug Administration (FDA) for use in obese patients, is administered subcutaneously at 0.6–1.8 mg once daily for type 2 diabetes and up to 3.0 mg for obesity [16]. Furthermore, liraglutide is also available as a FRC with the basal insulin degludec [10]. Dulaglutide is administered subcutaneously at 0.75–1.5 mg once weekly, with doses up to 4.5 mg approved in some regions [17]. Semaglutide is available in both injectable and oral formulations. The injectable formulation is administered subcutaneously once weekly at doses of 0.25–2.0 mg for type 2 diabetes and up to 2.4 mg for obesity [18,19]. Oral semaglutide, the first approved oral GLP-1 RA, is administered once daily at 3–14 mg for type 2 diabetes [20]. Long-acting GLP-1 RAs are generally more effective in controlling fasting blood glucose than short-acting agents because they maintain stable plasma concentrations. Although their effect on delaying gastric emptying is less pronounced than that of short-acting agents, they exhibit consistent and sustained efficacy in improving glycemic control and promoting weight reduction [21].
Tirzepatide, a dual GLP-1 and GIP RA, is the first co-agonist targeting glucose-regulating peptides approved by the FDA for the treatment of both type 2 diabetes and obesity [22]. It is administered subcutaneously once weekly, beginning at 2.5 mg and titrated up to a maximum of 15 mg [23,24]. Moreover, several novel co-agonists are currently under development, including retatrutide, a triple GIP/GLP-1/glucagon receptor co-agonist; semaglutide combined with cagrilintide, an amylin agonist; and survodutide, a GLP-1 and glucagon co-agonist [25].
GASTRIC EMPTYING DELAY EFFECT OF GLP-1 RAS
GLP-1 RAs delay gastric emptying by inhibiting gastrointestinal motility through both PNS and CNS. Peripherally, as an incretin and enterogastrone secreted from the gut, GLP-1 activates GLP-1 receptors on afferent vagal fibers, initiating signals that inhibit gastrointestinal motility within the PNS [26]. More recently, a novel inter-organ neural circuit linking the intestine and stomach through sympathetic pathways has been proposed, in which GLP-1 stimulates intestinofugal neurons in the myenteric plexus, activating the celiac ganglion and postganglionic sympathetic nerves within the autonomic nervous system. This activation results in gastric distention and inhibition of gastric emptying [27]. Centrally, GLP-1 activates GLP-1 receptors located in the nucleus tractus solitarius and dorsal motor nucleus of the vagus within the brainstem, leading to inhibition of parasympathetic output and a reduction in gastric smooth muscle tone [2, 28]. Consequently, this promotes fundic relaxation, decreases peristalsis in the gastric antrum and duodenum, and increases pyloric sphincter tone, thereby delaying the transfer of gastric contents into the duodenum [29]. Nakatani et al. [30] evaluated gastrointestinal transit time using capsule endoscopy in patients with type 2 diabetes before and after liraglutide administration and reported delayed gastric emptying with reduced duodenal and small intestinal motility. Similarly, Sen et al. [31] performed preoperative gastric ultrasonography on 124 patients undergoing elective surgery under general anesthesia and found that those receiving once-weekly GLP-1 RAs (including semaglutide, dulaglutide, and tirzepatide) had a 30.5% higher incidence of increased gastric residual volume compared with the control group.
The effect of GLP-1 RAs on delaying gastric emptying varies according to the specific agent and duration of use. In general, short-acting agents produce a more pronounced delay in gastric emptying than long-acting agents [3, 15]. This difference is believed to result from tachyphylaxis associated with prolonged GLP-1 RA use, leading to attenuation of the gastric emptying–delaying effect, which becomes more evident with long-acting formulations [32,33]. In a study comparing gastric emptying time in patients with type 2 diabetes after 12 weeks of oral semaglutide treatment versus a control group, Dahl et al. [34] observed a significant delay in gastric emptying during the first postprandial hour in the semaglutide group (p = 0.005), but no difference after five hours. Similarly, Hjerpsted et al. [35] reported comparable findings in obese patients treated with subcutaneous semaglutide for 12 weeks; gastric emptying during the first hour was 27% slower in the semaglutide group than in the control group (p = 0.0012), but no significant difference was detected thereafter. However, both studies indirectly assessed gastric emptying through paracetamol absorption testing, a method limited by its inability to quantify the actual volume or consistency of gastric contents [36]. Therefore, the gastric emptying–delaying effect of GLP-1 RAs may be underestimated, warranting a more conservative approach in clinical practice.
PERIOPERATIVE RISKS AND CLINICAL CONSIDERATIONS IN PATIENTS USING GLP-1 RAS
Although the inhibitory effects of GLP-1 RAs on gastrointestinal motility and gastric emptying are advantageous for glycemic control, they present several clinical challenges that require careful consideration in the perioperative setting (Fig. 1). First, gastrointestinal side effects such as nausea, vomiting, and abdominal distension can increase the incidence of postoperative nausea and vomiting (PONV). The incidence of PONV is generally higher among patients receiving GLP-1 RAs than in the general surgical population. Shan et al. retrospectively analyzed 564 patients who underwent sleeve gastrectomy and found that the incidence of PONV was significantly higher among those treated with semaglutide or liraglutide compared with patients not receiving these agents [37]. These symptoms may extend beyond mild discomfort and lead to severe secondary complications, including surgical site bleeding and elevated intraocular or intracranial pressure, thereby warranting particular caution during ophthalmic or neurosurgical procedures [38,39].
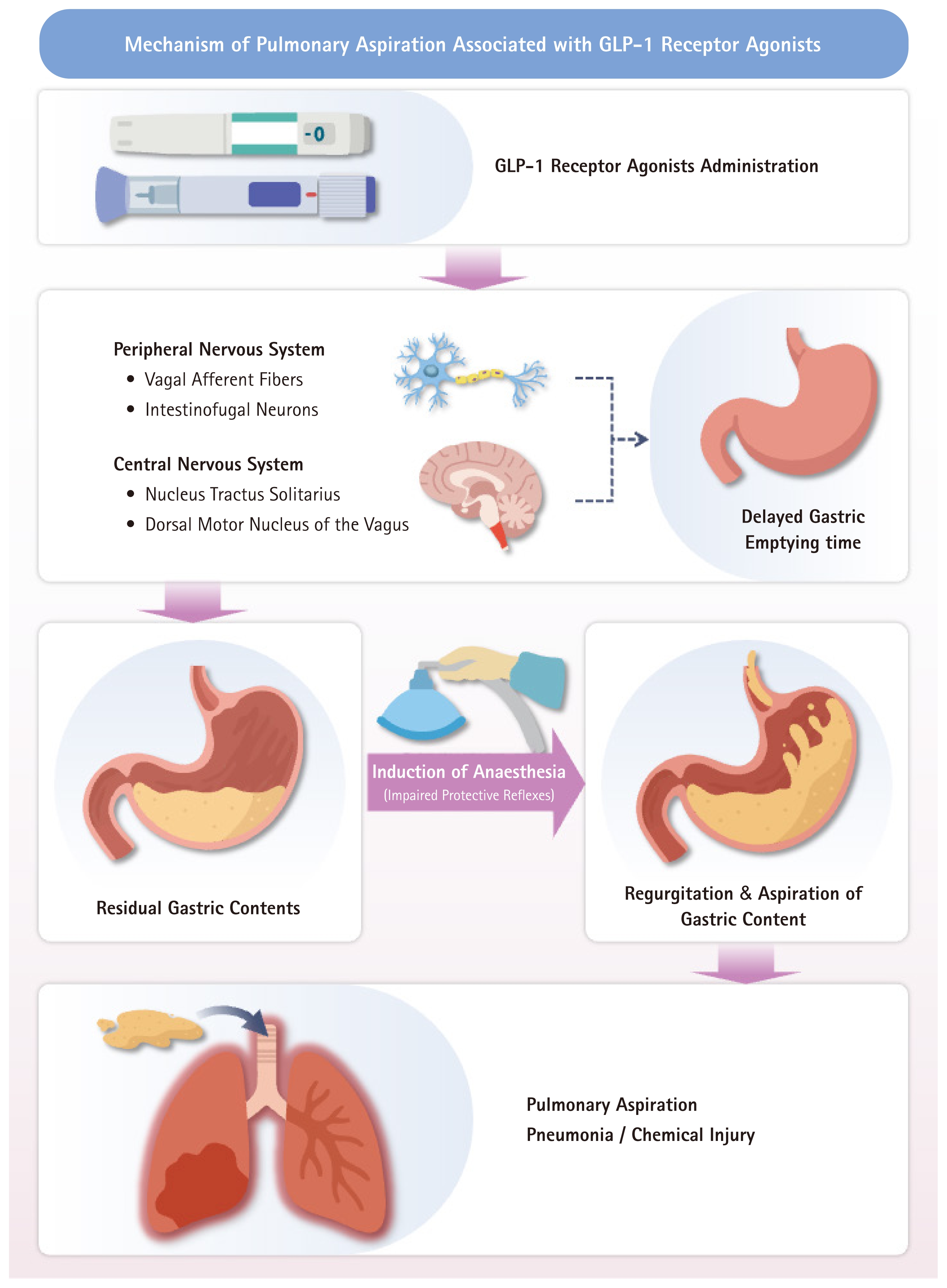
Proposed mechanism of delayed gastric emptying induced by GLP-1 receptor agonists and its implications for perioperative risk during anesthesia.
Second, delayed gastric emptying can alter the absorption of orally administered medications, resulting in unpredictable pharmacologic responses to premedication before anesthesia [40]. Premedication regimens typically include sedatives, anxiolytics, antihypertensives, and gastrointestinal motility–modifying agents. However, in patients receiving GLP-1 RAs, the absorption of these oral agents may be delayed or erratic, potentially leading to slower onset or reduced efficacy. This variability can directly influence the intended effects of premedication, including stable preoperative sedation, gastric acid suppression, and PONV prevention. To date, no direct pharmacokinetic studies have evaluated these effects in patients treated with GLP-1 RAs. Available evidence remains limited to the mechanism of delayed gastric emptying, without confirmed alterations in the absorption of specific premedication drugs. Therefore, in patients on GLP-1 RAs, any modification in the timing or route of premedication should be based on theoretical considerations and individualized clinical judgment.
Third, GLP-1 RAs can increase the risk of serious perioperative complications such as pulmonary aspiration by heightening the likelihood of regurgitation and aspiration of gastric contents during anesthesia. General anesthesia inherently increases the risk of aspiration because of a combination of factors, including diminished airway protective reflexes, decreased lower esophageal sphincter tone, and the use of positive pressure ventilation. This risk is particularly pronounced during tracheal intubation and extubation [41]. Because patients treated with GLP-1 RAs are more likely to retain gastric contents (RGC), the risk of aspiration may persist even when standard preoperative fasting guidelines are followed. Several studies have reported cases of regurgitation, vomiting, and aspiration in patients using GLP-1 RAs despite adherence to the fasting guidelines established by the American Society of Anesthesiologists (ASA), which recommend fasting for clear liquids up to two hours before surgery, light meals up to six hours before, and fatty meals up to eight hours before anesthesia induction [42]. For instance, Klein et al. [43] described a 42-year-old male patient receiving once-weekly semaglutide for weight loss who aspirated a substantial amount of solid and liquid gastric contents during anesthesia induction despite an 18-hour fasting period. Similarly, Gulak and Murphy [44] reported a 48-year-old female patient using semaglutide for weight loss who regurgitated and aspirated a large volume of liquid gastric contents during induction. Beam and Hunter Guevara [45] also documented a 50-year-old female patient treated with tirzepatide for obesity who vomited a large quantity of solid gastric contents immediately before extubation during anesthesia recovery. Similar adverse events have also been reported during other procedures, such as gastroscopy, resulting in procedural interruption [46]. Although these findings originate from individual case reports, they suggest a possible association between GLP-1 RAs and adverse perioperative events—including RGC and pulmonary aspiration—during general anesthesia, even with strict adherence to fasting guidelines.
While multiple studies have reported increased RGC among patients receiving GLP-1 RAs, both the optimal duration of drug discontinuation and whether elevated RGC—independent of other confounding factors—directly increases the risk of pulmonary aspiration remain subjects of debate. Sen et al. [31] observed a significantly higher incidence of increased RGC in patients treated with weekly GLP-1 RAs (semaglutide, dulaglutide, or tirzepatide) compared with control patients, with no correlation between the duration of drug discontinuation (up to seven days) and RGC levels. These findings indicate that temporarily withholding the medication for a standard period may be insufficient to mitigate the risk of retained gastric contents [31]. In a cohort study of patients undergoing upper gastrointestinal endoscopy, Alkabbani et al. [47] compared individuals with type 2 diabetes treated with various GLP-1 RAs (both short-acting and long-acting) with those receiving SGLT-2 inhibitors and found no significant increase in pulmonary aspiration risk. However, the rate of endoscopic procedure interruption was higher in patients treated with GLP-1 RAs, possibly due to increased gastric contents. Conversely, Yeo et al. [48], in a propensity score–matched cohort study including patients using a broad range of GLP-1 RAs, reported that GLP-1 RA therapy may be associated with an increased risk of aspiration pneumonia during endoscopic procedures.
Beyond the controversy surrounding the association between GLP-1 RAs and aspiration risk, perioperative risk assessment should also consider patients’ baseline glycemic control and diabetes-related complications [49]. Delayed gastric emptying is a well-recognized manifestation of diabetic gastrointestinal autonomic neuropathy and is more prevalent among individuals with long-standing or poorly controlled diabetes, independent of GLP-1 RA therapy [50]. Chronic hyperglycemia contributes to impaired gastric motility through autonomic and enteric neuronal dysfunction, and patients with microvascular complications have demonstrated a higher prevalence of gastroparesis and retained gastric contents [51,52]. Meanwhile, abrupt discontinuation of GLP-1 RAs during the perioperative period may worsen glycemic control, increase insulin requirements, and negate established cardiometabolic and renal benefits, potentially introducing additional perioperative risk [7]. Therefore, discontinuation decisions should be individualized by balancing aspiration risk against metabolic stability, taking into account glycemic control, diabetes duration, and the presence of diabetes-related complications rather than adhering to uniform discontinuation protocols.
Accordingly, gastrointestinal symptoms and delayed gastric emptying associated with GLP-1 RA therapy have important implications beyond glycemic control, serving as key factors in preoperative evaluation and surgical planning. Although several observational studies have examined these associations, their findings remain inconsistent. A recent systematic review and meta-analysis by Kamarajah et al. [53] evaluated 21 studies comprising 97,059 patients and found no definitive evidence of increased perioperative complications among preoperative GLP-1 RA users (pooled odds ratio 0.78, 95% confidence interval 0.59–1.05). However, the overall certainty of the evidence was rated as very low because of substantial heterogeneity and the predominance of observational data, underscoring the need for high-quality randomized controlled trials. Notably, determining the optimal discontinuation period remains challenging, as clinicians must weigh the therapeutic advantages of GLP-1 RAs—including improved glycemic control and cardiovascular and renal protective effects—against potential perioperative risks.
PREOPERATIVE RECOMMENDATIONS FOR GLP-1 RA MANAGEMENT
The ASA and the British Journal of Anaesthesia (BJA) published clinical recommendations in 2023 and 2024, respectively, addressing the perioperative management of patients treated with GLP-1 RAs [4,5]. In its 2023 consensus-based guidance, the ASA proposed a conservative approach, recommending preoperative discontinuation of GLP-1 RAs because of concerns regarding aspiration risk associated with delayed gastric emptying. Specifically, daily GLP-1 RAs should be withheld on the day of the procedure, whereas once-weekly formulations should be discontinued one week before surgery. For patients with diabetes, consultation with an endocrinologist was advised to facilitate bridging therapy. If gastrointestinal symptoms such as nausea, vomiting, or abdominal distension are present on the day of the procedure, postponement or reassessment of risk is recommended. If the medication was not withheld as advised or if symptoms persist, the patient should be considered non-fasting, and additional measures—such as preoperative gastric ultrasonography—should be undertaken. In emergency situations, patients should always be assumed to be non-fasting. However, concerns regarding potential deterioration of glycemic control prompted the ASA, in collaboration with four related societies, to issue updated guidance in 2024 [7]. These new recommendations advised that most patients continue GLP-1 RA therapy until surgery, except for those at high risk of gastrointestinal adverse effects. The ASA emphasized an individualized, risk-based approach, particularly for patients in the initial dose-escalation phase (typically 4–8 wk after initiating therapy) or those experiencing severe gastrointestinal symptoms.
The BJA, in contrast, advocates a more flexible and individualized management strategy. According to its analysis, the gastric emptying–delaying effect diminishes over time due to the development of tachyphylaxis with prolonged GLP-1 RAs use. Gastrointestinal motility may recover in patients who have received GLP-1 RAs for more than 12 weeks, thereby permitting adherence to standard preoperative fasting guidelines and allowing surgical suitability to be determined through clinical judgment. For short-term users (less than 12 wk), the BJA recommends basing perioperative decisions on the presence or absence of gastrointestinal symptoms because of insufficient reliable evidence. The statement also acknowledges the practical challenge of discontinuing once-weekly GLP-1 RA formulations, as four to five dosing intervals are typically required for complete drug clearance given their long half-lives. Erythromycin, although primarily an antibiotic, possesses motilin-mediated prokinetic properties that enhance gastric motility. Based on this mechanism, the BJA consensus statement suggests that erythromycin may be considered as an adjunctive measure for patients at high risk of GLP-1 RA–related delayed gastric emptying; however, the evidence supporting this use is limited, and administration should be undertaken with caution [5].
The most recent multidisciplinary consensus statement [54] recommends that patients without significant gastrointestinal symptoms—defined as severe nausea, vomiting, or intolerance to oral intake—should continue GLP-1 RA therapy regardless of the specific agent, with modifications to fasting protocols. Solid foods should be discontinued 24 hours before surgery, high-carbohydrate clear liquids (≥ 10% glucose) permitted up to 8 hours before surgery, and low-carbohydrate clear liquids (< 10% glucose) permitted up to 4 hours before surgery. The consensus underscores that ensuring an adequate fasting duration is paramount. Patients with significant gastrointestinal symptoms should receive individualized management, and in emergency procedures, all patients should be treated as having a full stomach.
In summary, across recommendations from multiple societies [4,5,6,7, 54,55] (Table 2), the initial guidance for preoperative management of GLP-1 RAs advocated a conservative strategy, recommending temporary discontinuation for safety considerations. However, recent studies have reported no significant differences in respiratory complications between users and non-users of GLP-1 RAs, indicating the need for continued discussion and evidence refinement [56,57]. Current preoperative assessment for patients treated with GLP-1 RAs emphasizes individualized evaluation based on the route of administration, duration of therapy, and presence of gastrointestinal symptoms, suggesting that tailored management according to patient-specific risk is more appropriate than a uniform prolongation of fasting periods.
NECESSITY FOR COLLABORATION BETWEEN ENDOCRINOLOGY AND ANESTHESIOLOGY AND THE DEVELOPMENT OF STANDARDIZED MANAGEMENT GUIDELINES
Currently, no official guidelines for the preoperative management of patients treated with GLP-1 RAs—based on collaboration and consensus between endocrinology and anesthesiology—have been established in Korea. Therefore, international recommendations must be contextualized and adapted to the Korean clinical environment. Close interdisciplinary collaboration between endocrinologists and anesthesiologists is essential for the safe surgical management of patients receiving GLP-1 RAs. First, these patients frequently present with endocrine–metabolic disorders such as diabetes or obesity, making perioperative glycemic regulation and metabolic optimization critical. Second, the delayed gastric emptying effect of GLP-1 RAs constitutes a potential anesthetic risk factor, requiring careful preoperative assessment and preparation by anesthesiology teams. Consequently, multiple specialty-specific considerations converge for each patient, making it challenging to achieve optimal perioperative outcomes through the efforts of a single department alone. A noteworthy example of successful interdisciplinary collaboration is demonstrated by Duke University Medical Center, where diabetic patients with glycated hemoglobin levels exceeding 7.5% achieved improved perioperative glycemic control and reduced surgical complications while awaiting surgery [58]. This case illustrates that a standardized, multidisciplinary care framework can enhance the quality of perioperative management and could be effectively extended to patients treated with GLP-1 RAs.
A coordinated perioperative strategy is essential for patients treated with GLP-1 RAs. First, a systematic preoperative screening process to confirm GLP-1 RA use is crucial. Because many patients are prescribed GLP-1 RAs not only for diabetes but also for obesity, these agents should be explicitly listed in preoperative questionnaires and electronic medical records to prevent omission. Furthermore, patients should be educated at the time of prescription to inform healthcare personnel of their GLP-1 RA use before surgery.
Next, individual patient risk must be assessed. Particular attention should be given to factors that increase the likelihood of delayed gastric emptying. Such risk factors include early treatment phase, persistent gastrointestinal symptoms such as nausea or vomiting, and comorbid conditions such as diabetic gastroparesis or Parkinson’s disease. When these factors are present, patients should be classified as high risk for aspiration during anesthesia; otherwise, they can be categorized as low risk. Additionally, both the urgency and the type of surgery (e.g., gastrointestinal versus non-gastrointestinal procedures) should be evaluated. Adjustments to surgical timing and method, as well as implementation of additional preventive measures, are warranted for high-risk patients. For elective procedures, surgery should be postponed to allow sufficient time for patients early in their GLP-1 RA treatment course. Dietary modification to liquid or clear fluids 24 hours before surgery is also recommended to minimize gastric contents. Furthermore, when available, preoperative gastric ultrasonography should be performed to evaluate gastric contents. If a substantial amount of solid material is detected, surgery should be delayed or interventions such as gastric decompression should be considered. For low-risk patients, surgery may proceed according to standard fasting guidelines.
A critical element in developing clinical guidelines is tailoring recommendations to the characteristics of Korean patients. Since the publication of the ASA consensus guidance in 2023, multiple international societies have released perioperative management recommendations for patients treated with GLP-1 RAs; however, large-scale randomized controlled trials remain lacking. Because GLP-1 RAs differ in half-life and in their effects on gastric emptying across agents, the degree of tachyphylaxis and severity of gastrointestinal symptoms may vary among individuals. Therefore, it is necessary to establish personalized discontinuation schedules and fasting protocols that reflect the pharmacologic properties of each drug, duration of therapy, and gastrointestinal status of individual patients. Future studies that stratify patients by drug class and clinical characteristics will be essential to support the development of context-appropriate guidelines for Korean clinical practice.
Beyond preoperative assessment, further research is required to develop standardized anesthesia protocols tailored to Korean clinical contexts. The Canadian Anesthesiologists’ Society (CAS), in its 2023 safety alert, recommended that patients receiving semaglutide be considered non-fasting even after adequate fasting [6]. The guidance further advised assessment for residual gastric contents using preoperative gastric ultrasonography, avoidance of general anesthesia or deep sedation when possible, and implementation of rapid sequence induction if such procedures are unavoidable. Additional precautions—including intraoperative gastric suction and delayed extubation until airway protective reflexes are secured—were also recommended. Because strategies for managing pulmonary aspiration during general anesthesia or deep sedation have been well described even before the widespread use of GLP-1 RAs, adapting international protocols to national clinical settings is a reasonable and appropriate approach.
CONCLUSION
The rapid rise in the use of GLP-1 RAs has brought perioperative management of these agents to the forefront as a critical clinical concern. Current recommendations from the United States and the United Kingdom emphasize a differentiated approach based on treatment duration and individual symptom profiles. However, standard fasting guidelines alone are often inadequate to prevent delayed gastric emptying and aspiration risk in all patients, thereby necessitating objective evaluations such as preoperative gastric ultrasonography. In Korea, where official national guidelines are not yet established, there is an urgent need to develop patient screening tools, standardized perioperative management frameworks, and anesthesia protocols tailored to local clinical practice while referencing international recommendations. Furthermore, establishing an integrated collaborative system between endocrinology and anesthesiology, along with improving patient education on preoperative medication disclosure, is essential. In the future, large-scale studies and sustained multidisciplinary collaboration are expected to facilitate the creation of effective, evidence-based management strategies and national guidelines.
Notes
CRedit authorship contributions
Donghee Kang: conceptualization, methodology, investigation, writing - original draft, project administration; Dughyun Choi: conceptualization, writing - review & editing, visualization, project administration, funding acquisition
Conflicts of interest
The authors disclose no conflicts.
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (No. NRF-2021R1G1A1009254). This work was supported by the Soonchunhyang University Research Fund.