Incidence and risk factors of rebleeding in Crohn’s disease patients with acute lower gastrointestinal bleeding
Article information
Abstract
Background/Aims
Acute lower gastrointestinal bleeding (LGIB) in Crohn’s disease (CD) is a rare but potentially life-threatening complication. This study aimed to investigate the incidence and risk factors of rebleeding in CD patients with LGIB.
Methods
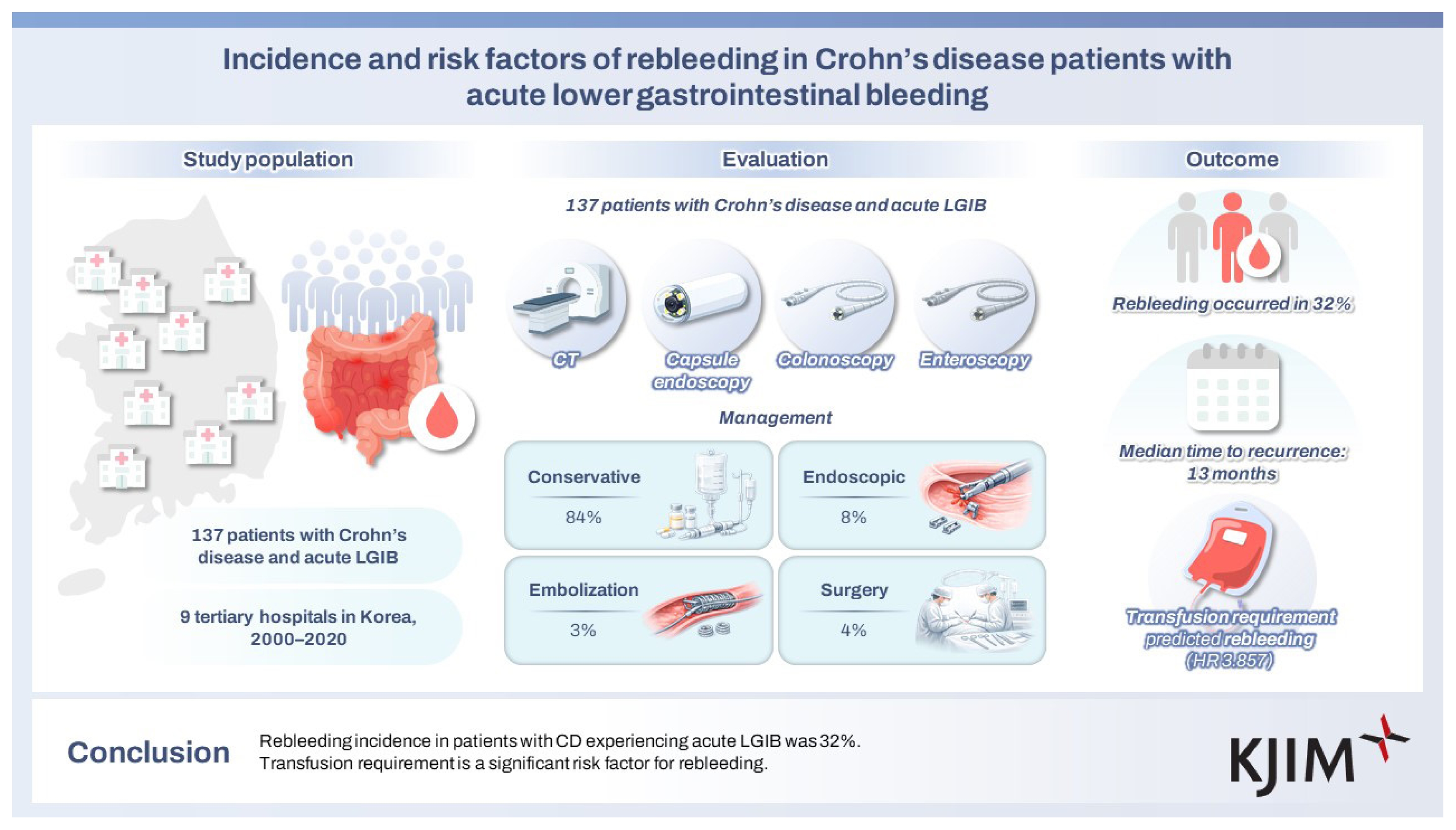
We retrospectively analyzed 137 CD patients with acute LGIB from nine tertiary university hospitals in Korea from January 2000 to October 2020. Patient characteristics, disease features, and treatment modalities were assessed, and rebleeding incidence, clinical features, and risk factors were analyzed.
Results
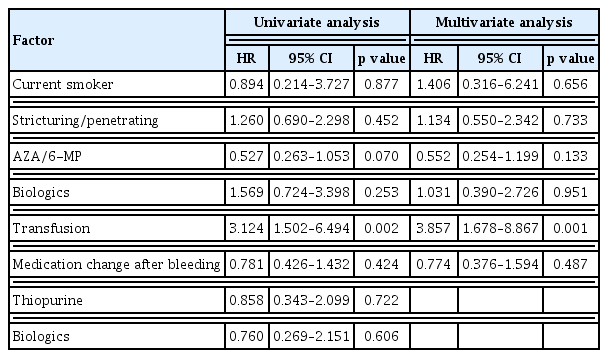
The median age of patients was 26.5 years, and the male-to-female ratio was 3.72:1. The median duration from diagnosis to first bleeding was 30.6 (0–243) months. Ileocolonic involvement (72 patients, 53%) and inflammatory phenotype (85, 62%) were most prevalent. During index bleeding, 20 and 40 patients were receiving biologics and azathioprine/6-mercaptopurine (AZA/6-MP), respectively. Bleeding focus was confirmed in 58 patients, primarily with endoscopy (33 cases). Management included conservative therapy (115 cases, 84%), endoscopic treatment (11, 8%), embolization (5, 3%), and surgery (6, 4%). After the index bleeding, 77 (56.2%) patients started or changed AZA/6-MP or biologics. Rebleeding occurred in 44 (32%) patients. The median time to recurrence was 13.0 (0.1–209) months. Transfusion requirement was significantly associated with rebleeding in univariate (hazard ratio [HR] = 3.124, 95% confidence interval [CI] = 1.502–6.494; p = 0.002) and multivariate (HR = 3.857, 95% CI = 1.678–8.867; p = 0.001) analyses.
Conclusions
Rebleeding incidence in patients with CD experiencing acute LGIB was 32%. Transfusion requirement is a significant risk factor for rebleeding.
INTRODUCTION
Acute severe lower gastrointestinal bleeding (LGIB) is a rare complication of Crohn’s disease (CD) affecting 0.6%–6.0% of patients [1–4]. However, it remains a challenging problem due to high recurrence (21.4–41.4%) [2,3, 5–7], surgery (7.1–39.7%) [2,3, 5–7], and mortality (0–8.2%) rates [3,5]. Further, it is one of the most difficult complications because the precise bleeding site is challenging to locate and treat. Studies regarding acute severe LGIB in CD are lacking, and most of the reported studies are limited due to a small sample size. According to domestic data, bleeding occurred in 5% of patients with CD, and the overall risk of rebleeding was 33.0% after 1 year and 72.3% after 10 years [2]. Regarding risk factors, a previous study indicated that early thiopurine administration and female sex may reduce the risk of acute LGIB [8]. Another study revealed that azathioprine/6-mercaptopurine (AZA/6-MP) treatment may reduce the risk of acute LGIB [2]. Regarding rebleeding, several studies have indicated that anti-tumor necrosis factor (anti-TNF) treatment may reduce the risk of rebleeding compared with conventional medical therapy (CMT) [2, 9]. Acute severe LGIB is an important clinical problem in patients with CD; however, only a few studies have focused on the prevalence, clinical features, outcomes, and risk factors of rebleeding. Hence, we aimed to investigate the clinical features, treatments, outcomes, incidence, and risk factors of rebleeding in patients with CD presenting with LGIB.
METHODS
Patients and data collection
This study retrospectively reviewed the medical records of 137 adult patients with CD presenting with symptomatic acute LGIB in nine tertiary university hospitals in Korea from January 2000 to October 2020. Patient features, disease characteristics, diagnostic methods, and treatment modalities were evaluated. Further, rebleeding incidence, clinical features, and risk factors were analyzed.
The Institutional Review Board of Yeungnam University Hospital (2020-10-006) and all participating hospitals approved this study.
Definitions and classifications
CD was diagnosed based on conventional clinical, radiological, endoscopic, and histopathological criteria [9]. Disease location and behavior were defined following the Montreal classification [10].
Acute severe LGIB was defined as acute overt rectal bleeding that caused (1) an abrupt decrease in the hemoglobin level to < 9 g/dL [11] or at least 2 g/dL below the baseline [7] and/or (2) transfusion of at least two units of blood within 24 hours [4]. The ligament of Treitz was considered the anatomic landmark that distinguished LGIB from upper GI bleeding. This study excluded patients with (1) upper GI bleeding, (2) anal lesions, (3) bleeding within 30 days postoperatively, and (4) no evidence of symptomatic bleeding.
The bleeding site was confirmed when the exact bleeding site was detected using computed tomography (CT), colonoscopy, angiography, radionuclide bleeding scan, capsule endoscopy, or enteroscopy. The identification of the lesion was defined as follows: on CT, the presence of extravasation or a neoplastic lesion; and on colonoscopy, enteroscopy, and capsule endoscopy, the presence of exposed vessels, oozing, spurting, or localized areas with adherent clot. Therapeutic modalities were classified as pharmacological treatment, including hemostatics, endoscopic treatment (injection, cauterization, and clipping), radiologic embolization, or surgery.
Rebleeding was defined as the occurrence of overt bleeding that met the criteria for acute severe LGIB after discharge.
Statistical analyses
The frequency of rebleeding was calculated as the percentage of patients experiencing bleeding that had recurred at least once after discharge.
Categorical variables are presented as numbers with percentages, whereas continuous variables are expressed as medians with ranges. Clinical features were compared between the rebleeding and no-rebleeding groups. Continuous variables are presented as mean and standard deviation, with an independent two-sample test or the Wilcoxon rank sum test used for comparison. The chi-square or Fisher’s exact test was used to analyze categorical variables. To identify rebleeding predictors, Kaplan–Meier survival analysis was conducted to compare the time to rebleeding, and the log-rank test was used to compare survival curves between groups according to key clinical variables. Further, Cox proportional hazards regression analysis was conducted to identify independent risk factors for rebleeding. For all tests, a p-value of < 0.05 indicated statistical significance. IBM Statistical Package for the Social Sciences Statistics version 21 (IBM Corp., Armonk, NY, USA) was used for statistical analyses.
RESULTS
Baseline and clinical characteristics of patients
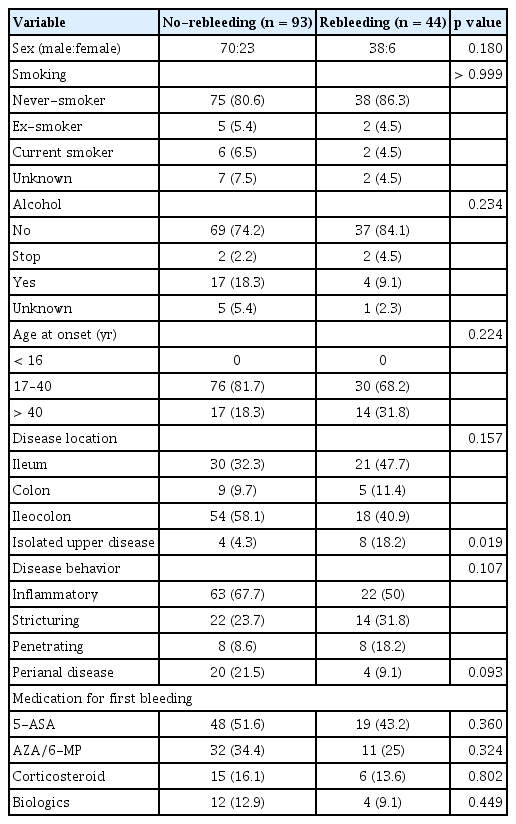
The median age of the patients was 26.5 years (range, 18–79 yr), and the male-to-female ratio was 3.72:1. The median disease duration was 30.6 months (range, 0–243 mo). Based on the Montreal classification, the most common disease location was ileocolonic in 72 (53%) patients. Inflammatory phenotype was most common in 85 (62%) patients. Mean CD activity index score at index bleeding was 245.8. Biologics were administered to 20 patients, whereas 40 patients were receiving AZA/6-MP. Isolated upper diseases were significantly more prevalent in rebleeding groups (p = 0.019) (Table 1).
Clinical manifestations, treatments, and outcomes of index bleeding
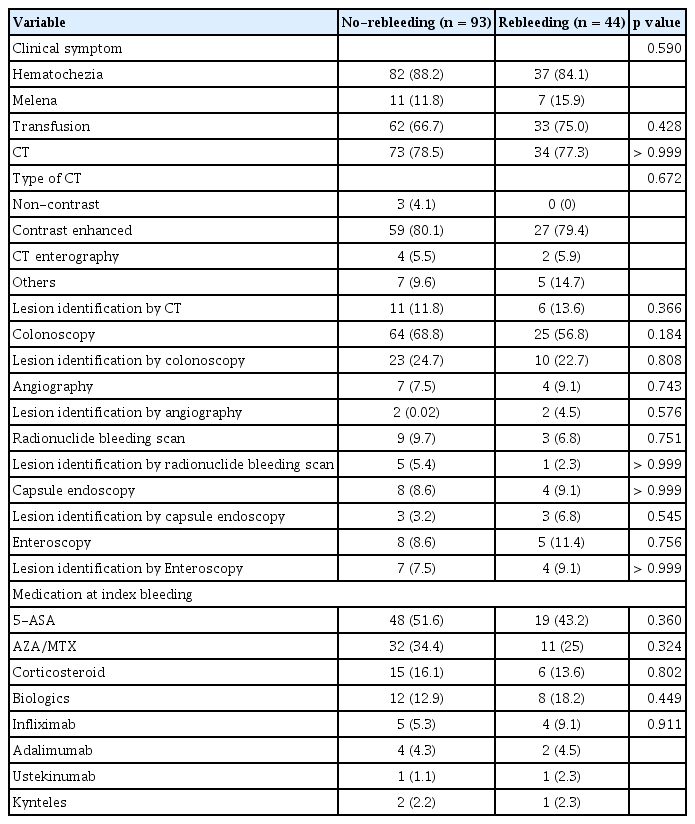
Clinical symptoms at index bleeding were hematochezia in 119 patients and melena in 18 patients. Except for 1 case with no information on blood transfusions, 95 of 136 (69.9%) patients required transfusions.
CT scan was performed in 107 patients, colonoscopy in 89 patients, capsule endoscopy in 12 patients, and enteroscopy in 13 patients. Bleeding sites were identified in 58 (42.3%) patients through CT, colonoscopy, angiography, bleeding scan, capsule endoscopy, or enteroscopy. Crohn’s lesion was ileocolonic in two cases where the disease and the bleeding location were different, and the bleeding location was confirmed in the more proximal ileum.
Bleeding sites were detected in 17 (15.9%) of 107 patients who underwent CT. Colonoscopy was conducted in 89 patients and revealed bleeding sites in 33 (37.0%) patients. Mesenteric angiography was performed in 11 patients, 4 (36.4%) of whom demonstrated bleeding spots. Of the 12 patients who underwent radionuclide bleeding scan, 6 (50.0%) exhibited bleeding sites. Bleeding focus was detected in 6 (50.0%) of the 12 patients who underwent capsule endoscopy. Enteroscopy detected bleeding focus in 11 (84.6%) patients.
Overall, 58 cases were identified with bleeding focus, and the bleeding site included the colon in 25 (43.1%) patients and the small intestine in 33 (56.9%) patients.
Conservative treatment (115 [84%] cases), endoscopic treatment (11 [8%]), embolization (5 [3%]), and surgery (6 [4%]) were performed for hemostasis. Escalation or change in medication was introduced in 78 (56.9%) patients. Among them, new introduction or dose escalation of AZA/6-MP was initiated in 32 (41.0%) patients, and change or new introduction of biologics was started in 30 (38.5%) patients.
Rebleeding occurred in 44 (32%) patients, and the median time to rebleeding was 13.0 (0.1–209) months. No statistically significant difference was observed in transfusion between the no-rebleeding (62 [65.3%] patients) and rebleeding (33 [34.7%]) groups (p = 0.428). No significant difference in terms of diagnostic modalities and bleeding focus was found between the non-rebleeding and rebleeding groups (Table 2).
Clinical outcome and factors associated with rebleeding
During the follow-up period, 27 patients experienced rebleeding once, whereas the other 17 patients had two or more rebleedings, with seven times of rebleeding noted in 1 patient. The frequency of rebleeding (one or more) was not significantly associated with the medications (e.g., aspirin, nonsteroidal anti-inflammatory drugs, antiplatelet agents), CD medications (e.g., 5-ASA, AZA/6-MP, corticosteroid, and biologics), or blood transfusion. No statistically significant difference in smoking and drinking was observed between the bleeding and rebleeding groups.
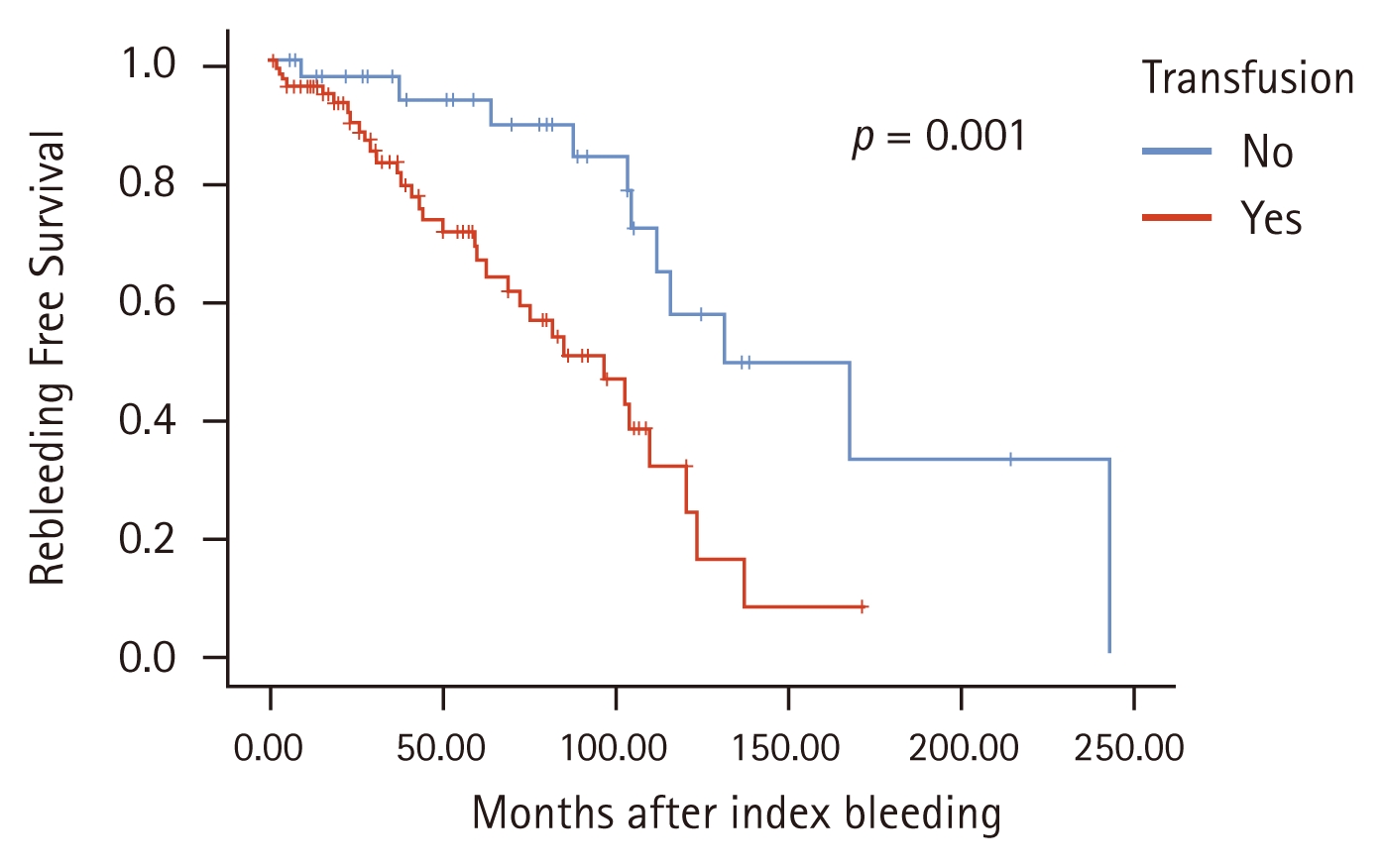
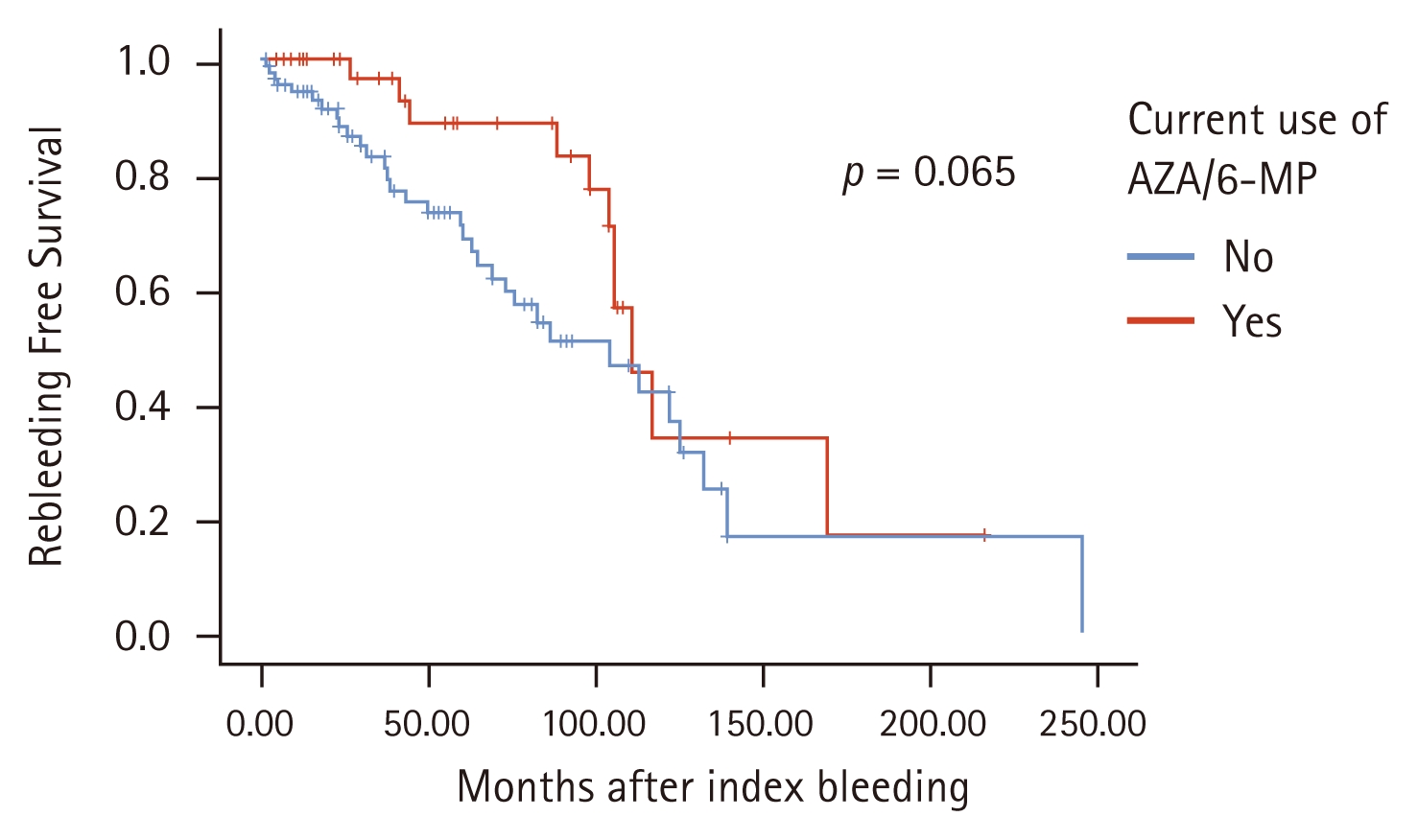
A significant difference in the necessity of transfusion was observed between the rebleeding and no-rebleeding groups based on univariate (hazard ratio [HR] = 3.124, 95% confidence interval [CI] = 1.502–6.494; p = 0.002) and multivariate (HR = 3.857, 95% CI = 1.678–8.867; p = 0.001) analyses (Fig. 1). The current AZA/6-MP administration tended to recur less despite no statistical significance (Table 3, Fig. 2).
DISCUSSION
Acute LGIB is one of the rare but serious complications of CD. In this study, we investigated the incidence and risk factors of rebleeding in patients with CD presenting with LGIB.
In our study, the rebleeding rate was 32% (44/137 patients), and the median time to recurrence was 13.0 (0.1–209) months. A previous Korean study revealed a cumulative risk of rebleeding of 33.0% after 1 year and 72.3% after 10 years [2].
A few studies focused on risk factors for rebleeding; however, several investigations have examined the risk factors of acute severe LGIB in CD. One Korean study reported that the female sex was associated with a lower risk of bleeding [8]. In another study, left colon involvement and bleeding history were associated with a higher risk of bleeding [3].
In our study, the necessity of transfusion at index bleeding was positively associated with the risk of rebleeding. The need for blood transfusion reflects substantial blood loss caused by a severe underlying lesion. Therefore, when transfusion is required, more intensive disease control should be considered, as these patients are at higher risk of rebleeding. No other statistically significant factors were identified, which is due to the small number of patients enrolled or because the analysis was retrospective.
Another study indicated that AZA/6-MP administration significantly and independently reduced the risk of acute severe LGIB [2]. Another study reported that early thiopurine administration was associated with a lower risk of bleeding [8]. Other research on risk factors for rebleeding revealed that anti-TNF therapy was associated with a lower risk of rebleeding compared with CMT [12]. Conversely, our study found that AZA/6-MP and biologic administration did not significantly reduce the risk of rebleeding. We could not identify the exact reason; however, it may be because the lesion, combined with bleeding, was one of the most difficult to be controlled with medication. Actually, most of the patients were already taking AZA/6-MP during the inquiry in our study, which could also be associated. Considering that 16 bleedings occurred even under biologic therapy, it is possible that the lesions were so severe that they could not be easily controlled by biologics. This could be a confounding factor. Further, a small number of cases or hidden confounding factors could also be associated.
One previous study investigated whether anti-TNF therapy was associated with a lower risk of rebleeding compared with CMT or surgical therapy and revealed that anti-TNF treatment was effective in preventing rebleeding in patients with CD presenting with acute severe LGIB [12]. However, our study revealed that changes in AZA/6-MP or biologics after bleeding had no statistically significant values as rebleeding risk factors, but demonstrated a tendency to reduce rebleeding risk. Considering the tendency in our results and previous findings [2, 8,12], active treatment with immunomodulatory or biologics could be helpful. Further prospective study with a large number of patients is warranted.
No difference was observed in rebleeding risk according to the location, and the presence or absence of previous bleeding could not be investigated because the first index bleeding patient was targeted.
This study has several limitations. First, because of its retrospective design, important clinical information such as the patients’ exact condition and concomitant medications at the time of index bleeding could not be fully collected. Second, the inclusion period was relatively long, during which meaningful changes in treatment strategies likely occurred following the introduction of the treat-to-target concept. Moreover, various biologics and small-molecule agents became available in the mid-2010s, which may have influenced clinical outcomes. However, index bleeding occurred in 67 cases before 2016 and in 70 cases after 2016, and the rebleeding rates did not differ significantly between the two periods (37.3% vs 27%, p = 0.203). Although the influence of temporal variation cannot be completely excluded, the absence of a statistically significant difference suggests that the impact of time period may be limited. Finally, the relatively small number of patients may have affected the robustness of the risk factor analysis.
Despite these limitations, our study has several strengths. This is the first multicenter study in Korea that included nine tertiary hospitals and utilized 20 years of data. Further, we focused on rebleeding, not just index bleeding, and this is the different point with most of the previous studies.
In conclusion, the incidence of rebleeding in CD patients with acute LGIB was 32%. Our findings indicate that severe index bleeding requiring transfusion is a significant risk factor for rebleeding. Although the effect did not reach statistical significance, initiation of AZA/6-MP or biologics after the index bleeding appeared to reduce the risk of recurrence. Therefore, patients with CD who experience severe bleeding necessitating transfusion require close monitoring and more aggressive disease control because of the increased risk of rebleeding.
KEY MESSAGE
1. The incidence of rebleeding in patients with CD experiencing acute LGIB was 32%.
2. Our results revealed that transfusion requirement was a significant risk factor for rebleeding.
3. Changes in AZA/6-MP or biologics after bleeding did not significantly decrease the risk of rebleeding but demonstrated a tendency to reduce the risk of rebleeding.
4. Therefore, patients with CD experiencing acute LGIB who need blood transfusions must be continuously monitored and treated more actively for rebleeding prevention.
Notes
CRedit authorship contributions
Gabin Moon: investigation, data curation, formal analysis, validation, software, writing - original draft, writing - review & editing; Kyeong Ok Kim: conceptualization, methodology, resources, writing - original draft, writing - review & editing, supervision, project administration, funding acquisition; Sung Noh Hong: resources, data curation, writing - review & editing; Seong Joon Koh: resources, data curation, writing - review & editing; Sung Hoon Jung: resources, data curation, writing - review & editing; Chang Hwan Choi: resources, data curation, writing - review & editing; Eun Mi Song: resources, data curation, writing - review & editing; Hong Sub Lee: resources, data curation, writing - review & editing; Yoo Jin Lee: resources, data curation, writing - review & editing; Ki Bae Bang: resources, data curation, writing - review & editing
Conflicts of interest
The authors disclose no conflicts.
Funding
This study was supported by the 2023 Yeungnam University Research Grant.