Comparison of the efficacy and safety of endoscopic and fluoroscopic balloon dilatation in benign esophageal strictures
Article information
Abstract
Background/Aims
Balloon dilatation under endoscopic or fluoroscopic guidance is regarded as the gold standard for treating benign esophageal strictures. However, no prior studies have compared endoscopic balloon dilatation (EBD) with fluoroscopic balloon dilatation (FBD). We aimed to compare the efficacy and safety of the two methods.
Methods
Subjects who underwent balloon dilatation under endoscopic or fluoroscopic guidance for benign esophageal strictures between 2001 and 2018 were eligible. Their medical records were retrospectively reviewed, and clinical characteristics were investigated.
Results
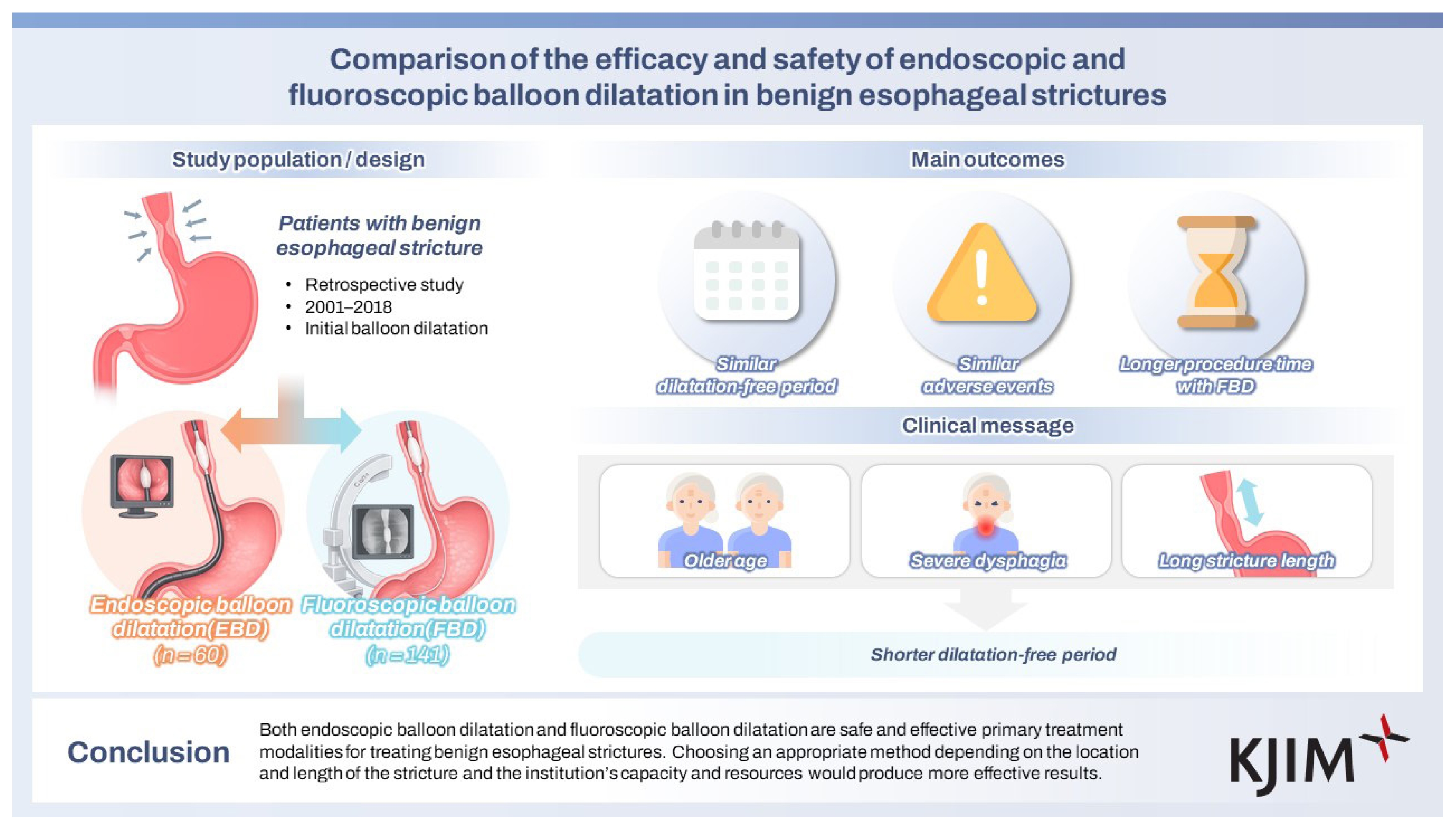
From 201 patients, 60 patients underwent EBD, and the remaining 141 underwent FBD. The mean age was higher in the FBD group than in the EBD group (69.77 vs. 58.63 yr, p < 0.001). The most frequent etiology of the strictures was surgery in both groups. Longer procedure times, cervical esophageal location, and short stricture length were significantly associated with the FBD group. Multivariable analysis showed that old age (HR 1.028), higher dysphagia grade (HR 1.561 for grade II and HR 3.518 for grade III), and long stricture length (HR 1.943) were significantly associated with the short dilatation-free period. There were no significant differences in dilatation-free periods or adverse events between the groups.
Conclusions
Both EBD and FBD are safe and effective primary treatment modalities for treating benign esophageal strictures. Choosing an appropriate method depending on the location and length of the stricture and the institution’s capacity and resources would produce more effective results.
INTRODUCTION
Benign esophageal strictures result from various etiologies such as surgical or endoscopic resection, peptic injuries, radiation therapy, or caustic ingestion. Esophageal strictures can be classified as either simple or complex according to their length, shape, and diameter [1]. Simple strictures such as peptic strictures and Schatzki’s ring are short (< 2 cm) straight, focal, and allow passage of normal diameter endoscopes. Complex strictures are longer (≥ 2 cm), angulated, and often severely narrowed. Strictures caused by surgical or endoscopic resection, caustic ingestion, and radiation therapy are examples of complex strictures [2].
Benign esophageal strictures are treated by fluoroscopic balloon dilatation (FBD) or by endoscopic treatments such as endoscopic balloon dilatation (EBD), endoscopic incisional therapy (EIT), and endoscopic stenting. The goal of the intervention is to relieve dysphagia symptoms by increasing the diameter of the esophageal lumen.
Among the different treatment modalities, balloon dilatation, either endoscopic or fluoroscopic, is the gold standard [3]. In the literature, EBD and FBD were highly effective in treating simple esophageal strictures, requiring between one and three dilatations to achieve symptom relief, while complex esophageal strictures often required more than three dilatation sessions [4]. However, no prior studies have directly compared the clinical outcomes of EBD and FBD. Therefore, this study compares the efficacy and safety of EBD and FBD in treating benign esophageal strictures.
METHODS
Patients
We retrospectively reviewed cases of benign esophageal strictures treated with balloon dilatation either endoscopically by gastroenterologists or fluoroscopically by interventional radiologists between 2001 and 2018. We excluded patients with malignant strictures and any cases that had been treated in other institutions. Age, sex, etiology of stricture, clinical symptoms, endoscopic and fluoroscopic images, procedural outcomes, and complications were all analyzed. This study was approved by the Institutional Review Board of Asan Medical Center (IRB number: 2021-1096). The requirement for written informed consent was waived due to the retrospective nature of the study.
Definition
Stricture
Esophageal strictures were diagnosed as strictures that a gastroduodenoscope (GIF H260 or HQ290; Olympus, Ltd., Tokyo, Japan) with an outer diameter of 9.8 mm or 10.2 mm could not pass through or when patients complain of dysphagia even if the endoscope could pass through. In the EBD group, the grade of stricture was divided according to whether they could be accessed with a 9.8 mm, 10.2 mm scope, or 5.5 mm scope (GIF-XT260N or XT290N; Olympus, Ltd.). In the FBD group, the stricture diameter was measured using fluoroscopic imaging and was classified as 9.8 mm or more, between 5.5 mm and 9.8 mm, and 5.5 mm or less. In both groups, stricture length was classified as either short (≤ 1 cm) or long (> 1 cm). The severity of dysphagia was scored as either 1 (inability to eat solid food), 2 (inability to eat semisolids), or 3 (inability to swallow liquids). Simple strictures were defined as short (< 2 cm) and straight, allowing passage of a standard endoscope. Complex strictures were defined as long (≥ 2 cm), tortuous, or not traversable with a standard endoscope [2].
Adverse events
Perforation was defined as a loss of continuity in the esophageal wall. Perforation was diagnosed by endoscopic inspection both during and immediately after EBD. Follow-up esophagography or a computed tomography scan was performed at the physician’s discretion in the EBD group. By contrast, esophagography was performed immediately after FBD to check for perforation in the FBD group. Bleeding was defined as clinical symptoms of bleeding, a decrease of > 2 g/dL in hemoglobin level, or the need for endoscopic hemostasis. Fever was defined as a body temperature equal to or greater than 38°C. Contrast media aspiration was defined simply as the aspiration of contrast media during esophagography.
EBD
The procedures were routinely performed on inpatients by six experienced gastroenterologists. Through-the-scope balloon dilatation (TTS-BD) was performed under conscious sedation with midazolam and pethidine during direct endoscopic visual control. TTS-BD catheters (Boston Scientific, Marlborough, MA, USA) between 6 to 20 mm were utilized, with the diameter of the balloon catheter determined based on the estimated size of the stricture.
The catheter passed into the stricture through the working channel of the endoscope with or without fluoroscopic guidance. After placing the balloon through the stricture, the balloon was inflated with normal saline to the recommended pressure for 30–60 seconds. The balloon diameter was then gradually increased by 1–2 mm over 2–3 increments. Although the initial treatments were EBDs, recurring strictures were treated with varying endoscopic methods such as repeated EBD, endoscopic stenting, or EIT depending on the physician [5].
FBD
Local anesthesia (use of an oral lidocaine hydrochloride spray) was administered immediately before the procedure without the use of sedation or general anesthesia. A small amount of water-soluble contrast medium was swallowed to opacify the stricture. Then, a 0.035-inch angled exchange guide wire (Radifocus wire; Terumo, Tokyo, Japan) was inserted through the mouth under fluoroscopic guidance, across the stricture, and into the distal part of the esophagus or stomach. A deflated balloon catheter (Boston Scientific) was then advanced over the guidewire and positioned across the stricture site. The deflated balloon catheter was slowly inflated with diluted water-soluble contrast medium until the hourglass deformity created by the stricture disappeared from the balloon contour [6,7]. Balloon dilatation was performed with increasing balloon diameters beginning with small balloons and progressing to larger sizes in order to minimize the risk of perforation [4]. Most often, a 10–15 mm diameter balloon was initially used. Then, if dilatation was easily accomplished and the patient tolerated the procedure well, the caliber of the balloon catheter was normally increased up to 20–25 mm.
The diameter of the balloon was chosen by the operator according to the adjacent normal lumen and severity of the stricture. In the case of postoperative anastomotic strictures, a relatively large diameter balloon catheter, such as a 16–18 mm balloon, was usually used from the first procedure. Other esophageal strictures, such as corrosive or post-radiation-induced strictures, were usually dilated with a 10–12 mm balloon. Consequently, if the dilatation was easily accomplished or was not blood-tinged on the balloon catheter, the balloon diameter was increased by 2 mm at the next session. The inflations were usually performed one or two times for 2–3 minutes in total [8–10]. Patients swallowed a small amount of water-soluble contrast medium or pull-back esophagography was performed through the retracted balloon catheter to determine the presence of perforation after balloon dilatation.
Statistics
Data are expressed as frequency (%) for categorical variables and mean ± standard deviation or median (interquartile range, IQR) for continuous variables. The Fisher’s exact test or chi-squared test was used to analyze categorical variables. Continuous variables were analyzed using the student’s t-test or the Wilcoxon rank sum test. Cox proportional hazards regression analysis and logistic regression analysis were performed to compare the dilatation-free period and adverse event rates, respectively, between the two groups. To identify risk factors for a short dilatation-free period, univariate and multivariate analyses were performed to obtain the hazard ratio (HR) with a 95% confidence interval (CI). Probability values of < 0.05 denoted statistical significance. All statistical analyses were performed using SPSS version 21.0 (IBM Corp., Armonk, NY, USA).
RESULTS
Baseline characteristics of the patients and lesions
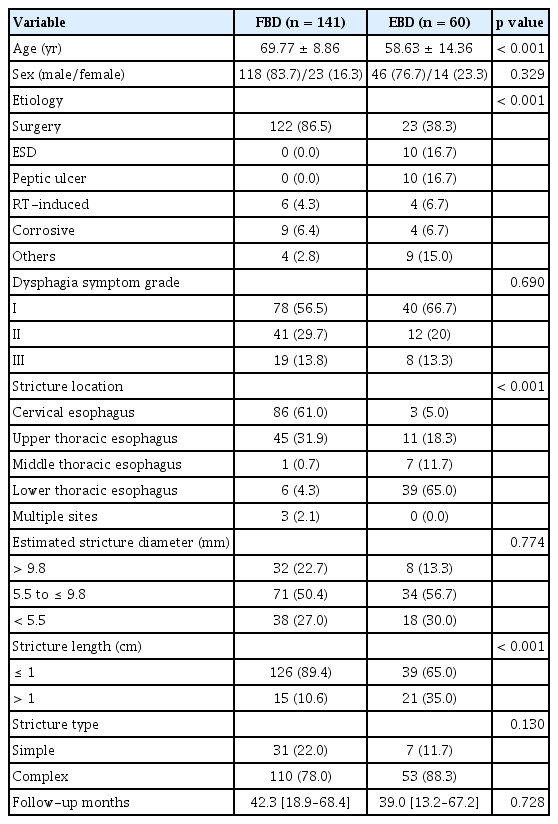
Baseline characteristics are summarized in Table 1. A total of 60 patients underwent EBD and 141 patients received FBD as their initial treatment for their benign esophageal strictures. For the EBD patients, 55 had treatment without fluoroscopic guidance, while the remaining five patients were treated with the assistance of fluoroscopy. The sex ratio was not significantly different between the groups, although the mean age was higher in the FBD group than in the EBD group (69.77 vs. 58.63 yr, p < 0.001). In the FBD group, 122 (86.5%) out of 141 strictures were caused by surgery. Out of 60 strictures in the EBD group, 23 (38.3%) were due to surgery and ten (16.7%) resulted from endoscopic submucosal dissection. The majority of strictures were in the cervical (61.0%) or upper thoracic part (31.9%) of the esophagus in the FBD group, while the majority of the strictures (65.0%) in the EBD group were in the lower thoracic part. In terms of the severity of the strictures, the estimated diameter of the strictures and dysphagia symptom grades showed no significant differences between the two groups. However, there were more short-length strictures (≤ 1 cm) in the FBD group compared with the EBD group (89.4% vs. 65.0%, p < 0.001). The median follow-up periods were not significantly different between the two groups (42.3 and 39.0 mo in the FBD group and EBD group, respectively).
Clinical outcomes
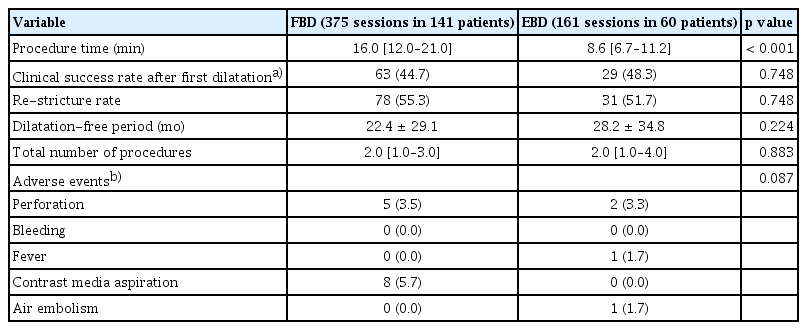
A total of 161 sessions (median two dilatations per patient; IQR 1–4) were performed on the 60 patients in the EBD group, and a total of 375 (median two dilatations per patient; IQR 1–3) were performed on the 141 patients in the FBD group. The maximal number of procedures was 12 in the FBD group and 11 in the EBD group. Re-stricture rates after the initial procedures were 55.3% in the FBD group and 51.7% in the EBD group (Table 2). The median procedure time was 16.0 minutes in the FBD group and 8.6 minutes in the EBD group (p < 0.001).
Patients who underwent FBD as the initial treatment option had subsequent treatments (either FBD or fluoroscopic stenting at the physician’s discretion) if symptoms recurred. Altogether, 30 patients (21.3%) needed four or more sessions to achieve symptomatic relief (Fig. 1). In the EBD group, patients with recurring symptoms received subsequent treatments such as EBD, endoscopic stenting, or EIT at the physician’s discretion, while 17 patients (28.3%) needed four or more sessions to achieve symptomatic relief. When combined with the FBD group and the EBD group, the recurrence rate after the first session was 54.2% (109/201). After the second and third sessions, patients who received balloon dilatation (either FBD or EBD) had recurring symptoms in 65.0% (65/100) and 63.6% (35/55) of cases, respectively, which were higher than the recurrence rate at the first session. In patients who received other treatment modalities than balloon dilatation at the second and third sessions, 66.7% (6/9) and 75.0% (12/16), respectively, had recurring symptoms. Regardless of group, cause of stricture and location of stricture, patients who had six or more procedures had longer strictures and more severe dysphagia symptoms, compared with those who had less than six procedures (Table 3).
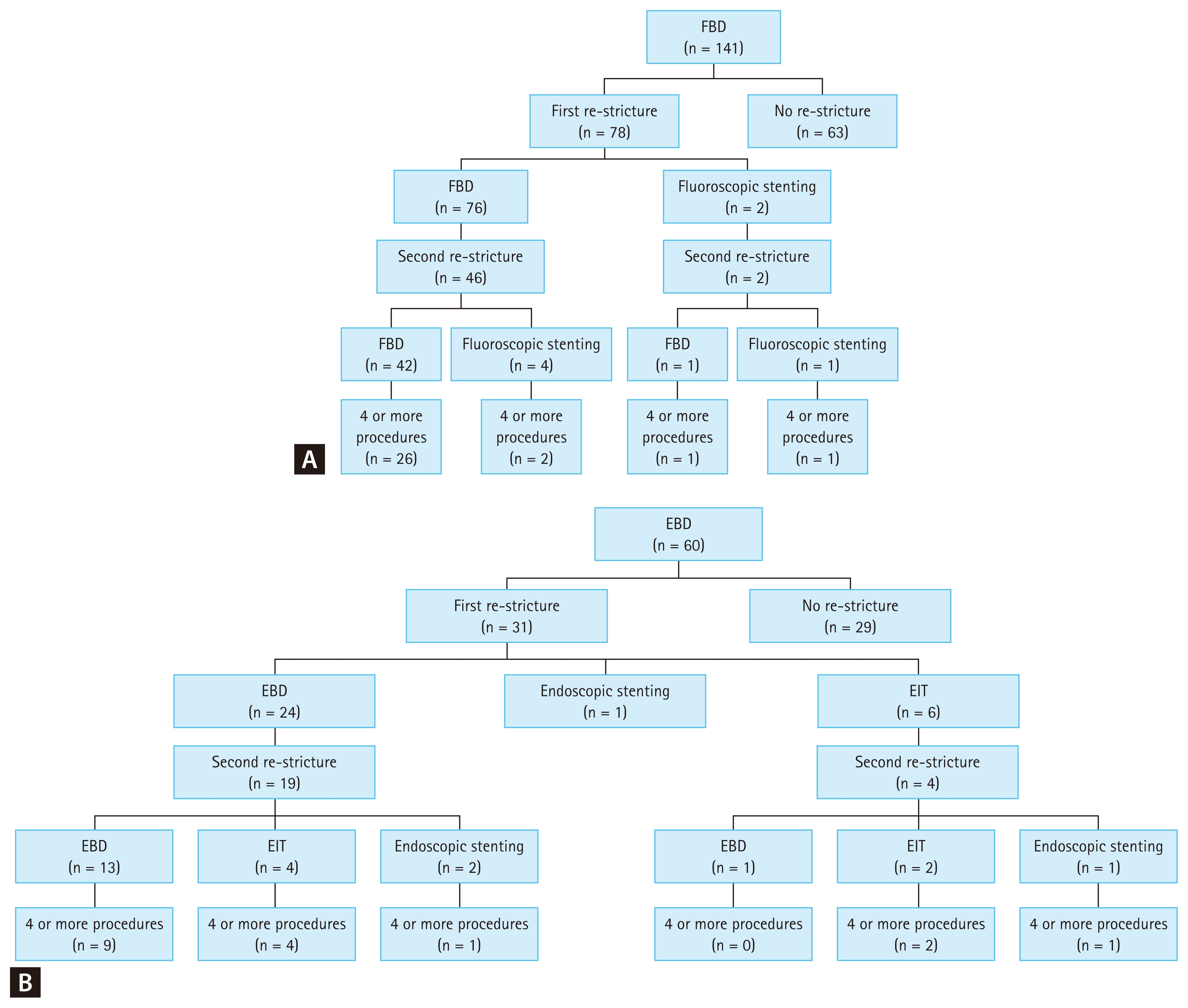
Flowchart of the stricture events: (A) FBD, (B) EBD. FBD, fluoroscopic balloon dilatation; EBD, endoscopic balloon dilatation; EIT, endoscopic incisional therapy.
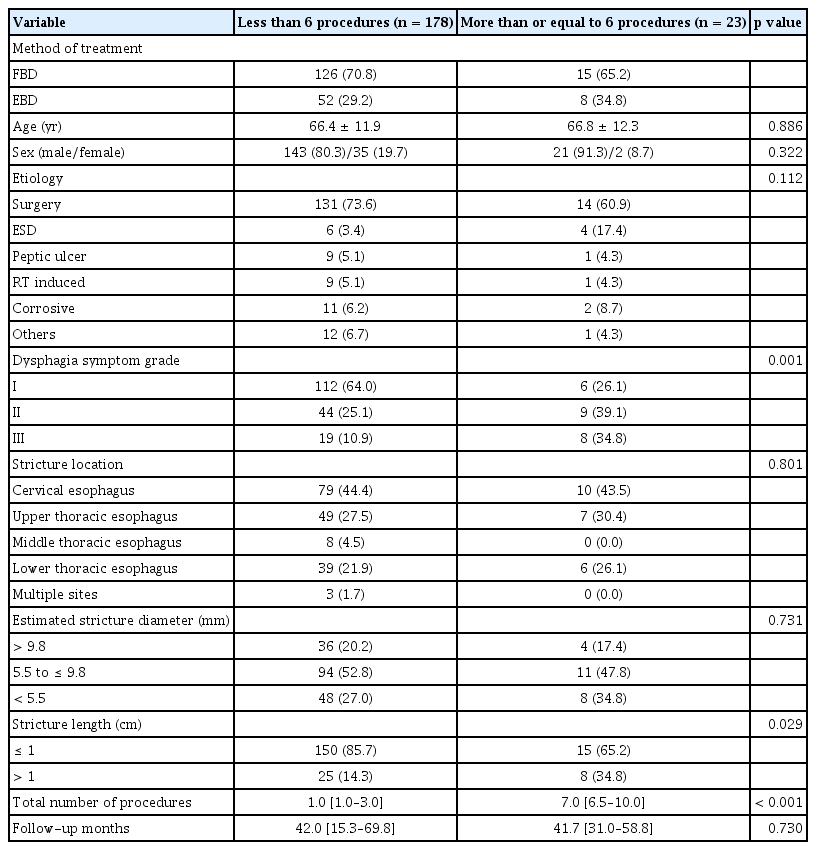
Comparison of characteristics between patients who had six or more procedures and those who had less than six procedures
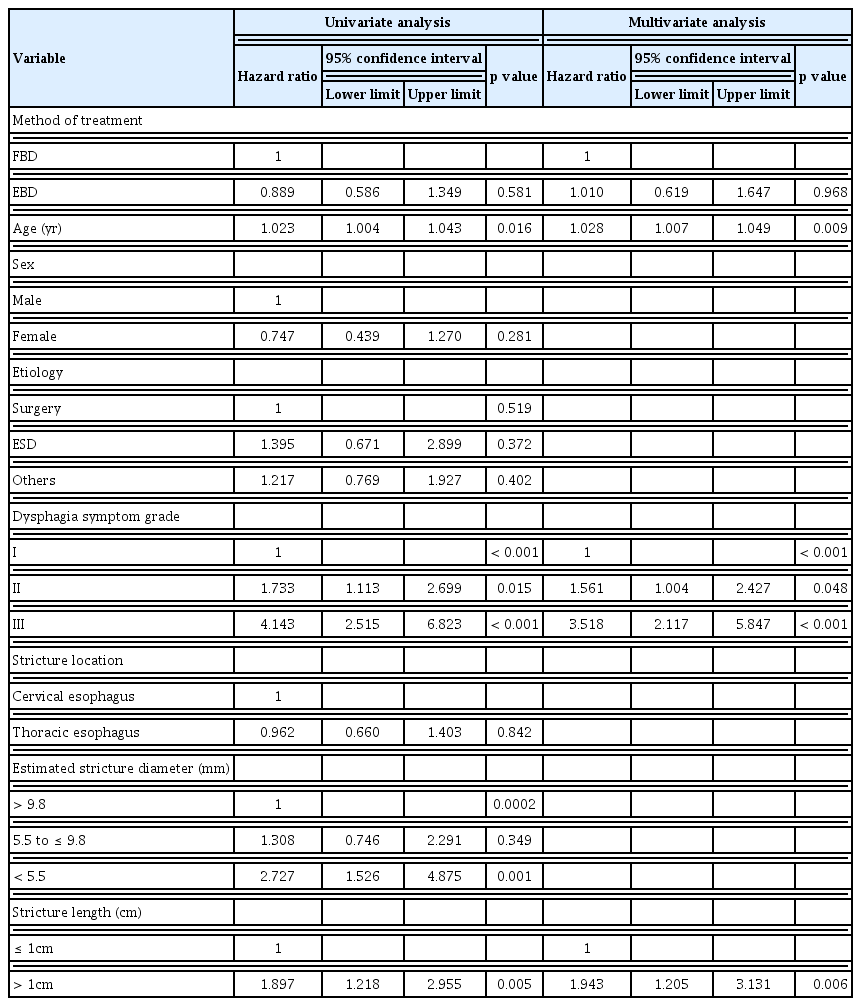
Univariate analysis, the method of treatment, sex, etiology, and stricture location did not show significant associations with a dilatation-free period. However, age, dysphagia symptom grade, estimated stricture diameter, and stricture length were significantly associated with a dilatation-free period (Table 4). Multivariate analysis was also carried out using the backward elimination technique and found that the method of treatment was not associated with a dilatation-free period (p = 0.968). However, age (HR, 1.028; 95% CI, 1.007–1.049; p = 0.009), severe dysphagia symptoms of grade 2 and 3 (HR, 1.561; 95% CI, 1.004–2.427; p = 0.048 and HR, 3.518; 95% CI, 2.117–5.847; p < 0.0001, respectively), and long stricture length (HR 1.943; 95% CI, 1.205–3.131; p = 0.006) were associated with the short dilatation-free period.
Adverse events
Logistic regression analysis showed that there were no significant differences between the groups regarding the occurrence of adverse events. Univariate and multivariate analysis showed that only stricture location was associated with adverse events. Multivariate analysis showed that cervical strictures were more associated with adverse events than thoracic strictures (OR 0.214; 95% CI, 0.043–0.802; p = 0.036).
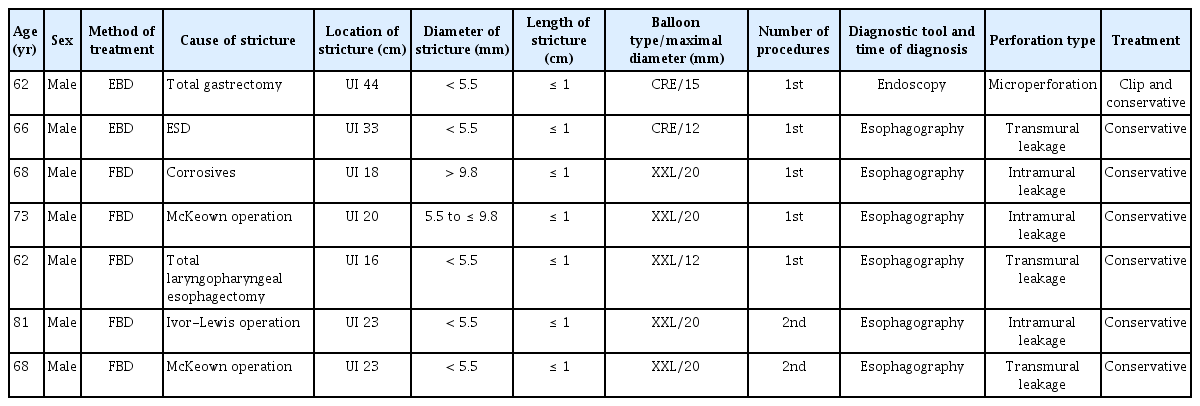
Although five perforations (1.3% per dilatation and 2.8% per patient) and two perforations (1.2% per dilatation and 3.3% per patient) occurred in the FBD group and EBD groups, respectively, all of the perforations were managed conservatively. The perforation cases are analyzed and summarized in Table 5. No bleeding events were reported in either group. One patient (0.6% per dilatation and 1.7% per patient) in the EBD group reported a high fever (≥ 38°C), which subsided the following day, while no cases of high fever were reported in the FBD group. In the FBD group, eight patients (2.1% per dilatation and 5.7% per patient) aspirated contrast media, compared with none in the EBD group. One patient (0.6% per dilatation and 1.7% per patient) in the EBD suffered a cerebral air embolism, which is a rare complication [11].
DISCUSSION
Benign esophageal strictures can negatively impact on an individual’s quality of life because of complications including dysphagia, regurgitation, or aspiration. Benign esophageal strictures are caused by various etiologies such as surgery, endoscopic intervention, peptic ulcer, radiation therapy, or caustic ingestions [4]. Strictures can be classed as simple and complex. Simple esophageal strictures are generally short and straight, and an endoscope can pass through. Peptic injuries and a Schatzki’s ring or web are common etiologies of simple esophageal strictures [12]. By contrast, complex esophageal strictures are often long and tortuous, and too narrow to access with an endoscope. Common etiologies of complex esophageal strictures include anastomotic strictures, caustic ingestion, and radiation therapy. Complex esophageal strictures often require multiple procedures [13].
EBD and FBD have been used as initial treatment modalities for benign esophageal strictures since they are considered safe and effective [4, 14]. In our study, EBDs were performed by experienced gastroenterologists on inpatients under conscious sedation. Endoscopy allowed the endoscopists to monitor the position of the balloon during inflation under direct vision. By contrast, FBDs were performed by experienced interventional radiologists without sedation in an outpatient setting. The position of the balloon was monitored under fluoroscopic guidance.
Regarding etiology, postoperative strictures were the most common cause of benign esophageal strictures in both groups. The mean age at the time of dilatation was older in the FBD group. Notably, there were significantly more cervical esophageal strictures in the FBD group compared with the EBD group (61% vs. 5%, respectively). This is probably because precise endoscopic manipulations are difficult in the cervical esophagus due to the contractions of the upper sphincter muscle and narrow working space. EIT may be a good alternative to EBD for cervical anastomotic strictures because EIT can be performed in narrower spaces and has shown particularly favorable outcomes in anastomotic strictures [5, 15–17]. Although estimated stricture diameters were not significantly different, there were more patients with short-segment strictures in the FBD group. This may be because the majority of the FBD group had short-segment anastomotic strictures.
This distribution likely reflects routine clinical practice patterns. In general, FBD is preferred for long or proximal strictures, particularly in the cervical or upper esophagus where endoscopic visualization of the distal end may be limited [18,19]. Conversely, EBD is typically more suitable for short-segment distal esophageal structures, where direct endoscopic control is feasible [20,21]. These considerations underscore the complementary roles of the two techniques and suggest that the apparent selection bias observed in our study is inherent to real-world practice.
A median of two dilatations were necessary to achieve symptom resolution in both groups. During the median follow-up periods of 42.3 months in the FBD group and 39 months in the EBD group, strictures recurred in more than 50% of patients after the first dilatation in both groups (FBD 55.32% and EBD 51.67%), which is consistent with previous literature [22,23].
Multivariable analysis showed that age (HR 1.028), high dysphagia grades (HR 1.561 for grade II and HR 3.518 for grade III), and long stricture lengths (HR 1.943) were associated with a short dilatation-free period. However, the method of treatment (whether fluoroscopic or endoscopic) was not associated with a dilatation-free period. This result is consistent with other literature that reported that the length and diameter of the esophageal strictures can significantly influence the effectiveness of dilatations. The literature also showed that narrower or longer strictures required more dilatations in order to achieve symptomatic relief [24].
The reported complications of esophageal balloon dilatation are perforation, bleeding, and bacteremia, with a perforation rate of between 0.1% and 0.4% reported [3,5,12,26]. In our study, perforation occurred in two patients in the EBD group (1.2% per dilatation) and five in the FBD group (1.3% per dilatation). All perforations were minor, and all patients recovered with conservative management. Reported perforation rates in the literature vary widely and are generally higher in complex strictures [3,4,7]. Because our cohort included a relatively high proportion of complex strictures, this may partly account for the comparatively elevated rate observed in our series. Most perforations occurred in the cervical or upper esophagus, and the majority were associated with very narrow strictures, findings that are consistent with established risk factors. Taken together, these findings suggest that perforation risk is closely related to stricture complexity, small luminal diameter, and proximal location. For very narrow strictures, initial dilation with a smaller balloon and increasing the diameter gradually may represent a safer approach. No significant bleeding events were reported in either group. One case of fever was noted in the EBD group, but the blood cultures were negative. Complication rates were not significantly different between the two groups.
Our study had some limitations. Firstly, the study design was retrospective in nature, meaning that selection bias is possible. Secondly, the two groups were not randomized and the treatment methods tended to depend on the referring doctor. More cervical anastomotic strictures were included in the FBD group and the non-random nature of the study may hinder the comparability of the two groups. Thirdly, the study was conducted in a single tertiary center, so the results may not be generalizable. A well-designed randomized controlled trial will be necessary in order to overcome these limitations. Lastly, the procedures were not fully standardized. Although techniques were relatively consistent within each group, variation in sedation, balloon type, inflation duration, and the use of fluoroscopy in a small number of EBD cases may have influenced the outcomes.
In conclusion, both FBD and EBD are safe and effective primary treatment options for benign esophageal strictures. FBD may be more appropriate for proximal or long strictures where endoscopic access is limited, whereas EBD may be better suited for short strictures in the distal esophagus. Selecting the treatment method based on stricture characteristics and available expertise is likely to yield optimal outcomes.
KEY MESSAGE
1. EBD and FBD had similar efficacy and safety for benign esophageal strictures.
2. Treatment modality itself did not affect the dilatation-free period.
3. Older age, severe dysphagia, and long stricture length predicted earlier recurrence after dilatation.
Notes
CRedit authorship contributions
Kwangbeom Park: data curation, formal analysis, writing - original draft; Chang Hoon Oh: data curation, writing - original draft; Do Hoon Kim: conceptualization, methodology, writing - review & editing, supervision; Ji Hoon Shin: writing - review & editing, supervision; Hee Kyong Na: data curation; Ji Yong Ahn: data curation; Jeong Hoon Lee: data curation; Kee Wook Jung: data curation; Kee Don Choi: data curation; Ho June Song: data curation; Gin Hyug Lee: data curation; Hwoon-Yong Jung: data curation
Conflict of Interest
The authors disclose no conflicts.
Funding
None