INTRODUCTION
Bone homeostasis is maintained by ensuring a balance between bone-resorbing osteoclasts and bone-forming osteoblasts. Many factors closely regulate bone resorption and bone formation in vivo to maintain bone homeostasis [1]. Bone erosion due to excessive osteoclast formation can occur in association with estrogen deficiency and increases in inflammatory diseases, such as rheumatoid arthritis (RA) and periodontal disease [2]. Most therapeutic drugs currently in use to treat osteoporosis inhibit osteoclast formation and thus bone resorption. Consequently, it is necessary to develop effective therapeutic drugs capable of improving the quality and quantity of bone.
Osteoclasts are the cells primarily responsible for bone resorption, and differentiation of these cells is regulated by multiple processes through differential gene expression. Osteotropic agents, including interleukin (IL)-1, IL-6, IL-11, IL-15, IL-17, tumour necrosis factor (TNF)-α, prostaglandin E2, and parathyroid hormone, cause bone loss by increasing osteoclast formation [1,3]. Receptor activator of NF-κB ligand (RANKL), an essential osteoclastogenic cytokine, recruits TNF receptor-associated factor (TRAF) proteins through binding to its receptor, RANK, on the surface of osteoclast precursors. TRAFs activate various signaling pathways, including c-Src, PI3-kinase/Akt, and mitogen activated protein kinase (MAPK) [4]. Several transcription factors, including c-Fos, nuclear factor of activated T-cells (NFAT) c1, PU1, and microphthalmia-associated transcription factor, are activated by RANKL and are important for osteoclast differentiation [5]. In particular, expression of c-Fos induced by RANKL plays an essential role in the initiation of osteoclast differentiation. c-Fos is responsible for the expression of NFATc1 during RANKL-mediated osteoclast differentiation [6,7].
Cudrania tricuspidata (CT) is a deciduous tree distributed over South Korea, China, and Japan, the cortex and root bark of which are used as a traditional medicine for curing inflammation and tumors [8]. Several reports have suggested various effects of CT extract, including antioxidant activity [9], inhibitory effects on nitric oxide synthase [10], and inhibitory effects on the proliferation of inflammatory immune cells and tumor cells [11,12]. However, there have been no investigations of the effects of ethyl acetate extract of CT (EACT) on the differentiation of osteoclasts, which play a crucial role in the pathogenesis of bone loss in several diseases, including RA.
We examined the inhibitory mechanism of EACT on IL-1β-stimulated RANKL-mediated osteoclast differentiation. Intracellular signaling factors were evaluated to define the mechanisms underlying the effects of EACT on osteoclast differentiation. We report here that EACT inhibits IL-1β-stimulated RANKL-mediated osteoclast differentiation by inhibiting extracellular signal regulated kinase (ERK) 1/2, p38 MAPK, c-Fos, and NFATc1 activation.
METHODS
Reagents and antibodies
Recombinant human IL-1β was purchased from R&D Systems (Minneapolis, MN, USA), and human soluble RANKL and macrophage colony stimulating factor (M-CSF) were purchased from PeproTech Inc. (Rocky Hill, NJ, USA). Monoclonal antibodies against ERK 1/2, phosphorylated ERK (p-ERK) 1/2, c-Jun amino-terminal kinase (JNK), phosphorylated JNK (p-JNK), p38, phosphorylated p38 (p-p38), NF-κB (p65), IκBα, and β-actin were purchased from Cell Signaling Technology (Beverly, MA, USA). Antibodies against c-FOS and NFATc1 were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). Fetal bovine serum was obtained from Gibco BRL Life Technologies (Grand Island, NY, USA).
Preparation of crude extract of C. tricuspidata
C. tricuspidata was collected from Jiri Mountain, Korea, and plant material was identified by an authority at the Rheumatology Laboratory and Research Center for Pulmonary Diseases, Chonbuk National University Medical School, Jeonju, Korea. Fresh stem bark was removed with a sickle and dried in a dark, well-ventilated room.
Air-dried stem bark (1 kg) was cut into pieces and extracted with 50% methanol at room temperature for several days. The methanol extract of CT was evaporated and partitioned consecutively with n-hexane, benzene, trichloromethane, ethyl acetate, and n-butanol as described previously [9]. The ethyl acetate fraction was used in this study after drying.
Isolation of osteoclast precursors and osteoclastogenesis
Osteoclast precursors were prepared as described previously with some modifications [13]. Briefly, bone marrow cells were obtained from the tibia and femur of 5-week-old male imprinting control region (ICR) mice. Red blood cells were removed in red blood cells lysis buffer (Sigma, St. Louis, MO, USA). Following centrifugation, bone marrow cells were suspended in α-MEM (Welgene, Seoul, Korea) containing 10% fetal bovine serum (Gibco BRL) and antibiotics and were cultured for 1 day in the presence of M-CSF (10 ng/mL). After 1 day, nonadherent cells were seeded on 10-cm culture dishes and cultured for 3 days in the presence of M-CSF (30 ng/mL); newly adherent cells were used as bone marrow cells. For the osteoclast differentiation assay, bone marrow macrophages prepared from bone marrow cells were cultured for 3 days with M-CSF (30 ng/mL) and RANKL (50 ng/mL). Bone marrow macrophages were cultured for 4 days with M-CSF (30 ng/mL) and RANKL (50 ng/mL) in the presence or absence of IL-1β (1 ng/mL) or EACT (100 µg/mL).
Cell viability analysis
Cell viability was determined using a CCK-8 kit (Dojindo Laboratories, Kumamoto, Japan) according to the manufacturer's instructions. Briefly, 2-(2-methoxy-4-nitrophenyl)-3-(4-nitropenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium (CCK-8) was reduced by dehydrogenases in cells to yield an orange-colored product (formazan) [14]. The amount of formazan dye generated by dehydrogenases in cells was directly proportional to the number of living cells. Bone marrow cells from the 5-week-old male ICR mice (1 × 104 cells per well in complete α-MEM media in 96-well plates) were cultured in 200 µL medium per well with M-CSF (30 ng/mL) and RANKL (50 ng/mL) or IL-1β (1 ng/mL) in the presence or absence of EACT (100 µg/mL) for 3 days. CCK-8 (20 µL) was added to each well of the plate, and the cells were incubated for 3 hours. Absorbance was measured at 450 nm using a microplate reader.
Immunoblotting
Cells were lysed in lysis buffer containing 50 mM Tris-Cl, 150 mM NaCl, 5 mM EDTA 1% Triton X-100, 1 mM sodium fluoride, 1 mM sodium vanadate, 1% deoxycholate, and protease inhibitors. The protein concentration was determined using Bio-Rad protein assay reagent (Bio-Rad Laboratories, CA, USA). Samples (50 µg) were prepared with four volumes of 0.5 M Tris buffer (pH 6.8) containing 4% SDS, 20% glycerol, and 0.05% bromophenol blue at 95℃ for 5 min. SDS-PAGE was performed in 10% slab gels, and the proteins were transferred onto nitrocellulose membranes. The membranes were washed in blocking buffer (10 mM Tris-HCl pH 8.0, 150 mM NaCl, 5% fat-free milk) for 60 min at room temperature with shaking and then washed with TBST (TBS, 0.01% Tween 20). Primary antibodies against ERK 1/2, p-ERK 1/2, p38, p-p38 MAPK, JNK, p-JNK, c-Fos, NFATc1, and β-actin at a concentration of 10 µg/mL were incubated at 4℃ for 4 hours. The secondary HRP-conjugated antibody was goat anti-rabbit IgG, anti-mouse IgG (Stressgen Biotechnologies Corporation, Victoria, Canada). Reactive proteins were detected using enhanced chemiluminescence (Amersham Life Sciences, Arlington, IL, USA) with Fuji Film LAS-3000 (Fuji Film, Tokyo, Japan).
RNA isolation and reverse transcription-polymerase chain reaction
Total RNA was extracted from cultured cells using the TRIsol reagent (Invitrogen, Carlsbad, CA, USA) in accordance with the manufacturer's instructions. Aliquots of 1 µg of RNA were reverse-transcribed using a Maxime RT Premix Kit (iNtRON Biotechnology, Seongnam, Korea). cDNA was amplified using the following primer sets: c-Fos sense, 5'-ATCGGAGGAGGGAGCTGACA-3'; c-Fos antisense, 5'-GGAACCGGACAGGTCCACAT-3'; NFATc1 sense, 5'-TGTGCAGCCAATTCCCCTG-3'; NFATc1 antisense, 5'-ATACCCCCCAGACCGCATC-3'; glyceraldehyde 3-phosphate dehydrogenase (GAPDH) sense, 5'-ACCACAGTCCATGCCATCAC-3'; GAPDH antisense, 5'-TCCACCACCCTGTTGCTGTA-3'. PCR products were electrophoresed on 1% agarose gels and visualized by staining with ethidium bromide. Densitometric analysis was performed on the relative intensity of each band using the Multi Gauge program, version 3.0 (Fuji Film).
Cytochemical assessment of osteoclast formation (tartrate-resistant acid phosphatase stain)
Cells were fixed with 3.7% formalin and permeabilized in 0.1% Triton X-100 for 10 minutes. The cells were stained with tartrate-resistant acid phosphatase stain (TRAP) solution using a leukocyte acid phosphatase kit (Sigma) according to the manufacturer's instructions. The numbers of osteoclasts were expressed as total counts per 48-well plate. Photographs were taken under a fluorescence microscope.
Resorption assay
Bone marrow cells (3 × 104 cells/well) were added to 4-well tissue culture plates coated with a calcium phosphate thin film (OAAS plates, Oscotec, Cheonan, Korea) prepared according to the manufacturer's instructions. After 2 weeks in culture, the cells were brushed away and OAAS plates were stained with toluidine blue. The area of resorption pits was measured in four randomly selected areas of each dentine slice using an image analysis system (Image Gauge, Tokyo, Japan).
Statistical analysis
All data are expressed as the means ± SD of triplicate measurements. Group mean values were compared by Student's t-test or analysis of variance as appropriate, and p < 0.05 was taken to indicate statistical significance. Statistical analyses were performed using SPSS version 12.0 (SPSS Inc., Chicago, IL, USA).
RESULTS
EACT inhibits RANKL-mediated or IL-1β-stimulated osteoclast differentiation
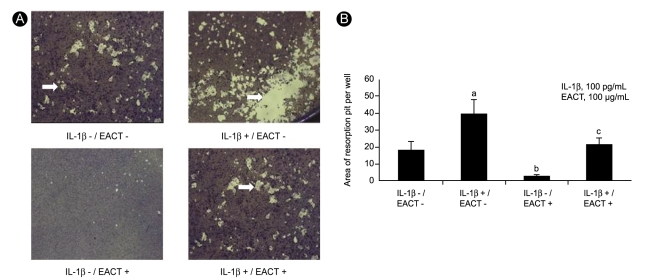
To determine the effects of EACT on RANKL-mediated or IL-1β-stimulated osteoclastogenesis, we initially measured the formation of TRAP (+) mononuclear cells as an indicator of osteoclast differentiation, which satisfied most of the morphological criteria of osteoclasts. IL-1β significantly increased the formation of TRAP (+) cells compared to the absence of this cytokine (550.6 ± 35.1 vs. 208.8 ± 20.3, respectively, p < 0.05). EACT significantly decreased RANKL-mediated or IL-1β-stimulated formation of TRAP (+) cells compared to the absence of EACT (84.7 ± 8.3 vs. 274.3 ± 13.8, respectively, p < 0.05, Fig. 1A and 1B).
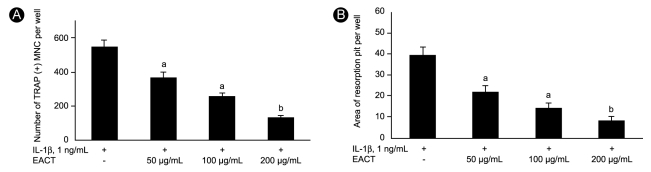
We also measured resorption area as another indicator of osteoclast formation. IL-1β significantly increased the resorption area compared to the absence of IL-1β (39.5 ± 8.3 vs. 18.3 ± 4.8, respectively, p < 0.05). EACT also significantly decreased the RANKL-mediated or IL-1β-stimulated formation of resorption pits compared to the absence of EACT (2.8 ± 0.8 vs. 21.5 ± 3.9, respectively, p < 0.05; Fig. 2A and 2B). EACT inhibited the RANKL-mediated or IL-1β-stimulated formation of TRAP (+) cells and resorption pits in a dose-dependent manner (Fig. 3A and 3B). These effects were also time dependent (data not shown).
EACT inhibits RANKL-mediated or IL-1β-stimulated survival of osteoclast precursors
To evaluate the effects of EACT on the growth properties of bone marrow cells from 5-week-old male ICR mice, we measured cell survival with M-CSF (30 ng/mL) and RANKL (50 ng/mL) in the presence or absence of EACT (100 µg/mL) or IL-1β (1 ng/mL) for 3 days as described in "METHODS." As shown in Fig. 4, IL-1β significantly increased the survival of osteoclast precursor cells compared to controls without IL-1β (p < 0.05). EACT also significantly inhibited the survival of osteoclast precursor cells compared to IL-1β (p < 0.05). However, there was no difference in survival of osteoclast precursor cells between controls and cultures with EACT. To determine the dose-dependent effects of EACT on IL-1β-induced survival of osteoclast precursor cells, we added various doses of EACT to the osteoclast precursor cell cultures with IL-1β (1.0 ng/mL) for 3 days and performed a CCK-8 assay. The inhibitory effects of EACT were significantly enhanced as the concentration of EACT increased (data not shown).
EACT suppresses c-Fos and NFATc1 expression induced by RANKL or IL-1β
Osteoclast differentiation is regulated by the induction of various genes in response to RANKL and other osteotropic agents, including IL-1β and TNF-α. IL-1β is a very important inflammatory cytokine in RA and causes bone loss by increasing osteoclast formation. Both c-Fos and NFATc1 play essential roles in the differentiation of osteoclast precursors [7]. Therefore, we examined whether EACT regulates the expression of c-Fos and NFATc1 in response to RANKL or IL-1β. The densitometric value of RT-PCR normalized by GAPDH intensity showed that EACT significantly decreased the RANKL-mediated or IL-1β-stimulated expression of c-Fos and NFATc1 mRNA (Fig. 5A). Consistent with the results of the RT-PCR analyses, expression of c-Fos and NFATc1 protein levels increased in response to RANKL and IL-1β, but the expression of both c-Fos and NFATc1 was significantly inhibited by EACT (Fig. 5B). Thus, EACT may inhibit osteoclast differentiation by inhibiting c-Fos and NFATc1 expression in response to RANKL and IL-1β.
EACT suppresses MAPKs signal pathways induced by RANKL or IL-1β
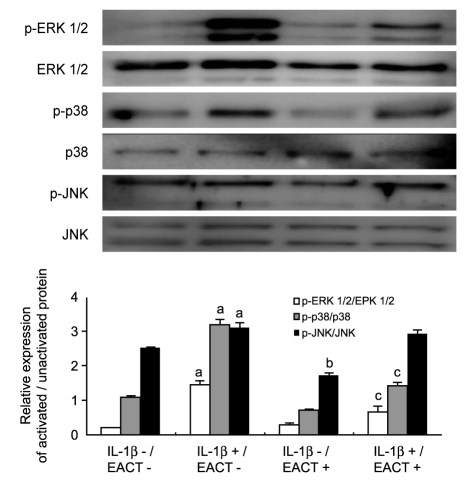
To determine the involvement of signal transduction and the mechanisms underlying the effects of EACT on IL-1β-stimulated RANKL-mediated osteoclast differentiation, we evaluated the activation of MAPKs in bone marrow cells. The densitometric values on immunoblots normalized according to β-actin intensity showed that IL-1β activated intracellular MAPKs, ERK 1/2, p38, and JNK (p < 0.05) and EACT significantly inhibited IL-1β-induced activation of intracellular MAPKs, including ERK and p38 (p < 0.05, Fig. 6). However, the effects of IL-1β on NF-κB activation were unaffected by EACT (data not shown). These results indicated that EACT inhibits RANKL-mediated or IL-1β-stimulated osteoclast differentiation via intracellular MAPKs pathways.
DISCUSSION
The results of this study show that EACT inhibits IL-1β-stimulated RANKL-mediated osteoclast differentiation in a dose-dependent manner by inhibiting activation of the ERK1/2 and p38 MAP kinases as well as inhibiting c-Fos and NFATc1 expression. These findings suggest that EACT plays an important role in inhibiting bone loss by preventing osteoclast formation and that it may be useful as a new therapeutic agent for managing osteoporosis or bone destruction in inflammatory diseases such as RA.
CT is a deciduous tree distributed over South Korea, China, and Japan, the cortex and root bark extract of are used as traditional treatments for inflammation, gastritis, and tumors [8]. However, there have been few studies of the pharmacological activity of CT extract extract of CT. Several reports have suggested various effects of CT extract extract of CT, including antioxidant activity [9], inhibitory effects on nitric oxide synthase [10], and inhibitory effects on proliferation of tumor cells [12]. Yet there have been few reports on the function and activity of CT extract extract of CT in inflammatory reactions. Chang et al. [11] reported that CT extract extract of CT could inhibit the proliferation of antigen-mediated spleen and T cells, which can accelerate inflammation and also reduce production of the proinflammatory cytokines IL-2 and IFN-γ in antigen-mediated T cells. Although these results suggest that CT extract extract of CT has effects on immune-mediated inflammatory joint diseases, including RA, the possible manner of osteoclast differentiation by which CT EACT inhibits inflammation-associated bone loss remains to be determined. There have been no studies of the effects on bone loss of inflammatory stimuli, especially IL-1, which plays a crucial role in the pathogenesis of bone destruction in RA.
The most striking features of RA are the hyperplasia of synovial fibroblasts in the lining layer and periarticular bone loss, which lead to joint destruction [15,16]. IL-1 plays an important role in the pathogenesis of inflammatory synovitis and joint destruction in RA by inducing the differentiation and activation of osteoclasts [17]. IL-1 can recruit and activate TRAF via heterodimeric IL-1 receptors. Thus, IL-1 differentiates and activates osteoclasts via intracellular signaling pathways, including MAPKs, c-Fos, and NFATc1 [18]. The results of the present study indicate that osteoclast differentiation induced by IL-1 can be significantly inhibited by EACT in a dose- and time-dependent manner. Thus, EACT may be an ideal treatment for bone loss in inflammatory arthritis.
There are more than 30 volatile components in EACT, and 8 of these compounds have antiinflammatory effects [11]. Further studies are needed to define which components of EACT are responsible for the observed results. This study shows that EACT inhibits the differentiation of osteoclasts in media with stimulating factors and inhibits IL-1-induced osteoclast differentiation. Further studies are required to determine the effects of EACT on osteoclast differentiation in the absence or presence of stimulating factors and to define the precise mechanisms of these effects.
A member of the TNF superfamily, RANKL mediates multiple signaling pathways via RANK in osteoclast precursors and activates critical transcription factors for osteoclast differentiation and function [1,4]. NFATc1 is one of the most important transcription factors and is expressed in osteoclast precursors through Ca2+ oscillation, MAPKs, and c-Fos or NF-κB in response to RANKL [19]. IL-1 differentiates and activates osteoclasts by recruiting and activating TRAF6 via downstream signaling pathways, including MAPKs, c-Fos, and NFATc1. We found that the inhibition of osteoclast differentiation by EACT coincides with the regulation of ERK 1/2, p38 MAPKs, c-Fos, and NFATc1.
This is the first study to indicate that CT EACT inhibits the IL-1β-stimulated RANKL-mediated differentiation of osteoclasts. This study also shows that CT EACT inhibits the phosphorylation of MAPK pathways and the activation of c-Fos and NFATc1. Taken together, these findings suggest that CT EACT may be useful for treating bone loss in inflammatory diseases, including RA. However, further studies are required to define the precise mechanism underlying the inhibition of osteoclast differentiation and to identify the active components in EACT.






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





