To the Editor,
Ozone is a component of photochemical air pollution; it is formed from volatile hydrocarbons, halogenated organics, and oxides of nitrogen in the presence of sunlight [1]. Ozone is a very strong oxidizing agent and can cause lung injury associated with edema, inflammation, and epithelial cell damage. It causes tissue injury not only in nasal airways [1,2] but also in pulmonary airways [3]. Ozone is known to decrease pulmonary function, increase airway hyper-responsiveness, and induce airway inflammation [3].
Proliferating cell nuclear antigen (PCNA) is a component of multi-protein complexes that are expressed during cell proliferation. PCNA is an auxiliary protein of DNA polymerase delta and is located in the nuclear compartment of proliferating normal and transformed cells [4]. The acute tracheobronchial epithelial injury and repair induced by ozone are thought to occur directly and indirectly through oxidative and inf lammatory mechanisms involving neutrophils [3]. Ozone induces epithelial necrosis, which leads to increased epithelial cell proliferation. Previously, we reported that ozone can induce alveolar epithelial cell proliferation in a dose-dependent manner, and that this proliferation is correlated with airway obstruction [5]. In this study, we investigated whether PCNA expression differs between airway and nasal skin.
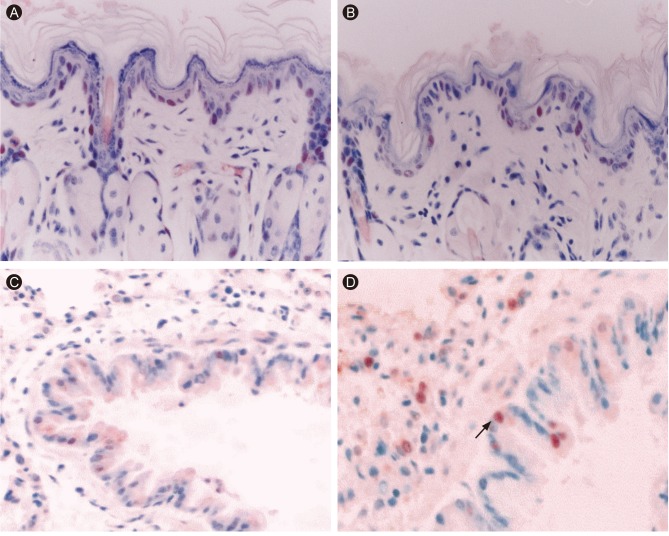
Female BALB/c mice 5 to 6 weeks of age were exposed to ozone concentrations of 0.12, 0.5, 1, and 2 ppm, or to filtered room air for 3 hours (n = 6/group). Ozone was generated using Sander model 50 ozonizers (Sander, Eltze, Germany) and monitored according to previously described methods [5]. Immunohistochemistry was performed on lung and nasal skin tissue mounted on Probe-On Plus slides (Fisher Scientific, Pittsburgh, PA, USA). All steps in the staining procedure were performed at 50℃ with a microprobe system that uses the capillary gap action produced by two opposed Probe-On Plus slides (Fisher Scientific). The slides were incubated for 15 minutes in anti-PCNA antibody (PharMingen, San Diego, CA, USA) diluted in primary antibody diluent (Research Genetics, Huntsville, AL, USA) at a final concentration of 5 µg/mL, and then for 10 minutes in goat anti-mouse IgG (Sigma, St. Louis, MO, USA). To block alkaline phosphatase activity, the slides were incubated for 2 minutes in Redusol (Biomeda, Foster City, CA, USA), and antigen/antibody complexes were detected via a 7-minute incubation with avidin alkaline phosphatase (DAKO, Glostrup, Denmark). The chromogen 3-amino-9-ethylcarbazole (Research Genetics) was applied for 10 minutes, followed by a 30-second application of hematoxylin. Samples of human spleen known to be PCNA-positive were used as positive controls, and were included with each staining reaction. For negative controls, the primary antibody incubation was omitted. All slides were examined by two observers using standard light microscopy. Lung sections were used to quantify PCNA-positive cells. PCNA-positive cells in the alveoli and nasal skin samples (Fig. 1) were counted by eye under a light microscope at × 400 magnification. The proliferative index was calculated as the number of PCNA-positive alveolar or nasal skin epithelial cells divided by the total number of each cell type. The PCNA index was calculated from a total of 10 fields selected at random. Following ozone exposure, the PCNA index increased in the cells lining airways (filtered air vs. 0.12 ppm vs. 0.5 ppm vs. 1 ppm vs. 2 ppm; 2.1% ± 0.4% vs. 5.7% ± 0.5% vs. 7.5% ± 1.1% vs. 13.4% ± 0.36% vs. 17.6% ± 0.52%, respectively; p < 0.01) (Fig. 2A). In contrast with airway, PCNA index in the skin of the nose decreased (filtered air vs. 0.12 ppm vs. 0.5 ppm vs. 1 ppm vs. 2 ppm; 13.2% ± 0.5% vs. 11.1% ± 0.3% vs. 9.6% ± 0.2% vs. 6.7% ± 0.25% vs. 3.5% ± 0.36%, respectively; p < 0.01) (Fig. 2B).
PCNA may be involved in both DNA repair and de novo DNA synthesis. Flow cytometry, tritiated thymidine labeling, bromodeoxyuridine incorporation, and Ki67 immunoreactivity have all revealed that PCNA immunoreactivity is correlated with other markers of the S phase of the cell cycle [2,5]. Ozone induces epithelial necrosis with a consequent increase in cell proliferation. Short-term exposure to 0.2 ppm ozone induces epithelial shedding and neutrophil recruitment. It also leads to increased CXC chemokine growth-related oncogene-α and interleukin-8 in bronchoalveolar lavage (BAL) fluid, and to decreased T-cell numbers, activation markers, and the CD4+/CD8+ ratio in BAL fluid [1-5]. The proliferating epithelial response following a single exposure to ozone is likely modulated through oxidative and inflammatory mechanisms involving neutrophils [1-5]. Exposure of animals to ozone induces airway and alveolar epithelial necrosis, together with an inflammatory cellular response characterized by an influx of neutrophils [3,5]. Blockade of PCNA production inhibits cell division, indicating that PCNA plays an important role in the process of cell proliferation [1]. This may be relevant to ozone exposure, because ozone damages DNA [1]. In this study, ozone induced epithelial injury in alveoli, resulting in a dose-dependent increase in alveolar epithelial cell proliferation and a dose-dependent decrease in PCNA in nasal skin. This finding suggests that cell proliferation responses in nasal skin and airway differ after short-term ozone exposure, which may reflect differences in epithelial cell damage and repair processes.




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





