INTRODUCTION
Extreme hyperlipidemia, defined as triglyceride levels > 1,000 mg/dL [1], is a rare condition in pregnancy that may be associated with abnormal lipoprotein lipase (LPL) activity. Partial LPL deficiency and dysbetalipoproteinemia are known to cause extreme gestational hyperlipidemia [2].
The effect of partial LPL deficiency and dysbetalipoproteinemia can be modulated by other genes, such as apolipoprotein (apo) E. Apo E2 has a lower binding affinity to the hepatic receptor [3,4], and patients with the apo E3/2 genotype are associated with higher plasma lipid levels compared to those without the apo E2 allele [5,6].
We report a case of a patient who experienced extreme recurrent gestational hyperlipidemia. Her first delivery was successful and without complications; however, she developed pancreatitis after her second delivery. She was diagnosed with a partial LPL deficiency without the LPL gene mutation in the presence of the apo E3/2 genotype. Several other similar cases have been reported, including patients with partial LPL deficiency who manifested extreme gestational hyperlipidemia [7]; however, the LPL gene mutation was associated in those cases. To our knowledge, this is the first reported case of gestational hyperlipidemia with a partial LPL deficiency but without an LPL gene mutation in the presence of the apo E 3/2 genotype. A patient without a LPL gene mutation has been reported, but the LPL activity was not measured in that case [8].
Implementation of extremely low-fat diets is usually adequate for the management of these patients. However, hyperlipoproteinemia and its associated complications may develop in patients who are unresponsive to dietary management with a progressive increase in triglyceride and cholesterol levels. Although drug therapy is contraindicated during pregnancy, treatment may be administered after a careful risk-benefit analysis [2].
CASE REPORT
A 28-year-old female patient presented to the hospital at 21 weeks of her second pregnancy. Her weight and height were 75 kg and 163 cm, respectively. She exhibited no clinical signs suggestive of hyperlipidemia such as tendon xanthomata, eruptive xanthomata, or other skin lesions. According to her family history, her mother had diabetes mellitus, and her father also had diabetes mellitus with hyperlipidemia. Her first pregnancy 1 year prior was influenced by gestational diabetes mellitus (GDM) and severe hyperlipidemia. Her first pregnancy progressed uneventfully, and other antenatal obstetrical examinations throughout her pregnancy were within normal limits. Labor was induced at the 38 weeks of gestation, and a healthy male infant weighing 4,050 g was delivered.
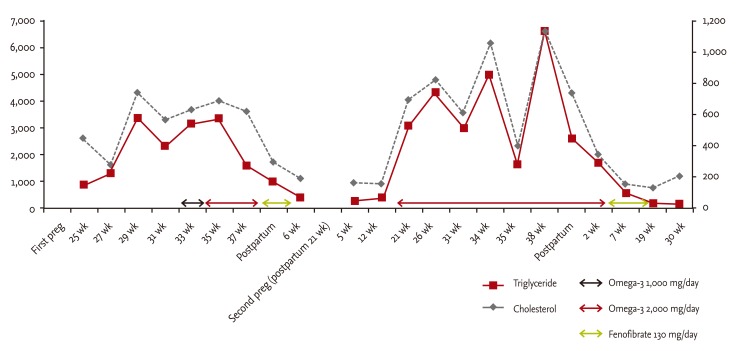
During the laboratory examination of her first pregnancy, functional tests of her liver, kidney, and thyroid were normal. At gestational week 25, she underwent a 50-g oral glucose tolerance test (OGTT) for screening of GDM; after 1 hour, her plasma glucose was 198 mg/dL. Next, 100-g OGTT was administered to the patient, and her plasma glucose levels at 0, 1, 2, and 3 hours were 79, 145, 175, and 169 mg/dL, respectively. Therefore, GDM was diagnosed by the Carpenter and Coustan criteria. Lipid levels indicated that her total cholesterol was 451 mg/dL, low density lipoprotein cholesterol (LDL-C) was 154 mg/dL, high density lipoprotein cholesterol (HDL-C) was 60 mg/dL, triglyceride was 1,128 mg/dL, and β-lipoprotein was 1,421 mg/dL. Supernatant samples exhibited a turbid, cloudy appearance. GDM and hyperlipidemia were managed by strict dietary control and the administration of omega-3 acid ethyl ester, which was started at 1 g/day. At 35 weeks of gestation, her lipid levels had increased with a cholesterol level at 622 mg/dL and triglyceride level at 3,360 mg/dL. Therefore, the omega-3 acid ethyl ester dose was doubled to 2 g/day. After delivery, the patient's medication was switched to fenofibrate (130 mg/day) at 6 weeks after delivery, her total cholesterol and triglyceride levels decreased to 195 and 413 mg/dL, respectively (Fig. 1).
At 21 weeks into her second pregnancy, extreme hyperlipidemia and GDM recurred with more severe laboratory findings. Upon administration of 100-g OGTT, her plasma glucose levels at 0, 1, 2, and 3 hours were 101, 158, 197, and 166 mg/dL, respectively. And lipid profile showed total cholesterol 693 mg/dL, LDL-C 99 mg/dL, HDL-C 42 mg/dL, triglyceride 3,108 mg/dL, β-lipoprotein 3,066 mg/dL, apo A1 188 mg/dL, and apo B 105 mg/dL, respectively. The patient was managed by strict dietary control and omega-3 acid ethyl esters at 2 g/day until labor. Her pregnancy progressed uneventfully, and labor was induced at the 38th week of gestation; a healthy male infant weighing 3,650 g was delivered. On the following day, she complained of epigastric pain. Peripheral blood tests showed that her cholesterol level was 1,137 mg/dL, triglyceride level was 6,600 mg/dL, amylase level was 179 U/mL (compared with normal ranges, 25 to 115), and lipase level was 133 U/L (compared with normal ranges, 13 to 60). Enhanced abdominal computed tomography revealed swelling of the pancreas with adjacent fluid collection consistent with acute pancreatitis (Fig. 2). After 4 days of complete parenteral nutrition and fluid therapy, her amylase and lipase levels were normalized. She was discharged 7 days after labor, with improved status of lipid levels with cholesterol 489 mg/dL and triglyceride 702 mg/dL (Fig. 1).
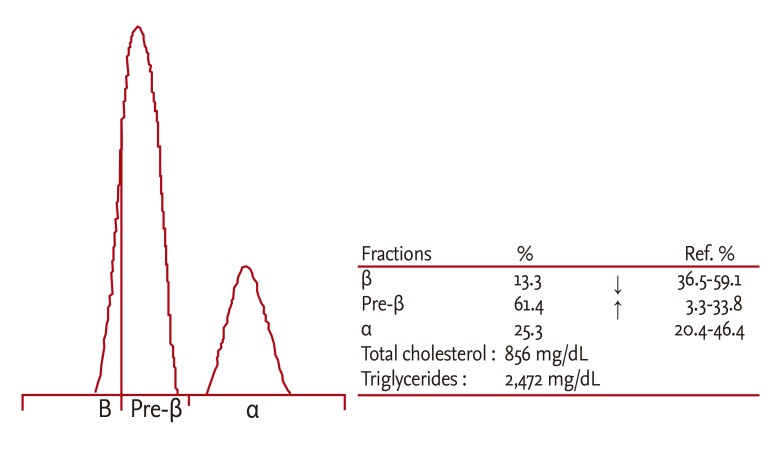
The patient experienced recurrent severe hyperlipoproteinemia limited to the gestational period. We performed lipoprotein electrophoresis, apoE genotyping and LPL gene analysis. Furthermore, we measured postheparin plasma LPL activity to elucidate the underlying etiology of the extreme gestational hyperlipidemia. Lipoprotein electrophoresis showed the increase of pre-β region and the decrease of β region (Fig. 3), and her apoE genotype was E3/2. We evaluated the LPL gene on chromosome 8q22, and detected no gene mutation. This test analyzed the entire exon, and the adjacent intron of the LPL gene was amplified using polymerase chain reaction (Model 9700, Applied Biosystems, Foster City, CA, USA), with direct gene sequencing using an automatic nucleotide sequencer (ABI 3130xl, Applied Biosystems). LPL activity was measured using an LPL enzyme-linked immunosorbent assay kit (American Diagnostica GmbH, Mannheim, Germany), performed in her nonpregnant state. A blood sample was collected from the patient after overnight fasting, and an intravascular injection of heparin was given (30 U/kg), with 5 mL of postheparin blood samples obtained 15 minutes after the injection [9]. LPL in samples was trapped on the plate due to the antigen-antibody reaction with the antibovine milk LPL mouse monoclonal antibody (anti-LPL monoclonal antibody) that was coated on the plate. Subsequently, antibovine milk LPL chicken serum (anti-LPL serum) was added, with horseradish peroxidase-labeled antichicken immunoglobulin G goat serum (enzyme-labeled antibody) applied to initiate the antigen-antibody reactions in each sample. The substrate o-phenylenediamine was then added for color development. The absorbance of the developed color was measured to determine LPL levels, which indicated an LPL activity level of 152 ng/mL (compared with normal values, 164 to 284).
After treatment for pancreatitis, her medication was switched to fenofibrate (130 mg/day), and her cholesterol level normalized, ranging from 130 to 200 mg/dL. In contrast, her triglyceride levels were mildly elevated, ranging from 139 to 567 mg/dL.
DISCUSSION
During a normal pregnancy, total cholesterol and triglyceride levels increase progressively due to hormonal changes, and significant dyslipidemia occurs more frequently in the third trimester. Extreme hyperlipidemia, defined as triglyceride level > 1,000 mg/dL [1], is a rare condition during pregnancy. Partial LPL deficiency and dysbetalipoproteinemia are known to cause extreme gestational hyperlipidemia [2]. LPL deficiency can cause hyperlipidemia, and patients with complete LPL deficiency frequently experience recurrent episodes of abdominal pain, pancreatitis, and eruptive xanthomas from early childhood. However, patients with partial LPL deficiency may have a normal lipid profile and may therefore go unnoticed until environmental stress, such as pregnancy or the use of estrogen-containing contraceptives or alcohol, is placed upon the defective lipolytic system. In this case, our patient showed no clinical signs suggestive of hyperlipidemia, such as tendon xanthomata, eruptive xanthomata or other skin lesions, and hyperlipoproteinemia was limited to the gestational period.
Dysbetalipoproteinemia is another condition that may cause severe hyperlipoproteinemia limited to the gestational period. Dysbetalipoproteinemia is a genetic disorder, which is caused by apo E deficiency or the expression of apo E isoforms that are defective in binding to lipoprotein receptors [2]. More than 90% of the incidence of dysbetalipoproteinemia is associated with homozygous apoE2, and a fraction of the β region is increased in lipoprotein electrophoresis. In this case, our patient's genotype was apoE3/2, with an increased pre-β region and a decreased beta region, suggesting a low probability of dysbetalipoproteinemia.
The effect of partial LPL deficiency can be modulated by other genes involved in the metabolism of triglyceride-rich lipoproteins, such as apoE. ApoE serves as one of the ligands for the hepatic remnant receptor and plays a key role in the clearance of chylomicron remnants after lipolysis. The apoE3 allele shows normal clearance function of chylomicron remnants. One apoE variant, apoE2, has a much lower binding affinity to the hepatic receptor [3,4]. Patients with the apoE2/3 genotype are more likely to have higher plasma lipid levels than patients without the apoE2 allele [5,6]. In this case, our patient's genotype was apoE3/2. The presence of the single copy of the apoE2, which delays the clearance of remnant particles, may have reduced the patient's tolerance of extreme hyperlipidemia during pregnancy, resulting in partially decreased LPL activity. Several other cases of severe gestational hyperlipidemia have been reported; however, this is the first reported case of gestational hyperlipidemia with a partial LPL deficiency but without an LPL gene mutation in the presence of the apoE3/2 genotype.
Statins and fibrates are used to treat combined hypercholesterolemia and hypertriglyceridemia, and reports have indicated successful use during the pregnancy with no side effects. However, statins are not generally recommended for the treatment of hyperlipidemia that develops during pregnancy, and fibrates are contraindicated [10]. Other treatment options for resistant cases include plasma exchange, plasmapheresis, intravenous heparin, and total parenteral nutrition. Omega-3 acid ethyl esters are also effective for treating hypertriglyceridemia. Furthermore, consumption during pregnancy is associated with improved neurodevelopmental outcome in the child. We managed the patient with strict dietary control and medicated with omega-3 acid ethyl esters.
The patient's first delivery was successful without complications; however, her condition was complicated by pancreatitis after her second delivery. Pancreatitis often occurs in individuals with extreme hyperlipidemia. Acute pancreatitis and other hyperlipidemic complications are associated with triglyceride and cholesterol levels [2], but correlations with delivery are unknown. In this case, the patient exhibited acute pancreatitis after delivery. Her second delivery period showed the highest serum triglyceride and cholesterol levels of her entire hospital stay; consequently, acute pancreatitis was suspected. The effect of delivery on the decline of plasma triglyceride levels can be immediate and dramatic. Therefore, decisions regarding the timing and route of delivery in the presence of extreme gestational hyperlipidemia are important and should be made carefully. It is possible that an induced early delivery or an elective caesarean section would have been appropriate during the second delivery in this case.




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





