INTRODUCTION
Drug-resistant tuberculosis (TB) is a global concern. However, the overall incidence of TB has been decreasing slowly since 2004 because of the World Health Organization (WHO) efforts through the Stop TB Strategy [
1]. Nevertheless, there is no definitive evidence that the incidence of multidrug-resistant (MDR) TB is decreasing, despite this global effort [
2]. Although considerable efforts in several countries, including South Korea, have led to a slow decrease in the overall incidence of TB, surveillance indicates that the incidence of drug-resistant TB is rising [
3,
4,
5]. Drug-resistant TB strains in South Korea account for 5.3% to 12.2% of new cases and 13.3% to 41.6% of previously treated cases, whereas MDR-TB strains account for 2.7% to 3.9% of new TB cases and 14.0% to 27.2% of previously treated cases [
3,
6,
7,
8].
Drug-resistance is a critical determinant of treatment success. Thus, to improve treatment success, the
Mycobacterium tuberculosis drug-resistance pattern in community-based care should be determined. Despite geographical variations, a few studies have reported that drug-resistant TB occurs less often in cases of extrapulmonary tuberculosis (EPTB) than in cases of pulmonary tuberculosis (PTB) [
9,
10]. However, insufficient data are available regarding the current status of anti-TB drug-resistance in South Korea since 2008. Furthermore, there are no remarkable drug-resistance data for EPTB. Therefore, we evaluated the prevalence of drug-resistance among
M. tuberculosis isolates, as well as differences in the drug-resistance pattern among the
M. tuberculosis isolates that cause PTB or EPTB.
DISCUSSION
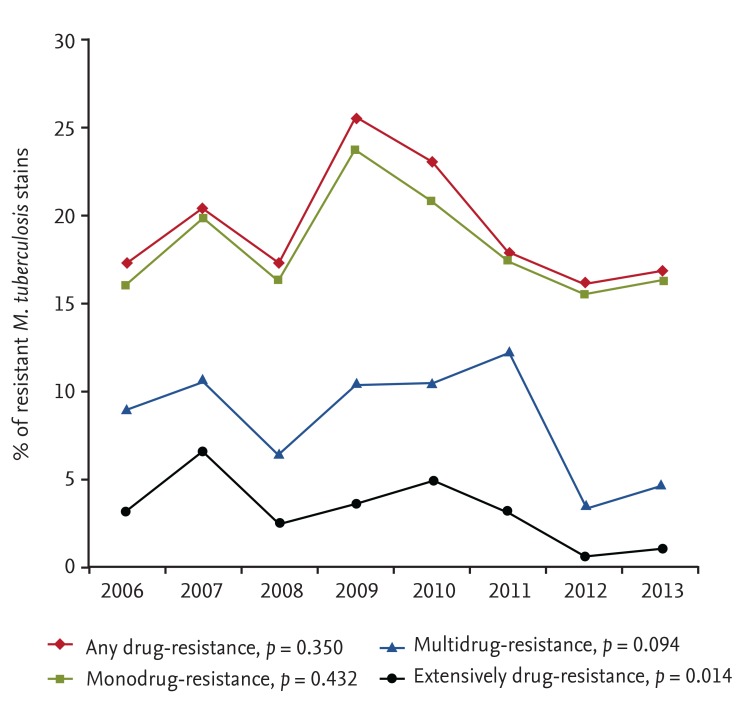
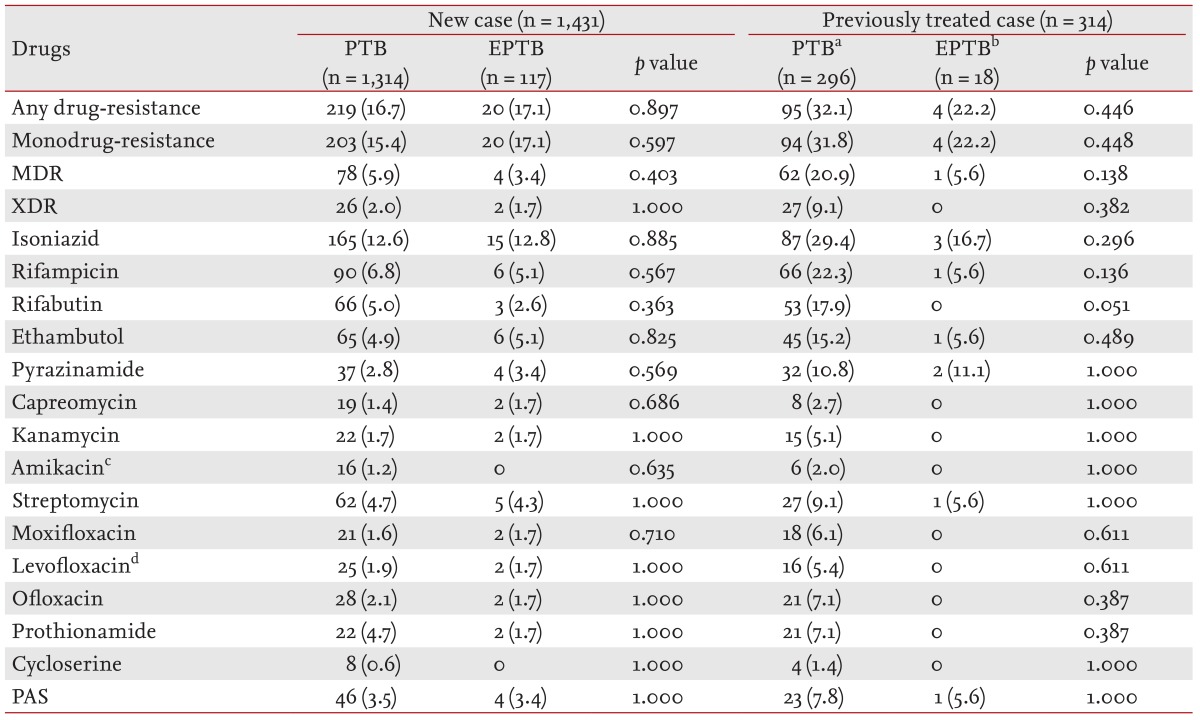
In this study, we investigated changes in TB drug-resistance patterns and differences in the drug-resistance pattern among new and previously treated TB cases. In addition, we compared the prevalence of drug-resistant TB observed in patients with PTB with that observed in patients with EPTB. The major findings were as follows. The rates of MDR and XDR TB in the private referral hospital remained high, but a slow decrease over time was suspected, particularly for previously treated TB cases. This finding may have been affected by changes in the proportions of relapse and TAI cases over time. In addition, although we evaluated a small number of EPTB cases, no difference in the drug-resistance rate between cases of PTB and EPTB was detected.
Determining the change in the TB drug-resistance rate over time and its current status in a region or nation are essential to adequately administer anti-TB regimens and achieve treatment success. According to the annual report of the Korea Centers for Disease Control and Prevention of 2012, 1,212 MDR TB cases were reported via the national TB monitoring system, which is a large burden in Korea despite a national effort [
15]. However, no information regarding whether these were new or previously treated cases is available, despite the fact that a TB treatment history is a potent risk factor for MDR TB [
16]. In addition, the proportions of patients with any-drug-resistant or monodrug-resistant TB are also unknown. According to the most recently published data regarding drug-resistance rates in 2009 in a public health center, the percentage of any-drug-resistant TB strains was 12.6% of new cases and 20.0% of previously treated cases. In addition, the percentage of MDR strains was 2.8% of new cases and 8.5% of previously treated cases [
17].
However, the number of TB cases treated in the private sector has been increasing continuously and was 90.4% in 2012, as assessed by the national TB monitoring system [
15]. Several studies have analyzed the TB drug-resistance rate in the Korean private sector [
3,
6,
17,
18,
19,
20,
21]. Unfortunately, no reported data are available regarding the drug-resistance rate in the private sector after 2009. Furthermore, patients diagnosed with MDR or XDR TB since 2011 must be hospitalized to prevent transmission of drug-resistant pathogens based on the revised Korean National Tuberculosis Control program. Therefore, a more accurate evaluation of the drug-resistance rate among TB cases in private sector clinical settings and the trend in the drug-resistance rate among TB cases is needed.
According to previous studies performed in the private sector based on TB strains identified before 2009 [
3,
6,
17,
18,
19,
20,
21], the any-drug-resistant TB rate was 15.0% to 20.4%, the monodrug-resistant TB rate was 6.4% to 17.6%, the MDR TB rate was 4.4% to 13.2%, and the XDR TB rate was 0.5% to 1.4%. In addition, MDR TB accounted for 2.9% to 5.1% of new cases and 9.3% to 41.8% of previously treated cases, whereas XDR TB accounted for 0.3% to 1.1% of new cases and 2.2% to 47.3% of previously treated cases. These studies show that any-drug-resistant and MDR TB decreased from 2001 to 2006. However, no definitive trend was observed for the gradual decrease from 2007 to 2008.
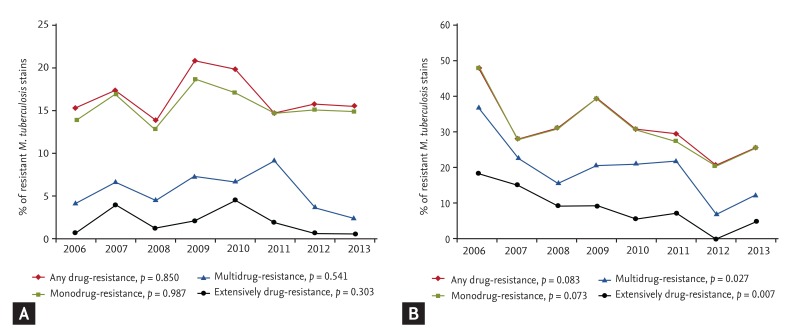
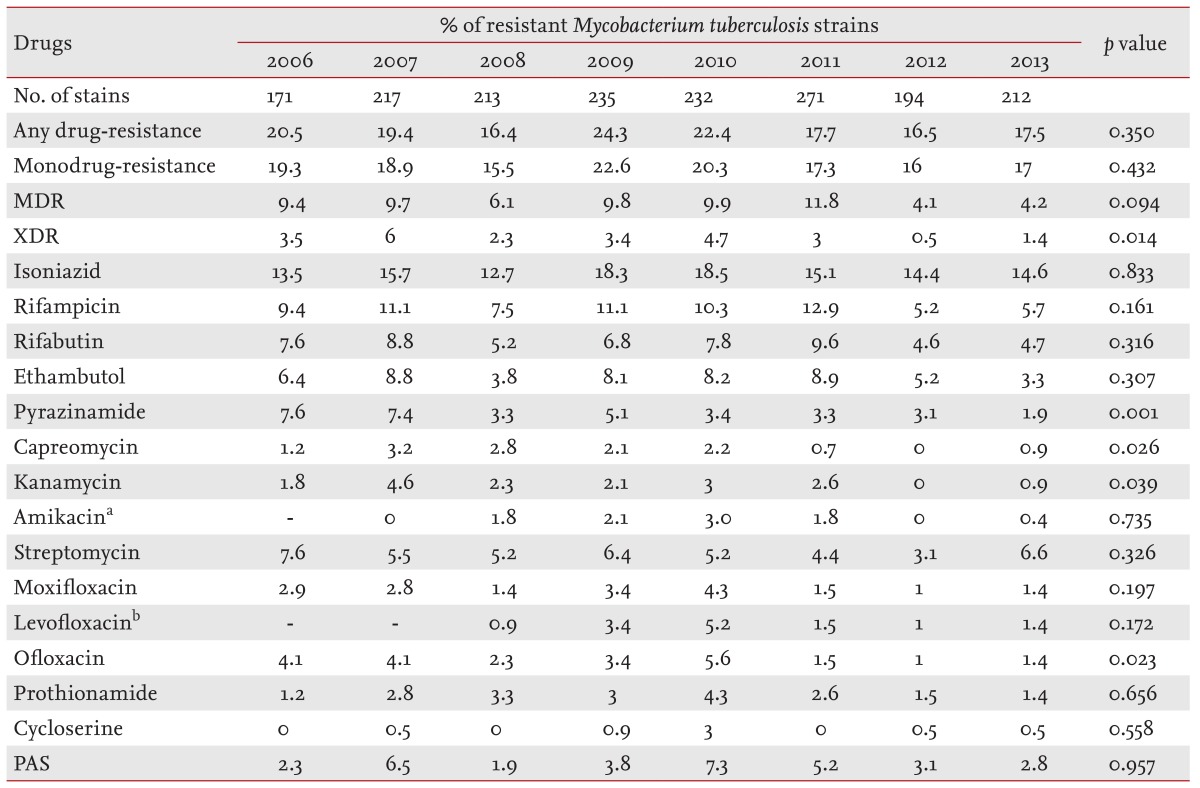
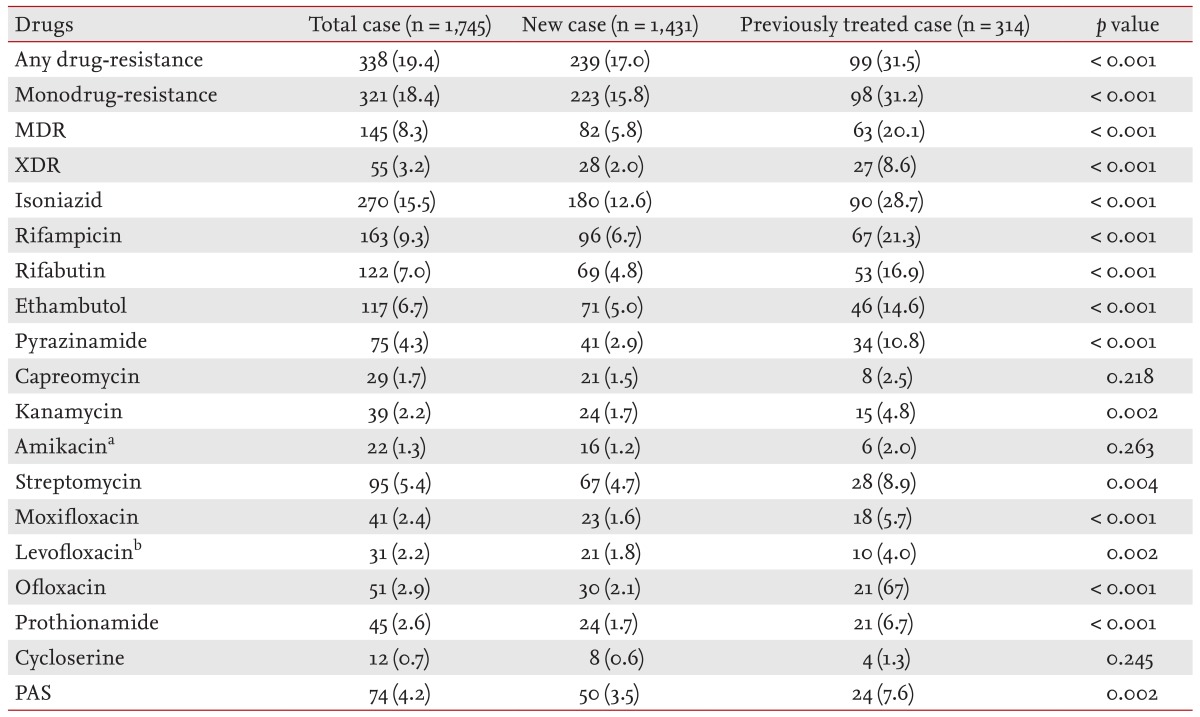
In our study, the any-drug-resistant TB rate was 19.4%, the monodrug-resistant TB rate was 18.4%, the MDR TB rate was 8.3%, and the XDR TB rate was 3.2%. In addition, any-drug-resistant TB accounted for 17.0% of new cases and 31.5% of previously treated cases. Monodrug-resistant TB accounted for 15.8% of new cases and 31.2% of previously treated cases. MDR TB accounted for 5.8% of new cases and 20.1% of previously treated cases. Finally, XDR TB accounted for 2.0% of new cases and 8.6% of previously treated cases. Although the drug-resistance trend among all TB cases did not decline remarkably over the study period, except XDR TB, the proportions of MDR and XDR TB decreased gradually among previously treated cases. This observation could be a result of the significant increase in the proportion of relapse cases and a decrease in the proportion of TAI cases during the study period. Because the drug-resistance rate is much higher in TAI cases than that in relapse cases, it may have affected the gradual decrease in the drug-resistance rate of the previously treated cases.
Although drug-resistant TB is a critical determinant of treatment success in a TB control program, few studies have compared the drug-resistance pattern of PTB with that of EPTB. In addition, three studies have reported that MDR TB occurs less frequently in EPTB cases than it does in PTB cases [
9,
10,
22]. Thus, we compared the pattern of drug-resistance in EPTB with that observed in PTB. However, we found no difference in the drug-resistance rate between PTB and EPTB according to previous treatment status.
MDR TB was less frequent among patients with EPTB than it was among those with PTB, which was similar to results reported previously. However, the drug-resistance rates of almost all anti-TB drugs, except that of Rfb, were not different. In reports from Taiwan and Russia [
10,
22], the monodrug-resistance rate in patients with EPTB was also significantly lower than that in patients with PTB.
A possible explanation for this finding is that all cases were culture-confirmed cases. Thus, it was possible that many drug-sensitive EPTB cases were excluded. In an actual clinical setting, many EPTB cases are diagnosed without culture because isolating
M. tuberculosis from an EPTB site is difficult, although the WHO recommends culturing specimens from a suspected extrapulmonary site. In fact, the proportion of EPTB cases among all TB cases was lower (7.7%) in our study than the 21.8% reported in the USA, the 27.5% reported in Russia, and the 21.4% reported in Taiwan [
9,
10,
22]. Another possible explanation is that the proportion of drug-resistant strains of infectious TB bacilli in a community affects similar drug-resistance to PTB and EPTB. In contrast, another possible explanation is that studies conducted outside Korea were based on the specimen site, without considering concurrent EPTB and PTB [
10]. In addition, another study excluded cases of concurrent EPTB and PTB [
9]. In our study, we classified pulmonary and EPTB separately, as recommended by the WHO [
12], although we found few concurrent EPTB and PTB cases (n = 45).
This study had several limitations. First, it was performed in a single tertiary referral center; thus, the results may not reflect the overall situation in South Korea. Second, a lack of information regarding the DST for previous TB infections in the previously treated TB cases precluded ascertaining whether drug-resistance was attributable to inadequate or incomplete treatment or whether it was the result of a previous infection with a resistant strain. We attempted to obtain past DST records of previously treated cases via the public health center; however, many of these records could not be traced. Third, some previously treated cases in the private sector may have been misclassified as new cases. Moreover, new drug-resistant patients may have a greater tendency to visit a tertiary hospital, which may have affected our results, as the new drug-resistant and MDR rates were higher than those reported from public health centers.
In conclusion, the TB drug-resistance rate, particularly that of MDR TB, remains high at a private referral hospital, and the drug-resistance rate did not decrease significantly from 2006 to 2013. However, the proportions of MDR and XDR TB cases since 2006 are suspected to have decreased consistently among previously treated cases. This finding may be a result of the significant increase in the proportion of relapse cases and a decrease in the proportion of TAI cases over time. In addition, we found no remarkable difference in the drug-resistance rate between PTB and EPTB. However, a national survey of the prevalence of drug-resistant TB should be conducted to obtain the most accurate drug-resistance status in South Korea, including the private sector. However, this will require a vast amount of time and financial investment.






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





