To the Editor,
Henoch-Schönlein purpura (HSP) is a systemic, small-vessel vasculitic syndrome characterized by palpable purpura, arthralgia, abdominal pain, and renal disease. The underlying pathogenesis of HSP remains unclear. Streptococcal infections, staphylococcal infections, vaccinations, medications including antibiotics, and insect bites have been implicated as possible triggers, although some cases lack a clear precipitating event [1,2]. One rare report indicated that infective endocarditis triggers HSP. We share an interesting case of HSP in a patient complicated with infective endocarditis.
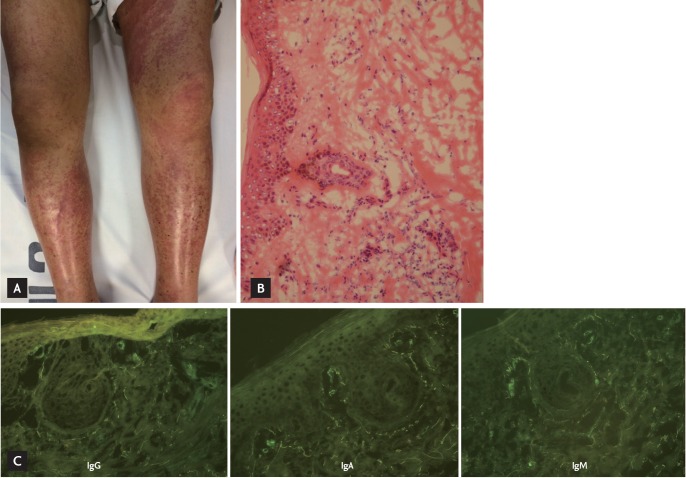
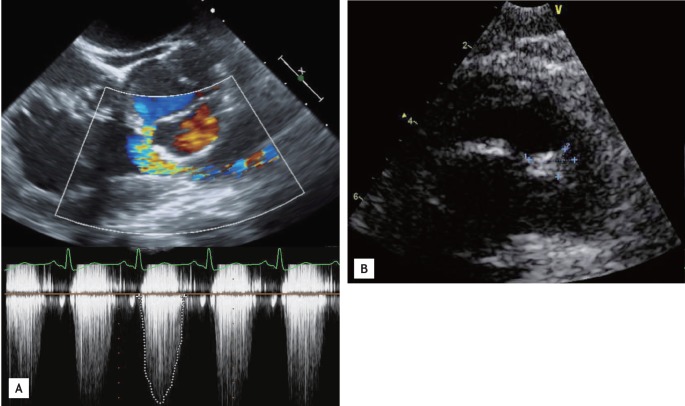
A 49-year-old male was transferred to our department with microscopic hematuria, proteinuria, decreased renal function, and purpura. He had been treated for arthralgia with nonsteroidal anti-inflammatory drugs (NSAIDs), acupuncture, and physical therapy for 3 months. However, he developed intermittent fever, edema, and a purpuric rash on both lower legs 1 month ago. Laboratory results obtained 10 days before revealed an increase in serum creatinine from 2.5 to 3.2 mg/dL, compared to the level 3 months prior. He had developed a high fever of 39℃, and pneumonic infiltration was detected on a chest X-ray 5 days prior. The patient had no remarkable history, except 3 months of NSAID treatment and 1 week of antibiotics (ceftriaxone and clarithromycin) for pneumonia. Vital signs at admission were: blood pressure, 127/76 mmHg; heart rate, 80 beats per minute; and body temperature, 36.2℃. He complained of intermittent fever, and a physical examination revealed marked pitting edema, a purpuric rash over the entire area of both legs (Fig. 1A), a regular heartbeat with a systolic murmur, and coarse breathing sounds with crackling in both lower lung fields. The laboratory results were: white blood cell count, 11,220/mm3; multi-nucleated neutrophils, 72.5%; hemoglobin, 9.1 g/dL; platelet count, 388,000/mm3; erythrocyte sedimentation rate, 37 mm/hr; C-reactive protein, 10.17 mg/dL; serum blood urea nitrogen, 31.4 mg/dL; creatinine, 2.5 mg/dL; protein, 8.8 g/dL; albumin, 2.3 g/dL, with a reversed albumin-globulin ratio; ferritin, 799.5 ng/mL; urinalysis, 4+ protein; and microhematuria on a urine sediment examination. A plain chest X-ray revealed multifocal consolidation in both lungs. Ceftriaxone and clarithromycin continued to be administered for pneumonia. We checked autoimmune antibodies, complement, immunoglobulins, and urine chemistry. A further laboratory evaluation revealed 1.2% fractional excretion of sodium, suggesting renal-type acute renal failure, a 1:100 positive antinuclear antibody titer (speckled and cytoplasmic type), increased rheumatoid factor of 852.1 IU/mL, and a decrease in the C3 level to 56 mg/dL. A skin biopsy was obtained from the purpuric rash on the left lower leg, and leukoclastic vasculitis with deposits of immunoglobulin G (IgG), IgA, and IgM were detected in the vascular endothelium by immunofluorescence (Fig. 1B and 1C). Seven glomeruli were obtained from the renal biopsy; three of the seven showed focal segmental glomerulosclerosis, and another three of seven were crescents with IgG, IgA, IgM, C3, C1q, and fibrinogen deposits detected in the mesangium on immunofluorescence (Fig. 2). The purpuric rash was localized to the lower limbs, with renal involvement, joint pain, and leukoclastic vasculitis, which were compatible with HSP. Steroids were considered due to the impaired renal function and the crescent glomeruli. A contrast-enhanced chest computed tomography (CT) scan and bronchoscopy were performed, as the pneumonia did not improve with antibiotics. No endobronchial lesion was detected on bronchoscopy, but the chest CT scan showed multiple cavitary nodules with ill-defined consolidation in the right lower lobe, left upper lobe, and left lower lobe, suggesting septic pneumonia associated with embolic infarction. A transthoracic echocardiogram was performed and revealed an echogenic mass in the right ventricular outflow tract, suggestive of vegetative growth, with significant pulmonary valve stenosis (PS) (Vmax, 4.7 m/sec; mean pressure gradient, 46.3 mmHg) and a small left to right shunt flow in the ventricular septum just below the aortic valve near the tricuspid valve, suggesting a perimembranous ventricular septal defect (VSD) (defect size, 4.1 mm). Therefore, a transesophageal echocardiogram was performed and also revealed subvalvular PS with right ventricular hypertrophy and a ~10-mm vegetative growth on the pulmonary valve (PV) (Fig. 3). Thus, he had a predisposing heart condition, vegetation on the PV, septic emboli in the lung, and renal involvement, which were compatible with the clinical criteria for infective endocarditis. The antibiotics were changed to piperacillin/tazobactam and gentamicin on hospital day 8. No steroids were administered, due to concern about aggravating the infective endocarditis. The fever subsided after changing the antibiotics, and the laboratory findings revealed improved leukocytosis and C-reactive protein level. Because the subvalvular PS with a small VSD was thought to be the reason for the vegetation on the PV as well as the septic embolism, he underwent closure of the VSD and had no postoperative complications. Creatinine decreased to 1.22 mg/dL, and urinary protein decreased from 4+ to trace. He was discharged and was scheduled for follow-up as an outpatient.
In this case, we were able to distinguish HSP from other vasculitis syndromes and conditions that can mimic vasculitis using the clinical features and histopathological findings. Infective endocarditis can also be detected by a physical examination and echocardiography. Because no detailed physical examination or cardiac evaluation had been performed at the primary hospital, it was difficult to determine the association between HSP and infective endocarditis. We did not culture his blood for bacteria, as he had already taken antibiotics for 1 week. We speculate that he had bacteremia, considering the vegetation on the PV and the pulmonary septic emboli. The infective endocarditis may have preceded HSP because infective endocarditis can be subacute, and we found no other preceding cause for HSP. Furthermore, the HSP improved after treating the infective endocarditis.
Renal failure can accompany up to one-third of endocarditis cases, and the pathology is similar to the deposition of immune complexes on the glomerular basement membrane and embolic renal infarct observed in this patient [1]. In this case, infective endocarditis was complicated by HSP. The pathogenic mechanism was deposition of immune complexes consisting of IgA and antigen in the small vessels of the skin and renal mesangium [2]. The mechanism for the development of HSP in a patient with infective endocarditis is unclear. Drug-induced HSP was less likely in this case, as arthralgia preceded antibiotic use. Further study is needed to evaluate the mechanism by which infective endocarditis leads to HSP.
A case of HSP complicating infective endocarditis is quite rare, as only eight cases have been reported, and none of the cases involved PS and VSD. No similar cases have been reported in Korea. All eight cases developed purpura on both legs with renal involvement, and arthralgia developed in five cases. Streptococcus was cultured in six cases, and Staphylococcus was cultured in the other two cases [1,2,3,4]. Our case resembled these eight reported cases. This case suggests an association between HSP and infective endocarditis but more cases are needed to identify the nature of the association.
Three crescents were observed among seven glomeruli in the renal pathological findings of our patient, which is HSP nephritis pathological grade 4. High-dose steroid therapy was needed because this pathological grade predicts a poor prognosis [5]. However, we could not use steroids due to concerns about aggravating the infective endocarditis. Although the patient was treated only with antibiotics and closure of the VSD for the infective endocarditis, renal function improved spontaneously. This finding suggests that HSP is self-limiting and that infective endocarditis induces HSP. Glomerulonephritis occurring in a patient with infective endocarditis usually improves only with antibiotic treatment.
Finally, the cardiac murmur detected on the physical examination may have been a cue to diagnose infective endocarditis in this case. Detecting the murmur led to finding the vegetative growth on echocardiography and the septic emboli with pneumonia on a chest CT scan. These findings suggest that clinical suspicion after a physical examination is important to diagnose infective endocarditis.




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





