 |
 |
| Korean J Intern Med > Volume 30(5); 2015 > Article |
|
Abstract
Background/Aims:
The purpose of this study was to determine the correlations between inflammatory factors—including absolute lymphocyte count, lactate dehydrogenase, β2-microglobulin, albumin, C-reactive protein, and ferritin—and the prognosis for survival in patients with multiple myeloma (MM) treated with induction chemotherapy containing thalidomide and who underwent autologous stem cell transplantation (ASCT).
Methods:
Data from patients at 13 university hospitals in South Korea were collected retrospectively between December 2005 and May 2013.
Results:
The median age of the 232 patients was 57 years (range, 33 to 77) and the male to female ratio was 1.09:1. In the multivariate analysis, fewer than two combined abnormal inflammatory factors was the only independent prognostic factor for superior progression-free survival (relative risk [RR], 0.618; 95% confidence interval [CI], 0.409 to 0.933; p = 0.022), and platelet count > 100 × 109/L and fewer than two combined abnormal inflammatory factors were independent prognostic factors for superior overall survival (RR, 4.739; 95% CI, 1.897 to 11.839; p = 0.001 and RR, 0.263; 95% CI, 0.113 to 0.612; p = 0.002, respectively).
Conclusions:
Patients with two or more than two combined inflammatory factors who were treated with thalidomide induction chemotherapy and who underwent ASCT showed significantly shorter survival compared to those with fewer than two combined inflammatory factors. These results could be helpful for predicting prognosis in patients with MM.
Many advances have been made in treatments for multiple myeloma (MM), as novel agents, such as thalidomide, bortezomib, and lenalidomide, have been developed [1-3]. In particular, thalidomide was the first novel agent introduced that improved the overall response rate (ORR) in transplant eligible or ineligible patients with MM [4-6]. It was first confirmed in 1999 that thalidomide was active in patients with relapsed and/or refractory MM. Since then, thalidomide has become an important part of MM treatment as initial therapy for previously untreated patients, as maintenance therapy following definitive treatment, and as salvage therapy [3-9].
Many prognostic factors have been investigated together with the development of MM treatments. The prognostic parameters correlated with survival in patients with MM are serum β2-microglobulin (β2MG), albumin, absolute lymphocyte count (ALC), C-reactive protein (CRP), lactate dehydrogenase (LDH), serum ferritin, bone marrow plasma cell percentage, serum creatinine, hemoglobin, platelet count, age, Eastern Cooperative Oncology Group performance status, and serum free light chains or their ratio [10-16]. Prognostic scoring systems have also been developed, such as the Durie-Salmon Staging System (D-S stage) and the International Staging System (ISS) [17,18]. However, the significance of the D-S stage or the ISS for predicting prognosis decreased after the development and introduction of novel agents. Therefore, a new prognostic model or factors are needed for a more precise prediction of prognosis. Cytogenetic abnormalities, which are confirmed by conventional chromosomal studies or fluorescence in situ hybridization (FISH), have emerged as important prognostic markers [11,19-21]. However, FISH is used less frequently to predict prognosis in clinical practice because of the high cost and time expenditure involved. We identified useful prognostic clinical or laboratory factors that are easily tested in patients with newly diagnosed MM.
Serological inflammatory markers, such as ALC, LDH, β2MG, albumin, CRP, and ferritin, have been correlated with the prognosis of patients with MM [10,13,15,16]. However, few studies of the prognostic impact of combined inflammatory status in patients with MM have been conducted.
Therefore, the purpose of this study was to determine the correlation between the frequency of combined inflammatory factors—including ALC, LDH, β2MG, and albumin—and the prognosis for survival in patients with MM treated with induction chemotherapy containing thalidomide who also underwent autologous stem cell transplantation (ASCT).
Data from patients at 13 university hospitals in South Korea were collected retrospectively between December 2005 and May 2013. All patients were treated with thalidomide-containing chemotherapy and then underwent ASCT. Patients who were treated with an induction chemotherapy regimen without thalidomide or those who underwent tandem ASCT were excluded. The thalidomide combination induction chemotherapy consisted of thalidomide (fixed oral dose of 50 to 100 mg on days 1 to 28) plus intravenous (IV) dexamethasone (20 mg/m2 or oral on days 1 to 4 and days 15 to 18) every 4 weeks (TD), cyclophosphamide (fixed oral dose of 150 mg on days 1 to 4 every 4 weeks) plus TD (CTD), doxorubicin (9 mg/m2 IV rapid infusion on days 1 to 4) plus TD (TAD) every 4 weeks, and thalidomide plus vincristine 4 mg/m2 IV infusion on days 1 to 4, 9 mg/m2 doxorubicin IV rapid infusion on days 1 to 4, and 20 mg/m2 dexamethasone IV or oral on days 1 to 4, 9 to 12, and 17 to 20 (VAD) every 4 weeks. Only four patients were treated with this thalidomide plus VAD regimen. Low-dose aspirin was used to prevent thrombosis during induction chemotherapy. A total of 221 patients who achieved PR or better underwent ASCT with high-dose melphalan (100 to 200 mg/m2).
Progression-free survival (PFS) and overall survival (OS) were estimated using clinical parameters in all patients, including age, sex, hemoglobin, platelet count, ALC, β2MG, serum albumin, LDH, cytogenetic risk, ISS score, response before ASCT, and inflammatory score. A conventional chromosomal study was performed in 204 patients and FISH including t(11;14), t(4;14), 13q deletion, 17p deletion, t(14;16), tri 1q, and t(14;20) were also performed in more than 100 patients. The cytogenetic risk, which was determined by conventional cytogenetics or FISH, was divided into standard, intermediate, and high risk. Standard risk included normal cytogenetics and hyperdiploidy, t(11;14), and t(6;14). Intermediate risk included t(4;14), 13q deletion, and hypodiploidy; and high risk included 17p deletion, t(14;16), and t(14;20) [19,21-23]. Each inflammatory factor of ALC level < 1.0 × 109/L, β2MG level > 3.5 mg/L, serum albumin < 3.5 g/dL, and LDH level above normal was defined as a combined abnormal inflammatory factor, and the sum of these abnormal factors was analyzed. Responses before and after ASCT were assessed according to the International Myeloma Working Group uniform response criteria [24].
We investigated independent prognostic factors associated with survival using the listed clinical and laboratory parameters. PFS was defined as the duration from the start date of induction chemotherapy to the date of disease progression, relapse, or death from any cause after ASCT. OS was calculated from the date of diagnosis to the date of death from any cause or the final follow-up date. Survival probabilities were calculated according to the Kaplan-Meier method and compared using the log-rank test. The Cox proportional hazards regression model was used for the multivariate analysis of independent prognostic factors of survival. Information about the baseline medical status and treatment modalities was collected from the medical records. Approval for this study was obtained from each Institutional Review Board.
The median age of the 232 patients was 57 years (range, 33 to 77), and the male to female ratio was 1.09:1. The median serum monoclonal protein level was 2.4 g/dL (range, 0.0 to 9.8), the median hemoglobin level was 10.1 g/dL (range, 4.2 to 15.4), and the median platelet count was 196.5 × 109/L (range, 37.0 to 454.0). Standard cytogenetic risk was found in 173 patients (74.6%), intermediate risk in 39 (16.8%), and high risk in 20 (8.6%). The frequencies of the combined abnormal inflammatory factors of 0, 1, 2, 3, and 4 were found in 64 (27.6%), 75 (32.3%), 67 (28.9%), 18 (7.8%), and five (2.2%) patients, respectively. Other clinical and laboratory characteristics are summarized in Table 1.
The response rates achieved before and after ASCT were as follows: complete response (CR) or stringent CR in 81 (34.9%) and 142 (60.2%) patients, very good partial response (PR) in 61 (26.3%) and 47 (20.3%), PR in 79 (34.1%) and 32 (13.8%), and < PR in seven (3.1%) and five (2.3%). The median follow-up duration was 24.5 months (range, 6.9 to 108.9). The median PFS was 31.93 months (range, 25.1 to 38.8), and the median OS was not reached during the follow-up (Fig. 1).
The following factors in the univariate analysis were associated with a greater than 2-year PFS (Table 2): low β2MG (< 3.5 mg/L vs. ≥ 3.5 mg/L, 74.8% vs. 54.5%, p = 0.022), normal LDH (normal vs. abnormal, 67.4% vs. 50.5%, p = 0.009), low cytogenetic risk (standard vs. intermediate vs. high, 68.5% vs. 48.5% vs. 61.5%, p = 0.018), and having fewer than two combined abnormal inflammatory factors (< 2 vs. ≥ 2, 72.1% vs. 53.3%, p = 0.004) (Fig. 2A). The following factors were associated with a greater than 2-year OS (Table 2): higher hemoglobin level (< 10 g/dL vs. ≥ 10 g/dL, 84.6% vs. 96.1%, p = 0.042), higher platelet count (< 100 × 109/L vs. ≥ 100 × 109/L, 48.5% vs. 93.1%, p < 0.001), lower β2MG (< 3.5 mg/L vs. ≥ 3.5 mg/L, 98.7% vs. 84.7%, p < 0.001), normal LDH (normal vs. abnormal, 91.6% vs. 69.5%, p < 0.001), lower cytogenetic risk (standard vs. intermediate vs. high, 92.1% vs. 90.3% vs. 83.8%, p = 0.028), lower ISS (I, II, and III, 98.0%, 90.3%, and 83.8%, respectively; p = 0.024), and having fewer than two combined abnormal inflammatory factors (< 2 vs. ≥ 2, 98.1% vs. 81.1%, p < 0.001) (Fig. 2B). The survival curves of 2-year PFS and OS according to the inflammatory factors (Fig. 2A and 2B) are compared to 2-year PFS and OS according to ISS in Fig. 2C and 2D. The factors correlated with longer survival in the univariate analysis were included in a multivariate analysis, excluding β2MG and LDH because they were already included as abnormal inflammatory factors. ISS was also excluded from the multivariate analysis because β2MG and albumin were already included as abnormal inflammatory factors. In the multivariate analysis, abnormal inflammatory factors (< 2) was the only independent prognostic factor for superior PFS (relative risk [RR], 0.618; 95% confidence interval [CI], 0.409 to 0.933; p = 0.022), and platelet count > 100 × 109/L, and fewer than two abnormal inflammatory factors were the only independent prognostic factors for greater OS (RR, 4.793; 95% CI, 1.897 to 11.839; p = 0.001 and RR, 0.263; 95% CI, 0.113 to 0.612; p = 0.002, respectively) (Table 3).
In this study, various parameters were estimated to identify prognostic factors for survival in patients who were treated with thalidomide-containing chemotherapy and who underwent ASCT. In the univariate analysis, higher β2MG (≥ 3.5 mg/L), abnormal LDH, poor cytogenetic risk, and two or more combined abnormal inflammatory factors were associated with a < 2-year PFS. Lower hemoglobin level (< 10 g/dL), lower platelet count (< 100 × 109/L), higher β2MG (≥ 3.5 mg/L), abnormal LDH, poor cytogenetic risk, higher ISS, and having two or more combined abnormal inflammatory factors were associated with < 2-year OS. Only two or more combined abnormal inflammatory factors was an independent prognostic factor for PFS in the multivariate analysis, and platelet count as well as combined abnormal inflammatory factors were independent prognostic factors for OS. Lower platelet count has been associated previously with a poor prognosis in a study reporting that low platelet count as well as low calcium, LDH, CRP, and performance status were consistent with rapid attrition in such patients due to disease aggressiveness or co-morbidities [25].
In the present study, the combined abnormal inflammatory factors included LDH, β2MG, albumin, and ALC. The reason for including albumin and ALC, which did not show significant results in the univariate analysis, was that the purpose of this study was not to show correlations between individual inflammatory factors and prognosis or to create a new prognostic index but to determine if there was an association between a number of combined inflammatory parameters and prognosis for survival. We demonstrated that having two or more combined abnormal inflammatory factors was correlated with short survival. These results suggest that patients with MM who have multiple combined inflammatory factors may show a poor prognosis. Some studies have reported mechanisms that may explain why inflammatory factors are associated with prognosis. Low serum albumin level has also been associated with advanced age and poor performance status, which are known poor prognostic factors in patients with MM [26]. Serum albumin level was previously shown to negatively correlate with serum interleukin 6 (IL-6) levels and reflects IL-6 effects on the liver, indicating its role as a potent myeloma cell growth factor in vitro [26]. Serum IL-6 level is also correlated with disease severity in patients with plasma cell dyscrasias [27]. Serum soluble IL-6 receptor (sIL-6R) level is correlated with β2MG, CRP, ferritin, and LDH concentrations and is believed to be correlated with the duration of disease-free survival [28,29].
Therefore, patients with two or more combined inflammatory factors who were treated with thalidomide induction chemotherapy and who underwent ASCT showed significantly shorter survival than patients with fewer than two combined inflammatory factors. These results might be helpful for predicting prognosis in patients with MM. However, the ORR before ASCT in this study was higher compared to those of other studies, including patients treated with thalidomide. This is likely because most hematologists consider ASCT only after patients achieve a response greater than PR in South Korea. Moreover, this study had some limitations including the fact that CRP, ferritin, and other cytokines, such as IL-6 and sIL-6R, which were regarded as inflammatory factors, were not analyzed independently because of insufficient retrospective data. Therefore, further prospective studies are needed to confirm the correlations between prognosis and these inflammatory factors in transplantation-eligible or -ineligible patients with MM treated with novel agents, such as thalidomide, bortezomib, or lenalidomide.
1. Serologic inflammatory markers have been reported to be correlated with the prognosis of patients with multiple myeloma.
2. The inflammatory markers were independent prognostic factors for progression-free survival and overall survival in patients who were treated with thalidomide induction chemotherapy and who underwent autologous stem cell transplantation.
Acknowledgments
This study was supported by a grant from Kosin University College of Medicine. The authors are indebted to Chul Won Choi for advising of this manuscript. A special acknowledgment is extended to the Korean Multiple Myeloma Working Party (KMMWP) for supporting our data.
Figure 1.
(A) Median progression-free survival was 31.93 months (range, 25.1 to 38.8), and (B) median overall survival was not reached during the follow-up in patients treated with thalidomide induction chemotherapy and who underwent autologous stem cell transplantation.
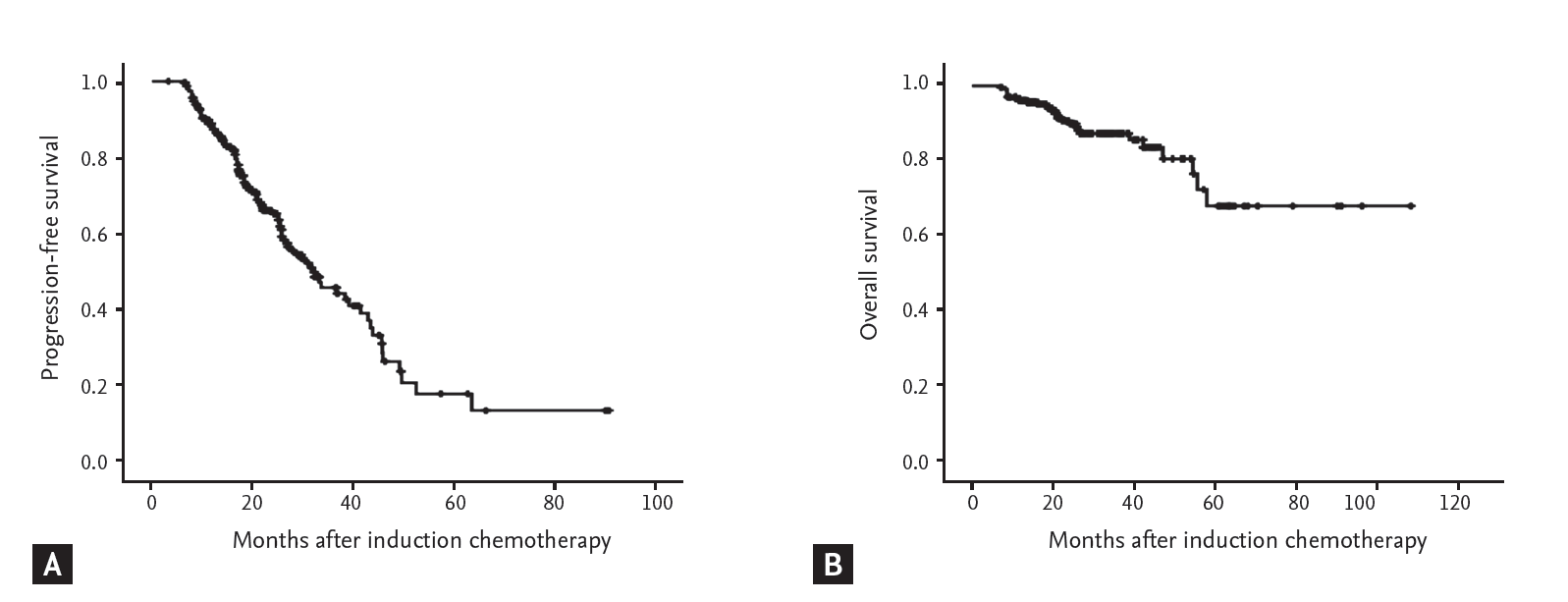
Figure 2.
The 2-year progression-free survival rate and 2-year overall survival rate were superior in patients with two or more combined abnormal inflammatory factors compared to those with fewer than two (A: 72.1% vs. 53.3%, p = 0.004; B: 98.1% vs. 81.1%, p < 0.001, respectively). The inflammatory factor survival curves in (A) and (B) were more significant than the International Scoring System (ISS) stage survival curves in (C, p = 0.062) and (D, p = 0.024).

Table 1.
Clinical and laboratory characteristics (n = 232)
| Characteristic | Value |
|---|---|
| Age, yr | 57 (33-77) |
| Gender | |
| Male | 122 (52.6) |
| Female | 111 (47.4) |
| Follow-up duration, mon | 24 (7-107) |
| Multiple myeloma subtype | |
| IgG, κ, or λ | 124 (53.4) |
| IgA, κ, or λ | 35 (15.1) |
| Light chain disease | 62 (26.7) |
| Othersa | 11 (4.7) |
| Serum monoclonal protein, g/dL | 2.4 (0.0-9.8) |
| Hemoglobin, g/dL | 10.1 (4.2-15.4) |
| Platelet count, x 109/L | 196.5 (37-454) |
| Absolute lymphocyte count, x 109/L | 1.730 (0.025-8.100) |
| Serum calcium, mg/dL | 9.1 (1.3-15.4) |
| Serum total protein, g/dL | 8.2 (2.9-15.2) |
| Serum albumin, g/dL | 3.65 (1-5.6) |
| Serum β2-microglobulin, mg/L | 3.4 (1.16-32) |
| Lactate dehydrogenase, IU/L | 310 (80-1,940) |
| Cytogenetic risk | |
| Standard | 176 (75.9) |
| Intermediate | 36 (15.5) |
| High | 20 (8.6) |
| International Staging System | |
| I | 78 (33.6) |
| II | 75 (32.3) |
| III | 69 (29.7) |
| Unknown | 10 (4.3) |
| Abnormal inflammatory factors | |
| 0 | 64 (27.6) |
| 1 | 75 (32.3) |
| 2 | 67 (28.9) |
| 3 | 18 (7.8) |
| 4 | 5 (2.2) |
| Treatment regimen | |
| TD | 129 |
| CTD | 96 |
| TAD | 3 |
| TVAD | 4 |
| Response before ASCT | |
| CR or sCR | 81 (34.9) |
| VGPR | 61 (26.3) |
| PR | 79 (34.1) |
| < PR | 7 (3.1) |
| Unknown | 4 (1.7) |
Values are presented as median (range), number (%), or number.
TD, thalidomide plus dexamethasone; CTD, cyclophosphamide plus thalidomide and dexamethasone; TAD, thalidomide plus doxorubicin and dexamethasone; TVAD, thalidomide plus VAD (vincristine, doxorubicin and dexamethasone); ASCT, autologous stem cell transplantation; CR, complete response; sCR, stringent complete response; VGPR, very good partial response; PR, partial response.
Table 2.
Clinical and laboratory values associated with progression-free survival and overall survival in the univariate analysis
Table 3.
Multivariate analysis for survival
REFERENCES
1. Palumbo A, Facon T, Sonneveld P, et al. Thalidomide for treatment of multiple myeloma: 10 years later. Blood 2008;111:3968–3977.


2. Cavo M, Tacchetti P, Patriarca F, et al. Bortezomib with thalidomide plus dexamethasone compared with thalidomide plus dexamethasone as induction therapy before, and consolidation therapy after, double autologous stemcell transplantation in newly diagnosed multiple myeloma: a randomised phase 3 study. Lancet 2010;376:2075–2085.

3. Morgan GJ, Davies FE. Role of thalidomide in the treatment of patients with multiple myeloma. Crit Rev Oncol Hematol 2013;88 Suppl 1:S14–S22.

4. Moreau P, Touzeau C. Initial treatment of transplant candidates with multiple myeloma. Semin Oncol 2013;40:585–591.


5. Ozaki S, Shimizu K. Autologous stem cell transplantation in elderly patients with multiple myeloma: past, present, and future. Biomed Res Int 2014;2014:394792.




6. Ahn SY, Jung SH, Joo YD, et al. Early response-based intensification of primary therapy in newly diagnosed multiple myeloma patients who are eligible for autologous stem cell transplantation: phase II study. Ann Hematol 2014;93:1571–1577.


7. Minarik J, Sandecka V, Maisnar V, et al. 10 Years of experience with thalidomide in multiple myeloma patients: report of the Czech Myeloma Group. Leuk Res 2013;37:1063–1069.


8. Vande Broek I, Jacobs P. Continuous treatment in multiple myeloma: the future? Transfus Apher Sci 2013;49:147–150.


9. Lokhorst HM, Schmidt-Wolf I, Sonneveld P, et al. Thalidomide in induction treatment increases the very good partial response rate before and after high-dose therapy in previously untreated multiple myeloma. Haematologica 2008;93:124–127.


10. Copur S, Kus S, Kars A, Renda N, Tekuzman G, Firat D. Lactate dehydrogenase and its isoenzymes in serum from patients with multiple myeloma. Clin Chem 1989;35:1968–1970.


11. Engelhardt M, Terpos E, Kleber M, et al. European Myeloma Network recommendations on the evaluation and treatment of newly diagnosed patients with multiple myeloma. Haematologica 2014;99:232–242.



12. Kyrtsonis MC, Maltezas D, Tzenou T, Koulieris E, Bradwell AR. Staging systems and prognostic factors as a guide to therapeutic decisions in multiple myeloma. Semin Hematol 2009;46:110–117.


13. Bataille R, Boccadoro M, Klein B, Durie B, Pileri A. C-reactive protein and beta-2 microglobulin produce a simple and powerful myeloma staging system. Blood 1992;80:733–737.


14. Kyrtsonis MC, Vassilakopoulos TP, Kafasi N, et al. Prognostic value of serum free light chain ratio at diagnosis in multiple myeloma. Br J Haematol 2007;137:240–243.


15. Strasser-Weippl K, Ludwig H. Ferritin as prognostic marker in multiple myeloma patients undergoing autologous transplantation. Leuk Lymphoma 2014;55:2520–2524.


16. Ege H, Gertz MA, Markovic SN, et al. Prediction of survival using absolute lymphocyte count for newly diagnosed patients with multiple myeloma: a retrospective study. Br J Haematol 2008;141:792–798.


17. Greipp PR, San Miguel J, Durie BG, et al. International staging system for multiple myeloma. J Clin Oncol 2005;23:3412–3420.


18. Durie BG, Salmon SE. A clinical staging system for multiple myeloma: correlation of measured myeloma cell mass with presenting clinical features, response to treatment, and survival. Cancer 1975;36:842–854.


19. Lim JH, Seo EJ, Park CJ, et al. Cytogenetic classification in Korean multiple myeloma patients: prognostic significance of hyperdiploidy with 47-50 chromosomes and the number of structural abnormalities. Eur J Haematol 2014;92:313–320.


20. Palumbo A, Rajkumar SV, San Miguel JF, et al. International Myeloma Working Group consensus statement for the management, treatment, and supportive care of patients with myeloma not eligible for standard autologous stem-cell transplantation. J Clin Oncol 2014;32:587–600.



21. Mikhael JR, Dingli D, Roy V, et al. Management of newly diagnosed symptomatic multiple myeloma: updated Mayo Stratification of Myeloma and Risk-Adapted Therapy (mSMART) consensus guidelines 2013. Mayo Clin Proc 2013;88:360–376.


22. Rajkumar SV. Multiple myeloma: 2013 update on diagnosis, risk-stratification, and management. Am J Hematol 2013;88:226–235.


23. Oh S, Koo DH, Kwon MJ, et al. Chromosome 13 deletion and hypodiploidy on conventional cytogenetics are robust prognostic factors in Korean multiple myeloma patients: web-based multicenter registry study. Ann Hematol 2014;93:1353–1361.


24. Cavo M, Rajkumar SV, Palumbo A, et al. International Myeloma Working Group consensus approach to the treatment of multiple myeloma patients who are candidates for autologous stem cell transplantation. Blood 2011;117:6063–6073.



25. Barlogie B, Bolejack V, Schell M, Crowley J. Prognostic factor analyses of myeloma survival with intergroup trial S9321 (INT 0141): examining whether different variables govern different time segments of survival. Ann Hematol 2011;90:423–428.



26. Kim JE, Yoo C, Lee DH, Kim SW, Lee JS, Suh C. Serum albumin level is a significant prognostic factor reflecting disease severity in symptomatic multiple myeloma. Ann Hematol 2010;89:391–397.


27. Bataille R, Jourdan M, Zhang XG, Klein B. Serum levels of interleukin 6, a potent myeloma cell growth factor, as a reflect of disease severity in plasma cell dyscrasias. J Clin Invest 1989;84:2008–2011.






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



