 |
 |
| Korean J Intern Med > Volume 30(5); 2015 > Article |
|
Abstract
Background/Aims:
A number of genome-wide and candidate gene association studies have identified polymorphisms associated with telomere length in Caucasian populations. This study was conducted to determine the impacts of 17 polymorphisms identified in Caucasians on telomere length in a Korean population.
Methods:
Ninety-four healthy individuals were enrolled in this study. Relative telomere length of chromosomes from peripheral blood samples was measured using quantitative polymerase chain reaction.
Results:
Two polymorphisms, rs10936599 of MYNN and rs412658 of ZNF676, were found to be associated w ith telomere length (under dominant model, p = 0.04; under recessive model, p = 0.001). Three polymorphisms, rs2853669, rs7705526, and rs2736108, at the TERT locus were also associated with telomere length (under recessive model, p = 0.01, p = 0.02, and p = 0.01, respectively). The genotypes of the five polymorphisms associated with short telomere length were considered bad genotypes; telomere length was significantly decreased with increasing number of bad genotypes (p= 1.7 ├Ś 10ŌĆō5).
Telomeres are repetitive, non-coding DNA sequences located at the ends of each chromosome. In humans, a six-nucleotide motif (TTAGGG) is repeated thousands of times, reaching a length of 10 to 15 kb [1]. By capping the ends of chromosomes, telomeres protect the chromosomes from degradation, end-to-end fusion, and atypical recombination [1]. Telomeres are progressively shortened during each mitotic cell division by 50 to 200 bp because of the incomplete replication of linear chromosomes by conventional DNA polymerase (also known as the end replication problem) [1-3]. Once telomeres become critically short, they lose their protective function, and cellular senescence or apoptosis occurs. This progressive shortening of telomeres limits the replicative capacity of human somatic cell division and serves as a mitotic clock for somatic cells [4]. Telomere shortening is also considered one of the mechanisms of human aging and age-related diseases, such as heart disease and atherosclerosis, as well as cancer [5-7]. Telomere shortening can be recovered by the action of telomerase. Human telomerase consists of two molecules, each comprising telomerase reverse transcriptase (TERT), telomerase RNA component (TERC), and dyskerin [8]. Alterations in telomerase activity are also associated with a diverse range of human diseases, including cancer [9-11].
A number of recent genome-wide association studies (GWASs) identified single nucleotide polymorphisms (SNPs) related to leukocyte telomere length [12-16]. A number of polymorphisms in TERT and TERC can alter telomerase activity and may therefore affect telomere length [17-19]. However, these studies were mainly conducted in Caucasian populations. The effects of genetic polymorphisms on telomere length may differ among ethnic groups. Therefore, the present study was performed to determine the impacts of the genetic variants identified in Caucasians on telomere length in a Korean population.
The study population consisted of 94 randomly selected individuals from a pool of healthy volunteers that visited the General Health Check-Up Center of Kyungpook National University Hospital between January and July 2005. All participants were ethnic Koreans residing in Daegu or the surrounding regions. Blood samples were provided by the National Biobank of Korea, which is supported by the Ministry of Health, Welfare, and Family Affairs. This study was approved by the Institutional Review Board of the Kyungpook National University Hospital (approval no. KNUHBIO-09-1018).
Genomic DNA was isolated from peripheral blood using the QuickGene DNA whole blood kit (Fujifilm, Tokyo, Japan) in accordance with the manufacturerŌĆÖs protocol. Relative telomere length was measured using a quantitative polymerase chain reaction (PCR) method described elsewhere [20,21]. The relative telomere length was expressed as the ratio of the telomere repeat copy number to the copy number of a single gene, i.e., the human ╬▓-globin gene.
Thirteen SNPs found to be associated with telomere length in five GWASs [12-16] were selected. Among these 13 SNPs, three (rs3027234, rs6028466, and rs654128) are very rare in Asians (according to HapMap data, minor allele frequency = 0.03, 0.05, and 0.07, respectively) and were therefore excluded from this study. Seven SNPs were selected from candidate gene association studies [17-19] that reported associations between polymorphisms in TERT or TERC and telomere length. A total of 17 polymorphisms were selected and genotyped using a sequenome mass spectrometry-based genotyping assay. Samples that could not be scored using this approach were re-genotyped by PCR-restriction fragment length polymorphism assay. For quality control, the genotyping analysis was performed in a blinded manner with regard to the subjects. Approximately 10% of the samples were selected randomly for repeat genotyping by a different investigator, and the results showed 100% concordance.
Statistical analysis was performed using R version 2.15.3. Means and standard deviations are reported for continuous variables, and numbers and percentages are reported for categorical variables. Telomere lengths of the global, dominant, or recessive model were compared by one-way analysis of variance or independent t test for each SNP. After adjusting for age, gender, and cigarette pack-years (for smokers), mean telomere lengths were compared by analysis of covariance.
The characteristics of the study population are shown in Table 1. The mean relative telomere length was 2.30 (standard deviation, 0.60). The mean telomere length was shorter in heavy smokers (pack-years > 34) than in lighter smokers (pack-years > 34; p = 0.05). No significant differences in telomere length related to age or gender were found (Table 1). The genotype distributions of the 17 SNPs were in Hardy-Weinberg equilibrium.
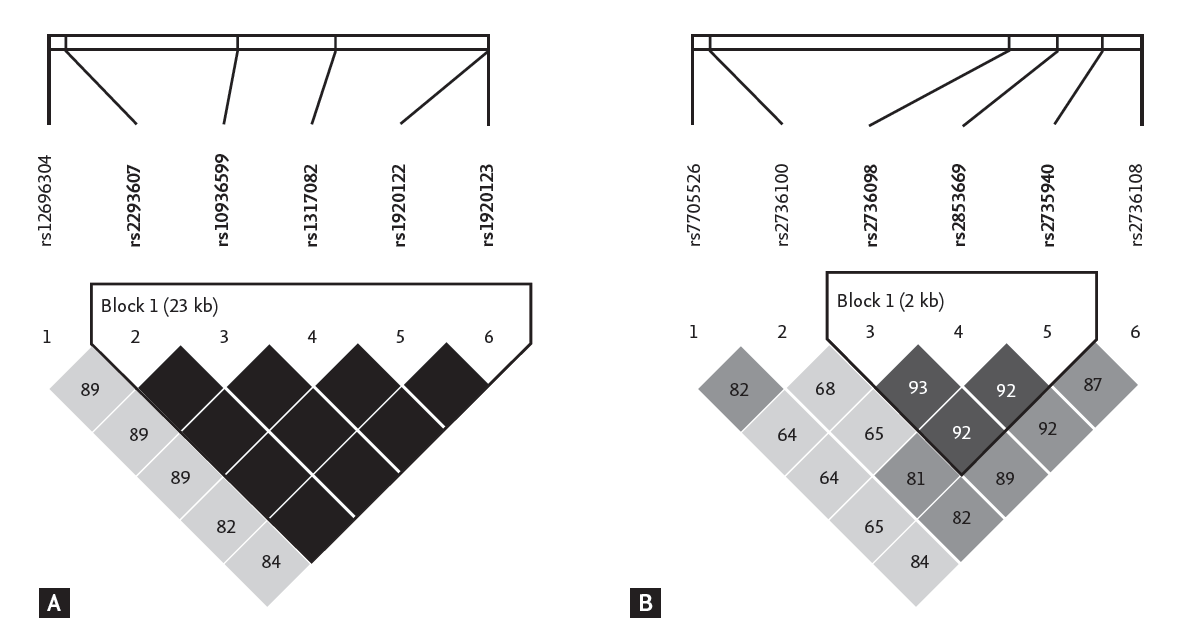
Among the 17 SNPs evaluated, eight were significantly associated with telomere length following adjustments for age, gender, and pack-years of smoking (Table 2). The rs10936599 and rs1317082 of MYNN and rs2293607 of TERC were associated with telomere length under the dominant model for the variant allele of each SNP (p = 0.04, p = 0.03, and p = 0.04, respectively). These three SNPs were within a region of approximately 15 kb on chromosome 3q26.2 (169482335 to 169497585 bases) and were in complete linkage disequilibrium (LD) (Fig. 1) [22]. The rs412658 of the zinc finger protein 676 (ZNF676) on chromosome 19p12 was also associated with telomere length under the recessive model (p = 0.001). Four SNPs (rs2853669, rs2736098, rs2736108, and rs7705526) in TERT showed significant associations with telomere length under the recessive model for the variant allele of each SNP (p = 0.01, p = 0.03, p = 0.01, and p = 0.02, respectively). The rs2853669 and rs2736098 of TERT were in strong LD (Fig. 1).
We further evaluated the combined effects of eight SNPs associated with telomere length. Among these eight SNPs, three (rs10936599, rs1317082, and rs2293607) were in complete LD, and two (rs2853669 and rs2736098) were in strong LD. Therefore, five SNPs (rs10936599, rs412658, rs2853669, rs2736108, and rs7705526) were evaluated for their combined effects. Each genotype associated with short telomere length was considered as a bad genotype for short telomeres. The telomere length decreased significantly with increasing number of bad genotypes (p = 1.7 ├Ś 10-5) (Table 3).
This study was performed to validate the associations between 17 genetic variants previously identified in Caucasians and telomere length in a Korean population. Among the 17 SNPs evaluated, eight were significantly associated with telomere length in the Korean population. Telomere length decreased significantly with increasing number of bad genotypes.
In the present study, rs10936599 and rs1317082 of MYNN and rs2293607 of TERC were significantly associated with telomere length. MYNN is located on 3q26.2 and encodes myoneurin, a member of the BTB/POZ and zinc finger domain-containing protein family, which is involved in the control of gene expression [23,24]. Myoneurin has been investigated in a mouse model, and its expression was shown to be developmentally regulated in mouse muscle [24]. In humans, myoneurin is expressed in the neuromuscular junctions and is thought to play a role in the control of either correct muscle attachment or the specificity of neuromuscular connections [24]. A number of studies have indicated that SNPs in MYNN are associated with telomere length [15,19,25,26]. Jones et al. [19] and Codd et al. [25] reported that rs10936599 is associated with telomere length. Mangino et al. [15] and Pooley et al. [26] reported associations between rs1317082 and telomere length. However, rs10936599 is a synonymous SNP, and rs1317082 is located within an intronic region of MYNN. Therefore, the associations between rs10936599 or rs1317082 and telomere length may be due to LD with another true functional variant. In the present study, rs2293607 was found to be in complete LD with rs10936599 and rs1317082. The rs2293607 is located in the promoter region of TERC, a component of telomerase. Telomerase plays an important role in the maintenance of telomere length and prevents progressive telomere shortening during cell division [1,8]. Differences in TERC mRNA expression level associated with rs2293607 have been reported [19]. rs2293607 was also found to be significantly associated with telomere length in the present study. Therefore, the association between rs10936599 or rs1317082 of MYNN and telomere length can be due to LD with rs2293607 of TERC.
Chromosome 19p12 is a recently identified locus associated with leukocyte telomere length variation [15]. This locus includes ZNF676. Zinc finger proteins are abundant in eukaryotic genomes and have various functions, including DNA recognition, RNA packaging, transcriptional activation, regulation of apoptosis, protein folding and assembly, and lipid binding [27]. Zinc finger proteins also play a role in telomere maintenance. By binding to telomeric DNA sequences and G-quadruplexes, zinc finger proteins can regulate telomere length [28]. Patel et al. [29] reported that telomerase activity was inhibited by an engineered zinc finger protein that binds to the G-quadruplex DNA structures of single-stranded human telomeric sequences. However, the effect of ZNF676 on telomere length is elusive. Mangino et al. [15] reported that the major allele of rs412658, which is located near the 3ŌĆÖ end of ZNF676, is associated with short leukocyte telomere length. In accordance with this finding, the major allele of rs412658 was associated with short telomere length in a Korean population. Consistent results across ethnic groups suggest that rs412658 may play a role in telomere length regulation. Additional studies, including functional studies, are required to establish the relationship between ZNF676 and telomere length.
TERT, one of the major components of telomerase, is located on 5p15.33. Mutations in TERT can also alter telomerase activity and telomere length [30]. The rs2736108 and rs2853669 are located in the promoter region of TERT. Bojesen et al. [18] reported that rs2736108 and rs2853669 were associated with telomere length in Caucasians. In the present study, rs2736108 and rs2853669 were also shown to be associated with telomere length in a Korean population. It was reported that the minor allele of rs2736108, together with the minor allele of rs2736109, reduced TERT promoter activity [31]. Hsu et al. [11] reported that telomerase activity was different according to the rs2853669 genotype in non-small cell lung cancer. Therefore, rs2736108 and rs2853669 may regulate telomere length by altering telomerase activity.
In the present study, associations between eight of the 17 SNPs and telomere length were verified in a Korean population. However, the remaining nine SNPs did not show significant evidence of association. Although replication is considered the gold standard for validating GWASs, non-replication of original results has also been reported in several subsequent studies [32-34]. Differences in genetic backgrounds or environmental factors among ethnic groups may be one reason for the discordant results on telomere length. It has also been reported that environmental stresses, such as oxidative stress, ethanol, caffeine, or temperature, may affect telomere length [35]. Furthermore, differences in LD patterns among ethnic groups should also be considered. The investigated variants may be linked to other polymorphisms that exhibit stronger effects on telomere length, and diverse LD patterns across ethnic groups could lead to inconsistent associations. Different minor allele frequencies should also be considered. The minor allele frequency of rs621559 is 0.06 in Caucasians but 0.28 in Koreans. The frequency of the ŌĆ£AŌĆØ allele of rs1317082 is 0.72 in Caucasians but 0.39 in Koreans. These differences in minor allele frequencies can also reduce a studyŌĆÖs power for replication [36]. Due to the relatively small sample size, the present study may not have had sufficient power for the detection of variants that exert small effects on telomere length. In particular, associations of rs398652, rs1975174, or rs2735940 with telomere length showed borderline significance in the present study. Additional studies using larger sample sizes are required to clarify these findings.
Age and gender, the best known confounding factors for telomere length, were adjusted on multivariate analyses in this study. Other minor factors, such as obesity and physical inactivity, may also be considered confounding factors. However, it is still controversial whether obesity and physical inactivity affect telomere length. In this study, no adjustments were made for obesity or physical inactivity.
In conclusion, we identified polymorphisms associated with telomere length in a Korean population. Similar results across ethnic groups suggest that polymorphisms identified in the present study may play roles in the regulation of telomere length.
Acknowledgments
This study was supported by the National R&D Program for Cancer Control, Ministry of Health and Welfare (0720550-2).
Figure┬Ā1.
Reconstructed linkage disequilibrium (LD) plots using informative single nucleotide polymorphisms in 94 healthy Koreans: TERC and MYNN (A) and TERT (B). The plots were generated using Haploview according to the method proposed by Gabriel et al. [22]. The black boxes that do not contain numbers indicate complete LD (|DŌĆÖ| = 1). The numbers in the squares are |DŌĆÖ| (├Ś 100) values. The triangles indicate haplotype blocks.
Table┬Ā1.
Characteristics of the study population
| Characteristic | No. (%) | Telomere length, mean ┬▒ SD | p value |
|---|---|---|---|
| Subject | 94 | 2.30 ┬▒ 0.60 | |
| Age, yr (range, 38ŌĆō78) | |||
| ŌĆāŌēż 62 | 46 (48.9) | 2.34 ┬▒ 0.65 | 0.55 |
| ŌĆā> 62 | 48 (51.1) | 2.26 ┬▒ 0.56 | |
| Sex | |||
| ŌĆāMale | 71 (75.5) | 2.29 ┬▒ 0.60 | 0.90 |
| ŌĆāFemale | 23 (24.5) | 2.31 ┬▒ 0.60 | |
| Smoking status | |||
| ŌĆāNever | 28 (29.8) | 2.29 ┬▒ 0.63 | 0.93 |
| ŌĆāEver | 66 (70.2) | 2.30 ┬▒ 0.59 | |
| Pack-yearsa in ever smokers (n = 66) | |||
| ŌĆāŌēż 34 | 33 (50.0) | 2.45 ┬▒ 0.67 | 0.05 |
| ŌĆā> 34 | 33 (50.0) | 2.16 ┬▒ 0.48 |
Table┬Ā2.
Association between 17 polymorphisms and telomere length
| Polymorphism | Gene or closest gene | Region | Number | Minor allele |
MAF |
ANOVA p value |
Adjusted p valueb |
||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Korean | JPTa | CEUa | Global | Dominant | Recessive | Global | Dominant | Recessive | |||||
| rs7235755 | CELF4 | Intergenic | 93 | A | 0.22 | 0.25 | 0.21 | 0.22 | 0.25 | 0.38 | 0.61 | 0.27 | 0.28 |
| rs2162440 | CELF4 | Intergenic | 93 | T | 0.22 | 0.24 | 0.21 | 0.14 | 0.16 | 0.30 | 0.31 | 0.13 | 0.33 |
| rs10936599 | MYNN | Synon | 93 | C | 0.41 | 0.32 | 0.22 | 0.15 | 0.05 | 0.53 | 0.09 | 0.04 | 0.53 |
| rs1317082 | MYNN | Intron | 85 | A | 0.39 | 0.32 | 0.72 | 0.11 | 0.04 | 0.48 | 0.07 | 0.03 | 0.49 |
| rs4387287 | OBFC1 | 5' UTR | 93 | A | 0.13 | 0.21 | 0.08 | 0.93 | 0.93 | - | 0.85 | 0.85 | - |
| rs398652 | PELI2 | Intergenic | 90 | G | 0.38 | 0.37 | 0.88 | 0.15 | 0.45 | 0.05 | 0.13 | 0.44 | 0.05 |
| rs2736428 | SLC44A4 | Intron | 89 | A | 0.42 | 0.45 | 0.36 | 0.19 | 0.46 | 0.19 | 0.88 | 0.43 | 0.20 |
| rs12696304 | TERC | 3' near gene | 84 | C | 0.27 | 0.23 | 0.76 | 0.87 | 0.68 | 0.65 | 0.60 | 0.66 | 0.65 |
| rs2293607 | TERC | 3' near gene | 94 | A | 0.40 | 0.33 | 0.75 | 0.13 | 0.04 | 0.52 | 0.08 | 0.04 | 0.52 |
| rs7705526 | TERT | Intron | 93 | A | 0.36 | - | 0.50 | 0.09 | 0.68 | 0.03 | 0.15 | 0.65 | 0.02 |
| rs2853669 | TERT | 5' near gene | 94 | C | 0.37 | - | 0.38 | 0.05 | 0.54 | 0.02 | 0.09 | 0.51 | 0.01 |
| rs2736098 | TERT | Synon | 94 | A | 0.37 | 0.25 | 0.37 | 0.08 | 0.57 | 0.03 | 0.11 | 0.52 | 0.03 |
| rs2736108 | TERT | 5' near gene | 93 | A | 0.26 | 0.17 | 0.35 | 0.04 | 0.23 | 0.01 | 0.04 | 0.23 | 0.01 |
| rs2735940 | TERT | 5' near gene | 94 | T | 0.42 | 0.44 | 0.61 | 0.19 | 0.50 | 0.07 | 0.11 | 0.44 | 0.05 |
| rs621559 | WDR65 | Intron | 84 | A | 0.28 | 0.31 | 0.06 | 0.94 | 0.83 | 0.75 | 0.66 | 0.81 | 0.54 |
| rs412658 | ZNF676 | 3' near gene | 92 | T | 0.44 | 0.33 | 0.33 | 0.01 | 0.80 | 0.01 | 0.03 | 0.63 | 0.001 |
| rs1975174 | ZNF729 | Intergenic | 93 | C | 0.27 | 0.28 | 0.52 | 0.24 | 0.14 | 0.20 | 0.07 | 0.13 | 0.13 |
MAF, minor allele frequency; ANOVA, analysis of variance; JPT, Japanese in Tokyo, Japan; CEU, Utah Residents (CEPH) with Northern and Western European Ancestry; Synon, synonymous.
a Data from NCBI (http://www.ncbi.nlm.nih.gov/SNP).
Table┬Ā3.
Combined effect of bad genotypes with telomere length
| Bad genotypea | Number | Telomere length | p value |
|---|---|---|---|
| 0 | 8 | 3.01 ┬▒ 0.80 | 1.7 ├Ś 10ŌĆō5 |
| 1 | 30 | 2.48 ┬▒ 0.43 | |
| 2ŌĆō3 | 45 | 2.14 ┬▒ 0.57 | |
| 4ŌĆō5 | 8 | 1.80 ┬▒ 0.38 |
REFERENCES
1. Blasco MA. Telomeres and human disease: ageing, cancer and beyond. Nat Rev Genet 2005;6:611ŌĆō622.


2. Levy MZ, Allsopp RC, Futcher AB, Greider CW, Harley CB. Telomere end-replication problem and cell aging. J Mol Biol 1992;225:951ŌĆō960.


3. Allsopp RC, Chang E, Kashefi-Aazam M, et al. Telomere shortening is associated with cell division in vitro and in vivo. Exp Cell Res 1995;220:194ŌĆō200.


6. Brouilette SW, Moore JS, McMahon AD, et al. Telomere length, risk of coronary heart disease, and statin treatment in the West of Scotland Primary Prevention Study: a nested case-control study. Lancet 2007;369:107ŌĆō114.


7. Willeit P, Willeit J, Mayr A, et al. Telomere length and risk of incident cancer and cancer mortality. JAMA 2010;304:69ŌĆō75.


8. Cohen SB, Graham ME, Lovrecz GO, Bache N, Robinson PJ, Reddel RR. Protein composition of catalytically active human telomerase from immortal cells. Science 2007;315:1850ŌĆō1853.


9. Kim NW, Piatyszek MA, Prowse KR, et al. Specific association of human telomerase activity with immortal cells and cancer. Science 1994;266:2011ŌĆō2015.


10. Chung IK, Hwang KY, Kim IH, et al. Helicobacter pylori and telomerase activity in intestinal metaplasia of the stomach. Korean J Intern Med 2002;17:227ŌĆō233.



11. Hsu CP, Hsu NY, Lee LW, Ko JL. Ets2 binding site single nucleotide polymorphism at the hTERT gene promoter: effect on telomerase expression and telomere length maintenance in non-small cell lung cancer. Eur J Cancer 2006;42:1466ŌĆō1474.


12. Mangino M, Richards JB, Soranzo N, et al. A genome-wide association study identifies a novel locus on chromosome 18q2 influencing white cell telomere length. J Med Genet 2009;46:451ŌĆō454.



13. Levy D, Neuhausen SL, Hunt SC, et al. Genome-wide association identifies OBFC1 as a locus involved in human leukocyte telomere biology. Proc Natl Acad Sci U S A 2010;107:9293ŌĆō9298.



14. Prescott J, Kraft P, Chasman DI, et al. Genome-wide association study of relative telomere length. PLoS One 2011;6:e19635.



15. Mangino M, Hwang SJ, Spector TD, et al. Genome-wide meta-analysis points to CTC1 and ZNF676 as genes regulating telomere homeostasis in humans. Hum Mol Genet 2012;21:5385ŌĆō5394.



16. Codd V, Mangino M, van der Harst P, et al. Common variants near TERC are associated with mean telomere length. Nat Genet 2010;42:197ŌĆō199.



17. Kote-Jarai Z, Saunders EJ, Leongamornlert DA, et al. Fine-mapping identifies multiple prostate cancer risk loci at 5p15, one of which associates with TERT expression. Hum Mol Genet 2013;22:2520ŌĆō2528.



18. Bojesen SE, Pooley KA, Johnatty SE, et al. Multiple independent variants at the TERT locus are associated with telomere length and risks of breast and ovarian cancer. Nat Genet 2013;45:371ŌĆō384.



19. Jones AM, Beggs AD, Carvajal-Carmona L, et al. TERC polymorphisms are associated both with susceptibility to colorectal cancer and with longer telomeres. Gut 2012;61:248ŌĆō254.



21. Jeon HS, Choi YY, Choi JE, et al. Telomere length of tumor tissues and survival in patients with early stage nonsmall cell lung cancer. Mol Carcinog 2014;53:272ŌĆō279.


22. Gabriel SB, Schaffner SF, Nguyen H, et al. The structure of haplotype blocks in the human genome. Science 2002;296:2225ŌĆō2229.


23. Alliel PM, Seddiqi N, Goudou D, et al. Myoneurin, a novel member of the BTB/POZ-zinc finger family highly expressed in human muscle. Biochem Biophys Res Commun 2000;273:385ŌĆō391.


24. Cifuentes-Diaz C, Bitoun M, Goudou D, et al. Neuromuscular expression of the BTB/POZ and zinc finger protein myoneurin. Muscle Nerve 2004;29:59ŌĆō65.


25. Codd V, Nelson CP, Albrecht E, et al. Identification of seven loci affecting mean telomere length and their association with disease. Nat Genet 2013;45:422ŌĆō427.



26. Pooley KA, Bojesen SE, Weischer M, et al. A genome-wide association scan (GWAS) for mean telomere length within the COGS project: identified loci show little association with hormone-related cancer risk. Hum Mol Genet 2013;22:5056ŌĆō5064.



27. Laity JH, Lee BM, Wright PE. Zinc finger proteins: new insights into structural and functional diversity. Curr Opin Struct Biol 2001;11:39ŌĆō46.


28. Isalan M, Patel SD, Balasubramanian S, Choo Y. Selection of zinc fingers that bind single-stranded telomeric DNA in the G-quadruplex conformation. Biochemistry 2001;40:830ŌĆō836.


29. Patel SD, Isalan M, Gavory G, Ladame S, Choo Y, Balasubramanian S. Inhibition of human telomerase activity by an engineered zinc finger protein that binds G-quadruplexes. Biochemistry 2004;43:13452ŌĆō13458.



30. Alter BP, Rosenberg PS, Giri N, Baerlocher GM, Lansdorp PM, Savage SA. Telomere length is associated with disease severity and declines with age in dyskeratosis congenita. Haematologica 2012;97:353ŌĆō359.



31. Beesley J, Pickett HA, Johnatty SE, et al. Functional polymorphisms in the TERT promoter are associated with risk of serous epithelial ovarian and breast cancers. PLoS One 2011;6:e24987.



32. Ranade K, Hsuing AC, Wu KD, et al. Lack of evidence for an association between alpha-adducin and blood pressure regulation in Asian populations. Am J Hypertens 2000;13(6 Pt 1):704ŌĆō709.


33. Kao WT, Wang Y, Kleinman JE, et al. Common genetic variation in neuregulin 3 (NRG3) influences risk for schizophrenia and impacts NRG3 expression in human brain. Proc Natl Acad Sci U S A 2010;107:15619ŌĆō15624.



34. Chakkera HA, Hanson RL, Kobes S, et al. Association of variants in the carnosine peptidase 1 gene (CNDP1) with diabetic nephropathy in American Indians. Mol Genet Metab 2011;103:185ŌĆō190.






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print



