To the Editor,
Churg-Strauss syndrome (CSS) is a systemic disorder characterized by bronchial asthma, hypereosinophilia, and systemic vasculitis. The American College of Rheumatology (ACR) classification criteria in patients with biopsy-proven vasculitis is the most widely used today. These criteria include asthma, greater than 10% peripheral eosinophilia, mono- or polyneuropathy, migratory pulmonary infiltrates, sinus anomalies, and extravascular eosinophils in the biopsy. The presence of four of these criteria in a patient with biopsy-proven vasculitis has a sensitivity of 85% and a specificity of 99.7% for CSS [1]. Involvement of the lung, skin, gastrointestinal tract, and nervous system are common in CSS, but involvement of the mediastinal lymph node (LN) and gallbladder (GB) containing stones are less common. We, herein, report and discuss a special CSS case involving the mediastinal LN and GB containing stones, as well as lung parenchyme.
A 43-year-old woman was admitted to our hospital because of dyspnea (Modified Medical Research Council IV) and wheezing. The associated symptoms were cough, sputum, and dyspnea for 7 days. On physical examination, auscultation of both lungs revealed inspiratory crackle and expiratory wheezing sounds localized to both lower lungs. The patientŌĆÖs body temperature was 39.0┬░C, and blood pressure and pulse rate were 140/85 mmHg and 120 beats per minute, respectively. The past medical records revealed a diagnosis of asthma 2 years earlier with regular use of an inhaled corticosteroid and a leukotriene antagonist.
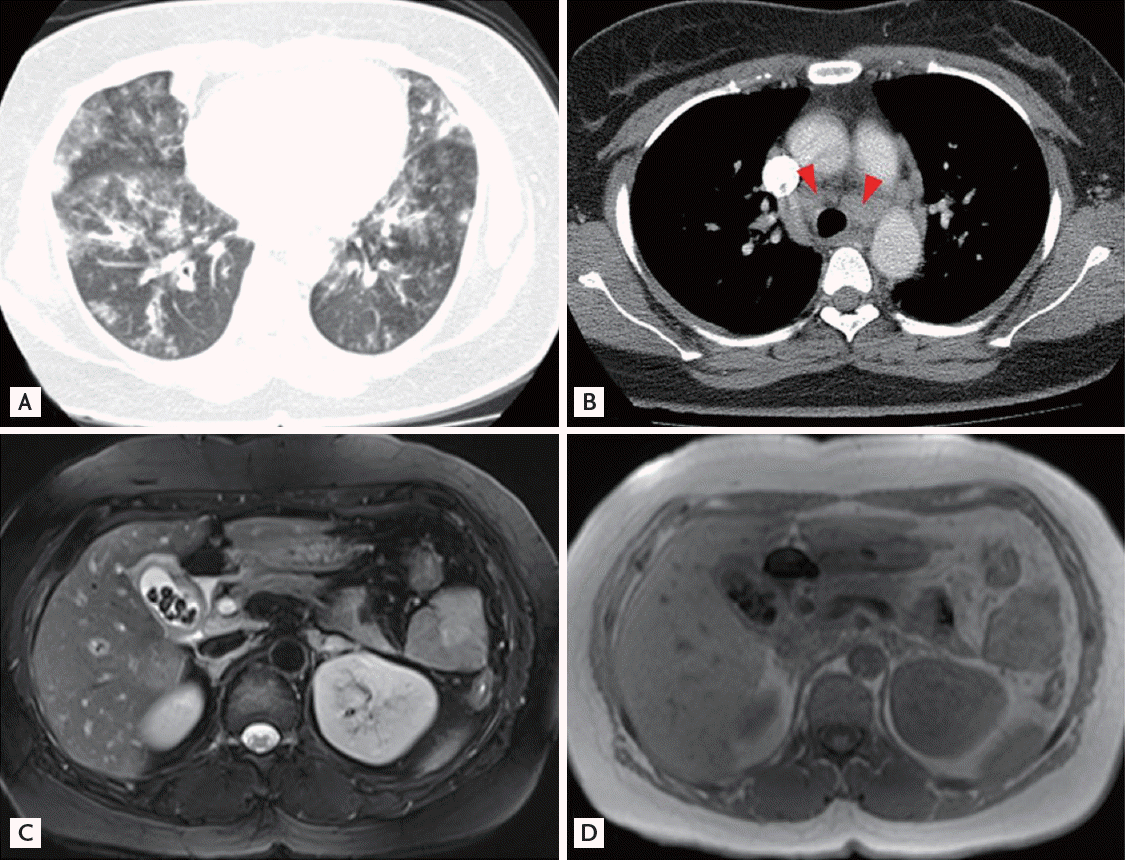
The laboratory results showed elevated plasma C-reactive protein 7.210 mg/dL (reference range, 0.03 to 0.3). The white blood cell count was elevated to 25,600/mm3 (reference range, 3,640 to 9,750) while the differential count was neutrophils 44% and eosinophils 45%. Hemoglobin was 12.5 g/dL (reference range, 11.2 to 14.7) and the platelet count was 305,000/mm3 (reference range, 150,000 to 400,000). Aspartate aminotransferase was 38 IU/L (reference range, 0 to 50) and alanine aminotransferase was 59 IU/L (reference range, 0 to 45) while alkaline aminotransferase was 215 IU/L (reference range, 35 to 160) and ╬│-glutamyl transpeptidase was 182 IU/L (reference range, 4 to 54). The serum blood urea nitrogen and creatinine were normal. The chest radiograph showed multifocal patchy consolidation in both lower lungs suggesting pneumonia. The chest computed tomography (CT) showed multiple lymphadenopathy at the right supraclavicular region, both hilar areas, and the mediastinum with multifocal pneumonic consolidations on both lower lungs (Fig. 1A and 1B). The initial arterial blood gas analysis was pH of 7.474, partial pressure of CO2 (PaCO2) of 30.3 mmHg, partial pressure of O2 (PaO2) of 56.3 mmHg, HCO3 of 21.8 mmol/L, and O2 saturation of 91.0%.
The patient was admitted to the intensive care unit for hypoxia. The patient was treated with methylprednisolone 60 mg/day and antibiotics with cefoperazone-sulbactam and ciprofloxacin assuming asthma exacerbation with pneumonia. Following 7 days of treatment, dyspnea improved, she maintained an afebrile state, and pulmonary infiltration had nearly disappeared on serial chest radiographs; thus, methylprednisolone was slowly tapered off to 30 mg/day over 7 days. However, peripheral eosinophilia did not decrease (Fig. 2).
Therefore, we considered eosinophilic infiltrative diseases accompanied by lymphadenopathy, hidden malignancy, parasite infection, or drug reaction as differential diagnoses. The following laboratory findings revealed the presence of anti-neutrophil cytoplasmic antibody and subsequent serum P-anti-neutrophil cytoplasmic antibody, and serum immunoglobulin E was elevated (> 1,000 IU/mL), even though medical records from 1 year prior showed a normal eosinophil count. The tumor markers such as ╬▒-fetoprotein, carcinoembryonic antigen, cancer antigens 125, and 19-9 and angiotensin-converting enzyme were all normal. Parasite laboratory findings were all negative. Urinalysis revealed microscopic hematuria and microalbuminuria (399.4 mg/day). Paranasal sinus radiographs showed left paranasal sinusitis. Based on asthma, eosinophilia, pulmonary infiltrates, and paranasal sinusitis, CSS was strongly suspected according to the ACR criteria, but not yet confirmed by biopsy.
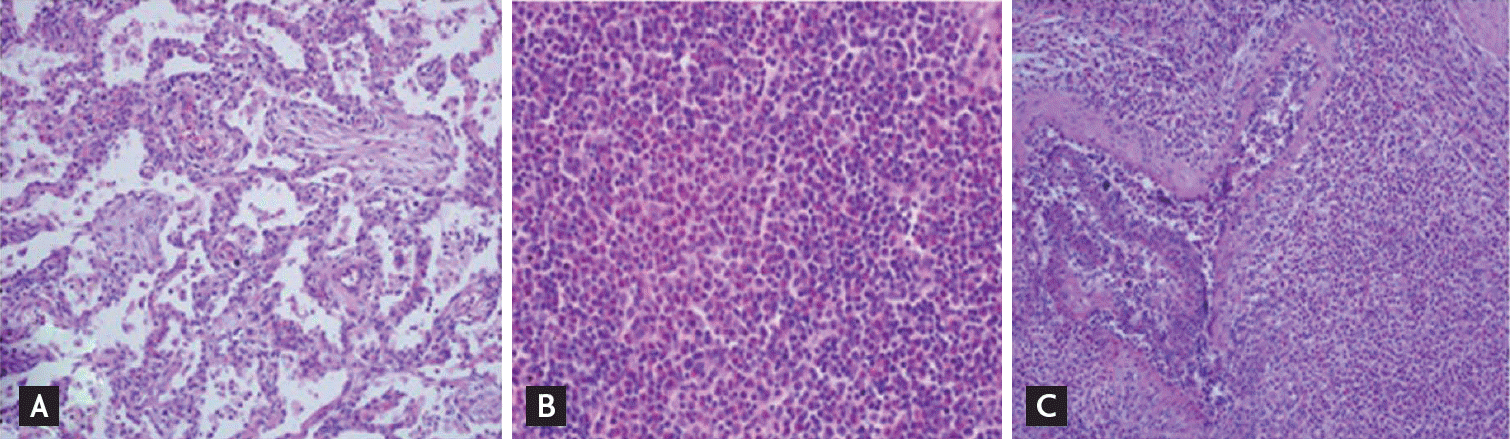
Subsequent follow-up chest CT scan compared with the previous chest CT scan taken 8 days prior showed that the multifocal patchy consolidations had largely disappeared, but there was no significant change in lymphadenopathy. Eight days after admission, she developed a fever and right upper quadrant abdominal pain with a positive MurphyŌĆÖs sign. Laboratory test results showed an elevated leukocyte count (27,900/mm3) with marked eosinophilia (45.6%). Liver enzymes were abnormal while bilirubin levels were normal. Aspartate aminotransferase was 67 IU/L, and alanine aminotransferase was 115 IU/L, while alkaline aminotransferase was 293 IU/L and ╬│-glutamyl transpeptidase was 470 IU/L. Abdominal sonography and magnetic resonance cholangiopancreatography revealed acute cholecystitis with multiple gall bladder stones and fatty liver (Fig. 1C and 1D). Because her abdominal pain was not relieved and the common bile duct was patent without stones on endoscopic retrograde cholangiopancreatography, cholecystectomy for the treatment of cholecystitis and concurrent wedge resection with mediastinal LN biopsy was performed for tissue confirmation. Histopathological examination of the resected GB, lung and LN showed necrotizing and granulomatous vasculitis with eosinophil infiltrates indicating CSS involvement, respectively (Fig. 3). A few days after the operation, she developed purpuric skin lesions on both dorsa and complained of a tingling sensation on both hands. Skin biopsy performed from purpuric lesions showed leukocytoclastic vasculitis with eosinophil predominance.
Finally, she was confirmed as having CSS based on asthma, eosinophilia, polyneuropathy, pulmonary infiltrates, paranasal anomalies, and extravascular eosinophils confirmed by biopsy of multiple sites, which together met all of the ACR criteria. The involvement of no other organ such as the gastrointestinal tract or heart was observed, as determined by gastrofiberscopic biopsy and two-dimensional echocardiography.
After 4 weeks of treatment with methylprednisolone (60 mg/day), her associated symptoms improved, and liver enzymes were decreased. Blood tests performed at subsequent outpatient department clinic visits indicated that the white blood cell count was 18,700/mm3 and eosinophils decreased to 22.3% (Fig. 2).
CSS is a rare disease that occurs in individuals with asthma, with a reported population incidence of 1/1,000,000, and is characterized histologically by pulmonary and systemic small vessel necrotizing vasculitis, peripheral eosinophilia, and granulomas. Perinuclear anti-neutrophil cytoplasmic antibodies are present in less than half of the cases [2]. The various anatomic sites involved in CSS include lungs, gastrointestinal tract, heart, peripheral nerves, and central nervous system (CNS). The involvement of mediastinal LN and GB containing stones in CSS have not been reported in Korea. Because CSS is a systemic disease as the case herein reported, suspicion of other organ involvement by CSS is prior to hasty conclusion that the manifestation is another disease entity for differential diagnosis.
And in a case of atypical allergic diathesis, simultaneous invasive, and noninvasive evaluations are needed for the confirmation of CSS [3], even though diagnosis for this case was delayed due to atypical manifestations. Furthermore, this patient was in serious condition with unstable vital signs in which prompt initial management took priority over invasive evaluation.
On the other hand, this patient was taking leukotriene antagonist (montelukast, Lukair, CJ, Seoul, Korea) since she had been diagnosed as asthma. Franco and Artes [4] reported the temporal relationship between leukotriene antagonist treatment and the development of pulmonary eosinophilia as a causal association, although cysteinyl leukotriene type 1 receptor antagonists remain safe and effective drugs for most patients with mild to moderate asthma. However, while the exact causes of the syndrome are not well understood, discontinuation of this drug is warranted for those with more severe disease who require systemic corticosteroids, especially if they show characteristics of the atypical allergic diathesis seen in the prodromal phase of CSS.
CSS can involve other organs simultaneously or later at time of diagnosis, so initial systemic check-up and appropriate follow-up care is necessary. In this case, CSS initially manifested as mediastinal lymphadenopathy with lung consolidation, but subsequently evolved into an acute calculous cholecystitis, and then, finally, as purpuric skin lesions and polyneuropathy.
High dose steroids are the mainstay of treatment and generally begin with a dose of 1 mg/kg/day (max 60 mg/day) of prednisolone, maintained until symptoms are controlled, and then slowly tapered for 1 year. In patients with factors which have a poor prognosis (e.g., serum creatinine > 140 mmol/L, proteinuria > 1 g/day, severe gastrointestinal tract involvement, cardiomyopathy, and/or CNS involvement) [5], the combination of steroids and cyclophosphamide produced better control and sustained remission rates, compared with steroids alone [1]. This case reported herein was without poor prognosis factors and hence initially had good response to steroid treatment alone, but then rapidly showed newly developed cholecystitis while steroids were tapered off. Thus, the mainstay of steroid treatment and the slow tapering off are important for the treatment of CSS.
In conclusion, we herein report an unusual case of CSS manifested by mediastinal lymphadenopathy with lung consolidation and necrotizing eosinophilic cholecystitis, which can be misdiagnosed as pneumonia with reactive LN enlargement and stone-induced cholecystitis. Thus, involvement of CSS in not only lung, skin, and the nervous system but also other organs, especially LN and GB, should not be overlooked. Furthermore, considering the probability of CSS presenting as a systemic disease and the likelihood of undefined accompanying condition, more effort is need to search for other involved organs in CSS.




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





