 |
 |
| Korean J Intern Med > Volume 33(1); 2018 > Article |
|
Abstract
Background/Aims
Methods
Results
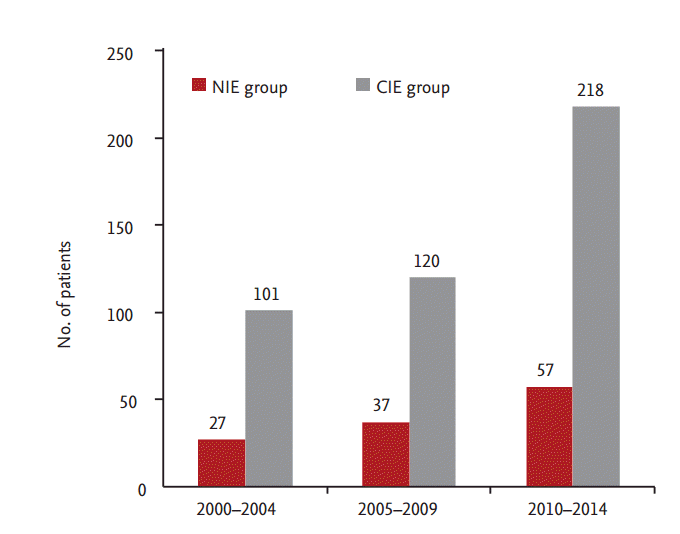
Figure┬Ā1.
Table┬Ā1.
Values are presented as mean ┬▒ SD or number (%).
NIE, nosocomial infective endocarditis; CIE, community-acquired infective endocarditis; COPD, chronic obstructive pulmonary disease; ILD, interstitial lung disease; TACE, transcatheter arterial chemoembolization; PPM, permanent pacemaker; ICD, implantable cardioverter defibrillator; HACEK, Haemophilus parainfluenzae, Haemophilus aphrophilus, Aggregatibacter actinomycetemcomitans, Cardiobacterium hominis, Eikenella corrodens, and Kingella kingae.
Table┬Ā2.
| Variable |
Crude |
Adjusted |
||
|---|---|---|---|---|
| OR (95% CI) | p value | OR (95% CI)a | p value | |
| Age | 1.03 (1.02ŌĆō1.04) | < 0.001 | 1.02 (1.001ŌĆō1.04) | 0.006 |
| Male sex | 1.58 (1.06ŌĆō2.38) | 0.027 | 1.43 (0.86ŌĆō2.38) | 0.170 |
| Liver cirrhosis | 3.65 (1.67ŌĆō8.00) | 0.001 | 4.57 (1.86ŌĆō11.21) | 0.001 |
| Diabetes mellitus | 1.94 (1.03ŌĆō3.67) | 0.042 | 1.47 (0.67ŌĆō3.25) | 0.340 |
| Chemotherapy | 10.76 (5.70ŌĆō20.31) | < 0.001 | 5.03 (2.29ŌĆō11.06) | < 0.001 |
| Central intravenous catheter | 23.59 (11.0 ŌĆō50.50) | < 0.001 | 10.18 (4.21ŌĆō24.62) | < 0.001 |
| Hemodialysis | 5.27 (2.59ŌĆō10.72) | < 0.001 | 2.58 (1.02ŌĆō6.53) | 0.046 |
| Endoscopy with procedure | 14.97 (1.66ŌĆō135.25) | 0.016 | 10.53 (0.98ŌĆō13.54) | 0.052 |
| Genitourinary tract manipulation | 7.47 (1.35ŌĆō41.59) | 0.021 | 11.22 (1.84ŌĆō68.36) | 0.009 |
Table┬Ā3.
Values are presented as mean ┬▒ SD or number (%).
NIE, nosocomial infective endocarditis; CIE, community-acquired infective endocarditis; COPD, chronic obstructive pulmonary disease; ILD, interstitial lung disease; NA, not assessed; TACE, transcatheter arterial chemoembolization; PPM, permanent pacemaker; ICD, implantable cardioverter defibrillator; HACEK, Haemophilus parainfluenzae, Haemophilus aphrophilus, Aggregatibacter actinomycetemcomitans, Cardiobacterium hominis, Eikenella corrodens, and Kingella kingae.
Table┬Ā4.
| Variable |
Crude |
Adjusted |
||
|---|---|---|---|---|
| OR (95% CI) | p value | OR (95% CI)a | p value | |
| Age | 1.03 (1.01ŌĆō1.06) | 0.045 | 1.04 (1.01ŌĆō1.07) | 0.037 |
| Male sex | 0.40 (0.17ŌĆō0.90) | 0.028 | 0.42 (0.17ŌĆō1.08) | 0.070 |
| Liver cirrhosis | 1.21 (0.35ŌĆō4.24) | 0.770 | 2.80 (0.64ŌĆō12.34) | 0.170 |
| Chemotherapy | 2.83 (1.21ŌĆō6.61) | 0.016 | 3.89 (1.18ŌĆō12.87) | 0.026 |
| Hemodialysis | 0.27 (0.06ŌĆō1.24) | 0.090 | 0.36 (0.07ŌĆō1.91) | 0.230 |
| Central intravenous catheter | 1.22 (0.53ŌĆō2.83) | 0.640 | 0.82 (0.26ŌĆō2.63) | 0.740 |
| On indication of cardiac surgery (but not done) | 2.93 (1.21ŌĆō7.08) | 0.017 | 2.18 (0.81ŌĆō5.88) | 0.120 |
| Fungus | 5.93 (1.03ŌĆō34.09) | 0.046 | 4.43 (0.61ŌĆō31.94) | 0.140 |
Table┬Ā5.
| Variable |
Crude |
Adjusted |
||
|---|---|---|---|---|
| Crude OR (95% CI) | p value | Adjusted OR (95% CI)a | p value | |
| Age | 1.06 (1.03ŌĆō1.09) | < 0.001 | 1.04 (1.01ŌĆō1.07) | 0.012 |
| Male sex | 1.07 (0.46ŌĆō2.45) | 0.880 | 1.09 (0.38ŌĆō3.16) | 0.870 |
| Liver cirrhosis | 2.79 (0.59ŌĆō13.15) | 0.200 | 1.13 (0.17ŌĆō7.30) | 0.900 |
| On indication of cardiac surgery (but not done) | 30.62 (12.36ŌĆō75.84) | < 0.001 | 29.83 (10.19ŌĆō87.31) | < 0.001 |
| Viridans group streptococci | 0.09 (0.01ŌĆō0.67) | 0.019 | 0.14 (0.01ŌĆō1.16) | 0.070 |
| MRSA | 5.55 (1.87ŌĆō16.49) | 0.002 | 9.58 (2.27ŌĆō40.45) | 0.002 |
REFERENCES
-
METRICS

- Related articles
-
Predictive factors of radiographic progression in ankylosing spondylitis2015 May;30(3)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print


