 |
 |
| Korean J Intern Med > Volume 33(3); 2018 > Article |
|
Abstract
The renin-angiotensin system (RAS) is an important regulator of cirrhosis and portal hypertension. As hepatic fibrosis progresses, levels of the RAS components angiotensin (Ang) II, Ang-(1–7), angiotensin-converting enzyme (ACE), and Ang II type 1 receptor (AT1R) are increased. The primary effector Ang II regulates vasoconstriction, sodium homoeostasis, fibrosis, cell proliferation, and inflammation in various diseases, including liver cirrhosis, through the ACE/Ang II/AT1R axis in the classical RAS. The ACE2/Ang-(1–7)/Mas receptor and ACE2/Ang-(1–9)/AT2R axes make up the alternative RAS and promote vasodilation, antigrowth, proapoptotic, and anti-inflammatory effects; thus, countering the effects of the classical RAS axis to reduce hepatic fibrogenesis and portal hypertension. Patients with portal hypertension have been treated with RAS antagonists such as ACE inhibitors, Ang receptor blockers, and aldosterone antagonists, with very promising hemodynamic results. In this review, we examine the RAS, its roles in hepatic fibrosis and portal hypertension, and current therapeutic approaches based on the use of RAS antagonists in patients with portal hypertension.
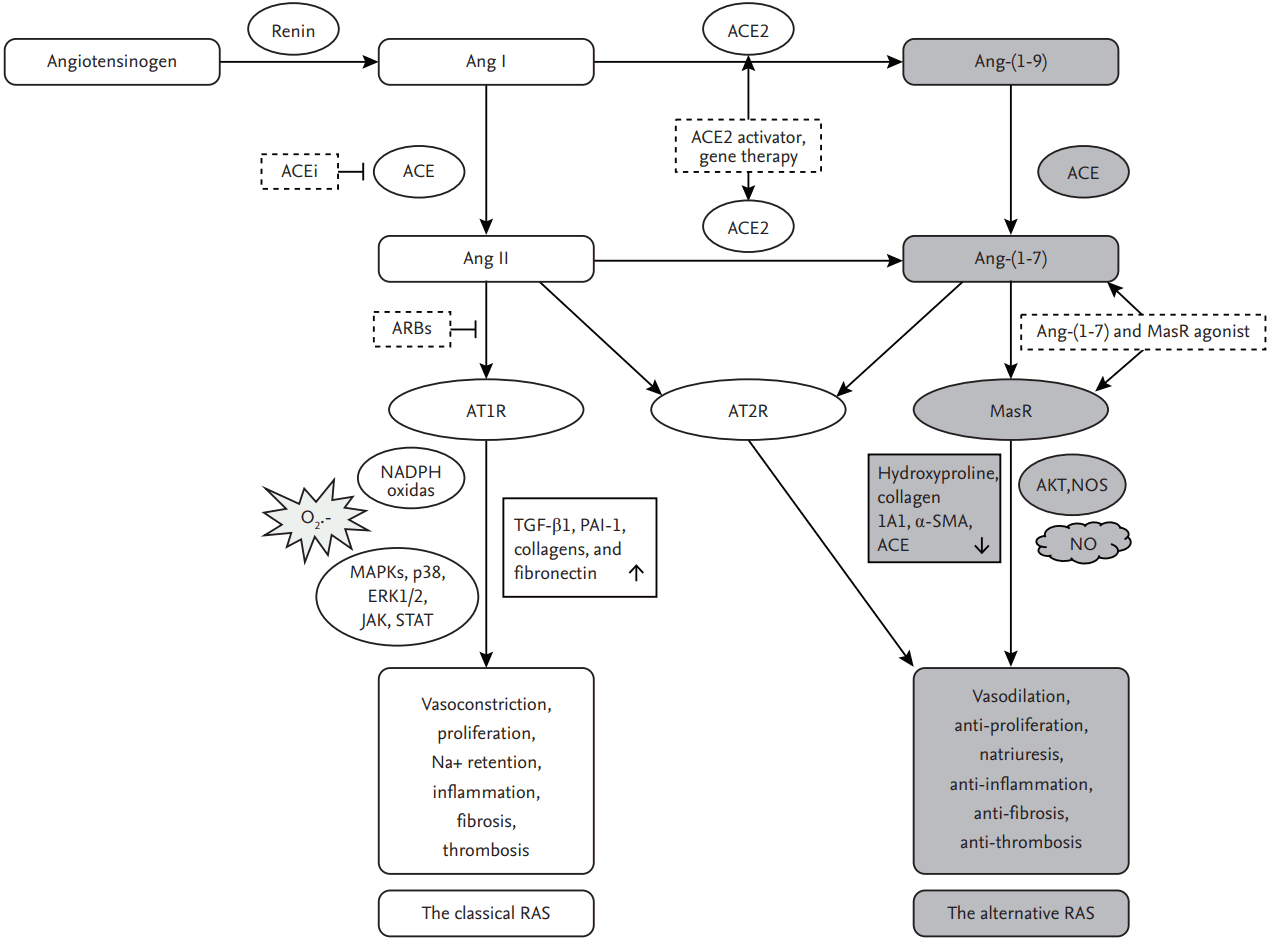
The renin-angiotensin system (RAS) is a physiological regulator of blood pressure, electrolyte balance, and fluid homeostasis. However, it is also involved in organ dysfunction and chronic tissue damage, via the vasoactive and profibrotic effects of angiotensin (Ang) II, a major effector octapeptide [1], and in the pathogenesis of hepatic fibrosis and portal hypertension [2-4]. This review provides an overview of the RAS, its role in hepatic fibrosis and portal hypertension, and current therapeutic approaches based on the use of RAS antagonists to treat patients with portal hypertension (Fig. 1).
The RAS precursor angiotensinogen and its cleavage enzyme, renin, have been extensively detected in normal and in injured liver tissue, but their expression does not increase immediately after liver injury [5]. Instead, the levels of angiotensin-converting enzyme (ACE) and Ang II type 1 receptor (AT1R) are remarkably increased after liver injury, particularly in fibrotic areas of the injured liver tissue and activated hepatic stellate cells (HSCs) [5-7]. ACE2 is expressed in healthy liver by endothelial cells, bile duct cells, and perinuclear hepatocytes, but its high level of expression in the parenchymal tissue of diseased livers results in increased expression of Ang-(1–7), produced from Ang II [8,9]. One cause of portal hypertension is an increase in intrahepatic resistance to portal flow, mediated by contraction of the sinusoidal vascular bed by HSCs and vascular smooth muscle cells [10-12]. HSC activation is facilitated by Ang II. These results link the RAS to liver fibrosis and portal hypertension [2-5,13,14], but they also indicate that the RAS can be targeted to ameliorate hepatic fibrosis. This has been demonstrated in experimental animal models and suggests that a reduction in portal pressure in humans can be achieved by suppressing Ang II-mediated intrahepatic vasoconstriction [15-18].
As the main effector of the RAS, the potent vasoconstrictor Ang I regulates arterial blood pressure and sodium homeostasis, but it also acts on cardiovascular processes such as remodeling [19]. The aspartyl protease renin, expressed by the juxtaglomerular apparatus of the kidney, converts liver-derived angiotensinogen into Ang I, which in turn is converted to Ang II by ACE. ACE2 catalyzes the conversion of Ang II to Ang-(1–7). Thus, Ang II levels are regulated by the balance between ACE and ACE2.
The various components of the RAS form two distinct pathways: the classical pathway, comprising Ang II, ACE, and AT1R, and the alternative pathway, comprising ACE2, Ang-(1–7), and Ang-(1–7) Mas receptor (MasR).
Vasoconstriction, sodium homoeostasis, fibrosis, cell proliferation, and inflammation are mediated by the classical RAS pathway [20-24]. The vasoconstrictor effect of Ang II is dependent on AT1R, a G-protein-coupled receptor present in most cells types, including the liver [25,26]. AT1R stimulates the activation of phospholipases A2, C, and D as well as L-type Ca2+ channels while inhibiting adenylate cyclase (reviewed in [27,28]). Triggered by Ang II, AT1R also induces cell proliferation by activating tyrosine phosphorylation, phospholipase C-γ, and downstream proteins, including mitogen-activated protein kinases (MAPKs), Janus kinases, and signal transducer and activator of transcription [23,29,30].
In addition to the classical RAS pathway, the (pro)-renin receptor, the ligands of which are renin and pro-renin, activates the extracellular signal-regulated kinase (ERK) 1/2 and p38 pathways, leading to up-regulation of profibrotic genes, including those encoding transforming growth factor β1 (TGF-β1), plasminogen activator inhibitor-1, collagens, and fibronectin [31,32].
The alternative RAS pathway, via its components ACE2, Ang-(1–7), and MasR, counterbalances the effects of the classical RAS pathway. Despite the > 40% homology between the catalytic domain of ACE and that of ACE2 [33,34], the latter removes a single amino acid at the C-terminus of Ang I to generate the nonapeptide Ang-(1–9), whereas ACE digests a dipeptide to convert Ang-(1–9) to Ang-(1–7) [34,35]. ACE2 also cleaves Ang II to produce Ang-(1–7), with greater efficiency than the conversion of Ang I to Ang-(1–9) [36]. In fact, the substrate preference of ACE2 for Ang II is approximately 400-fold higher than that for Ang I [36]. ACE2 is thus representative of a compensatory pathway in the RAS [34,37]. Ang-(1–7) and Ang-(1–9), produced by ACE2, react with MasR and AT2R, respectively [38]. MasR is a G-protein-coupled receptor encoded by the Mas proto-oncogene [38]. The ACE2/Ang-(1–7)/MasR axis counterbalances the effects of the ACE/Ang II/AT1R axis, increases vasodilation and the production of nitric oxide (NO), inhibits cell proliferation and cardiovascular remodeling, and improves endothelial function [39,40]. The ACE/Ang II/AT1R axis is also countered by the ACE2/Ang-(1–9)/AT2R axis, which stimulates vasodilation and exerts antigrowth, proapoptotic, and anti-inflammatory effects [41,42].
The hallmarks of cirrhosis are fibrotic septa, regenerating hepatocyte nodules, hepatic sinusoidal remodeling, and capillarization [43,44]. These architectural changes are related to increased intrahepatic resistance to portal blood flow, resulting in increased portal pressure and the development of portal hypertension, the most serious complication of cirrhosis [45]. Portal blood flow is also increased by splanchnic vasodilatation and hyperdynamic circulation [46]. Paizis et al. [5] demonstrated that ACE and AT1R levels are dramatically upregulated in the active fiber-forming region of the liver after hepatic injury; thus, linking the classical RAS to portal hypertension. In addition, ACE and AT1R are highly expressed by activated HSCs both in vivo and in vitro [6]. Upregulation of ACE2 at the gene and protein levels following liver injury in rats and humans implicates the alternative RAS in the response to cirrhosis and portal hypertension [8]. Herath et al. [9] reported the association of alternative RAS activation in chronic liver injury, based on the increase in plasma Ang-(1–7) induced by the upregulation of ACE2 and Mas as well as the hepatic conversion of Ang II to Ang-(1–7). These results clearly show that the classical RAS pathway promotes, while the alternative pathway antagonizes the progression of cirrhosis and portal hypertension.
Sustained and chronic liver disease, caused by hepatitis viruses, heavy alcohol use, certain medications, toxins, and autoimmune diseases, is characterized by the accumulation of excess extracellular matrix (ECM) proteins and changes in liver architecture, followed by the formation of fibrous scars and cirrhotic nodules [2]. Portal fibroblasts, circulating fibroblasts, and bone marrow-derived cells are involved in hepatic fibrogenesis [47], but the most pivotal cell type is HSCs, which secrete collagen types I and III [2]. One of the many mechanisms underlying activation of quiescent HSCs after liver damage is upregulation of RAS components during liver disease, including AT1R/AT2R and MasR, which promote and suppress fibrosis, respectively [5,9,48]. In human liver, quiescent HSCs do not express RAS components, nor do they release Ang II. However, both in vivo-activated HSCs isolated from human cirrhotic liver and culture-activated HSCs isolated from normal human liver highly express active renin and ACE and secrete Ang II [6]. Acting via AT1R, Ang II stimulates DNA synthesis and increases the contraction and proliferation of activated HSCs [49]. Ang II also mediates the proliferation and contraction of HSCs as well as their production of ECM via different signaling pathways, including MAPK pathways, phosphoinositide/Ca2+ pathway, and the generation of reactive oxygen species by phosphorylating the p47phox subunit of Nox [49-51]. HSCs are activated by reactive oxygen species, whereas fibrosis after liver injury is ameliorated in p47phox knockout mice [50]. In addition, in both activated and quiescent rat HSCs exposed to Ang II, the mRNA and protein levels of all TGF-β isoforms are upregulated via the ERK1/2- and Nox-dependent pathways, but independently of protein kinase C [52].
As described above, the alternative RAS axis produces antifibrotic effects via the components ACE2, Ang-(1–7), and MasR. In a rat model of hepatic fibrosis induced by bile duct ligation, the Ang-(1–7) and MasR agonist AVE 0991 improved fibrosis, reduced the content of hydroxyproline, a major component of collagen, and decreased the expression of collagen 1A1, α-smooth muscle actin, and ACE [53]. These antifibrotic effects were antagonized by pharmacological blockade of the MasR, which induced significant increases in hydroxyproline and total TGF-β1 levels [53-55]. In a mouse model of cirrhosis, ACE2, which is upregulated after liver injury [56], inhibited hepatic fibrosis via destruction of Ang II and production of Ang-(1–7). While the loss of ACE2 activity exacerbates experimental hepatic fibrosis, recombinant ACE2 attenuates hepatic fibrosis in chronic liver injury models, suggesting its therapeutic potential [57].
Taken together, these results demonstrate the important roles played by the classical and alternative RAS pathways in promoting and inhibiting fibrosis, as well as the therapeutic potential of classical RAS pathway antagonists and alternative RAS pathway agonists in patients with hepatic fibrosis.
Portal hypertension is a major cause of morbidity and mortality in patients with cirrhosis. Multiple factors contribute to its pathogenesis, including increased intrahepatic resistance following increased deposition of ECM, distortion of the hepatic vascular architecture [2], and splanchnic vasodilation in response to NO produced by endothelial NO synthase [58-60]. The increased vascular tone and ensuing hepatic resistance to portal inflow have also been attributed to contraction of the sinusoidal vascular bed by activated HSCs and vascular smooth muscle cells [11]. Because the activation of HSCs during liver injury is induced by Ang II, and activated HSCs express Ang II, ACE, and AT1R [5,49], the RAS is a key mediator of the pathogenesis of portal hypertension in cirrhosis [3,61,62].
In addition to increased intrahepatic resistance, the systemic and splanchnic vasodilation that characterizes cirrhosis reflects a hypo-responsiveness to vasoconstrictors such as Ang II, α-adrenergic agonists, and endothelin-1 [63,64]. In contrast to the vasoconstrictor activity of Ang II, Ang-(1–7) is a vasodilator [65] whose systemic levels are dependent on ACE2 activity during the progression of hepatic fibrosis [8,9]. Therefore, increased ACE2 expression may accelerate the transition from vasoconstriction to vasodilation in cirrhosis.
Following the report of Lebrec et al. [66] showing that propranolol, a non-selective β-blocker (NSBB), reduces portal venous pressure in patients with cirrhosis, NSBBs have become a standard treatment in patients with portal hypertension. NSBBs reduce cardiac output and splanchnic blood flow by blocking β-1 and -2 adrenergic receptors, resulting in splanchnic vasoconstriction and decreased portal pressure [67]. These drugs were also shown to reduce the incidence of bleeding (primary prophylaxis) and rebleeding (secondary prophylaxis) in patients with esophageal varices [68,69]. However, NSBBs do not produce optimal responses in all patients; 15% of patients do not tolerate NSBBs, and nearly 50% do not show a therapeutic reduction in the hepatic venous pressure gradient (HVPG < 12 mmHg or a decrease of > 20% from baseline) [70,71]. These resistant patients are instead treated with nitrates, which increase NO levels in the intrahepatic circulation, or with prazosin or clonidine, which inhibit α-adrenergic activity; modest reductions in HVPG were achieved in both groups [72-74].
Recently, very promising hemodynamic results have been achieved using other therapeutic agents that inhibit the RAS (e.g., captopril, losartan, and irbesartan) [75-77]. Decreases in portal pressure in patients with cirrhosis have been obtained with RAS antagonists such as ACE inhibitors (ACEis) and AT1R blockers (ARBs), and aldosterone antagonists (AAs), without adverse events [61,78]. Tandon et al. [61] conducted a systematic review and meta-analysis of 19 controlled trials (678 patients) to evaluate the efficacy and safety of RAS antagonists in reducing portal pressure. The ARBs and ACEis used in the clinical trials included losartan, candesartan, and irbesartan (ARBs) and captopril and enalapril (ACEis) [75-77,79-82]. Captopril effectively reduces portal pressure in patients with portal hypertension characterized by a low portal venous velocity [77]. Thus, captopril and other ACEis may be useful for treating this subset of patients with increased intrahepatic resistance. Encouraging results were also obtained with the AA spironolactone [83,84] and with ARB/ ACEi therapy, in which significant reductions in HVPG compared with a placebo were reported [75,76,79,85]. Although greater reductions in HVPG were achieved with NSBBs than with ARBs/ACEis, the difference according to the pooled individual patient data was not significant [77,80]. The HVPG of Child-Pugh class A patients treated with ARBs/ACEis or NSBBs decreased by 17% and 21%, respectively, whereas there was no significant change in the HVPG of Child-Pugh class B/C patients treated with ARBs/ACEis (3%) [61]. However, the difference in the HVPG between the AA-treated and placebo group was significant. While there were no adverse events in any of these groups according to Tandon et al. [61], individual data obtained from a systematic review and meta-analysis of the efficacy and safety of RAS antagonists in reducing portal pressure suggested a higher rate of adverse events in patients with more advanced liver dysfunction.
The antifibrotic effects of RAS antagonists, including ACEis, ARBs, and AAs, have been evaluated in several clinical studies [82,86-94]. The benefits of candesartan, an ARB, in patients with compensated alcoholic liver fibrosis were first reported as part of a well-established open-label randomized controlled trial (RCT) [82]. Candesartan resulted in significant histological improvements and reduced fibrosis scores, fibrotic areas, and α-smooth muscle actin and hydroxyproline levels [82]. Zhu et al. [94] conducted a systematic review and meta-analysis of RCTs to assess the efficacy and safety of ACEis/ARBs in liver fibrosis. They showed that RAS inhibitor therapy significantly lowered both liver fibrosis scores and the liver fibrotic area in patients with hepatic fibrosis; the good safety profile of these drugs was also demonstrated. Taking these results together, the use of RAS antagonists, such as ACEis, ARBs, and AAs, may decrease portal pressure and ameliorate fibrosis in patients with cirrhosis. However, high-quality RCTs using more accurate evaluation parameters are needed to confirm the effectiveness and safety of RAS antagonists for the treatment of hepatic fibrosis and portal hypertension.
Portal hypertension is a serious complication of cirrhosis that increases the morbidity and mortality rates of these patients. Several studies have demonstrated the role of RAS in the pathogenesis of hepatic fibrosis and portal hypertension [2-4], based on elevated levels of several RAS components (Ang II, Ang-[1–7], ACE, and AT1R) during the progression of hepatic fibrosis [5,6,8,49]. Acting through the ACE/Ang II/AT1R axis (i.e., the classical RAS), Ang II is the main effector regulating vasoconstriction, sodium homoeostasis, fibrosis, cell proliferation, and the inflammation that accompanies various diseases, including liver cirrhosis [20-22,24]. However, the ACE2/ Ang-(1–7)/MasR and ACE2/Ang-(1–9)/AT2R axes, which make up the alternative RAS, have vasodilatory, antigrowth, proapoptotic, and anti-inflammatory properties that counterbalance the effects of the classical RAS axis to reduce hepatic fibrogenesis and portal hypertension [39- 42,95,96]. Accordingly, the use of classical RAS antagonists (ACEi, ARB, and AA) to treat patients with portal hypertension has been examined, with significant and promising hemodynamic results reported thus far [75,76]. These findings suggest that classical RAS pathway antagonists and alternative pathway agonists are key pharmacological molecules that may offer strategies for treating and preventing chronic liver disease and portal hypertension.
Acknowledgments
This work was supported by a research grant from Yonsei University Wonju College of Medicine (YUWCM-2008-22), a grant from the Korea Health Technology R&D Project, through the Korea Health Industry Development Institute, funded by the Ministry of Health and Welfare, Republic of Korea (HI15C2364, HI17C1365), and from the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education (NRF-2017R1D1A1A02019212, -2017R1A2B4009199, and -2017R1A5A2015369).
Figure 1.
Overview of the renin-angiotensin system (RAS). In the RAS pathways, angiotensin (Ang) II is the main effector regulating vasoconstriction, sodium homoeostasis, fibrosis, cell proliferation, and inflammation in various diseases, including liver cirrhosis, via the angiotensin-converting enzyme (ACE)/Ang II/Ang II type 1 receptor (AT1R) axis, which defines the classical RAS. Mediators of the alternative RAS have vasodilatory, antigrowth, proapoptotic, and anti-inflammatory properties that counterbalance the effects of the classical RAS axis to reduce hepatic fibrogenesis and portal hypertension. Ang II type 2 receptor (AT2R), Ang-(1–7) Mas receptor (MasR), ACE inhibitor (ACEi), AT1R blocker (ARB), mitogen-activated protein kinase (MAPK), Janus kinase (JAK), and signal transducer and activator of transcription (STAT), extracellular signal-regulated kinase (ERK), transforming growth factor β1 (TGF-β1), plasminogen activator inhibitor-1 (PAI-1), nitric oxide (NO), NO synthase (NOS), α-smooth muscle actin (α-SMA). NADPH, nicotinamide adenine dinucleotide phosphate.
REFERENCES
1. Lavoie JL, Sigmund CD. Minireview: overview of the renin-angiotensin system: an endocrine and paracrine system. Endocrinology 2003;144:2179–2183.


3. Tox U, Steffen HM. Impact of inhibitors of the renin-angiotensin-aldosterone system on liver fibrosis and portal hypertension. Curr Med Chem 2006;13:3649–3661.


4. Kang SH, Kim MY, Baik SK. Novelties in the pathophysiology and management of portal hypertension: new treatments on the horizon. Hepatol Int 2018;12(Suppl 1):112–121.


5. Paizis G, Cooper ME, Schembri JM, Tikellis C, Burrell LM, Angus PW. Up-regulation of components of the renin-angiotensin system in the bile duct-ligated rat liver. Gastroenterology 2002;123:1667–1676.


6. Bataller R, Sancho-Bru P, Gines P, et al. Activated human hepatic stellate cells express the renin-angiotensin system and synthesize angiotensin II. Gastroenterology 2003;125:117–125.


7. Sakata T, Takenaga N, Endoh T, Wada O, Matsuki K. Diagnostic significance of serum angiotensin-converting enzyme activity in biochemical tests with special reference of chronic liver diseases. Jpn J Med 1991;30:402–407.


8. Paizis G, Tikellis C, Cooper ME, et al. Chronic liver injury in rats and humans upregulates the novel enzyme angiotensin converting enzyme 2. Gut 2005;54:1790–1796.



9. Herath CB, Warner FJ, Lubel JS, et al. Upregulation of hepatic angiotensin-converting enzyme 2 (ACE2) and angiotensin-(1-7) levels in experimental biliary fibrosis. J Hepatol 2007;47:387–395.


10. Rockey D. The cellular pathogenesis of portal hypertension: stellate cell contractility, endothelin, and nitric oxide. Hepatology 1997;25:2–5.


11. Kaneda K, Ekataksin W, Sogawa M, Matsumura A, Cho A, Kawada N. Endothelin-1-induced vasoconstriction causes a significant increase in portal pressure of rat liver: localized constrictive effect on the distal segment of preterminal portal venules as revealed by light and electron microscopy and serial reconstruction. Hepatology 1998;27:735–747.


12. Bhathal PS, Grossman HJ. Reduction of the increased portal vascular resistance of the isolated perfused cirrhotic rat liver by vasodilators. J Hepatol 1985;1:325–337.


13. Grace JA, Herath CB, Mak KY, Burrell LM, Angus PW. Update on new aspects of the renin-angiotensin system in liver disease: clinical implications and new therapeutic options. Clin Sci (Lond) 2012;123:225–239.


14. Kim G, Kim MY, Baik SK. Transient elastography versus hepatic venous pressure gradient for diagnosing portal hypertension: a systematic review and meta-analysis. Clin Mol Hepatol 2017;23:34–41.




15. Dickstein K, Kjekshus J, OPTIMAAL Steering Committee of the OPTIMAAL Study Group. Effects of losartan and captopril on mortality and morbidity in high-risk patients after acute myocardial infarction: the OPTIMAAL randomised trial. Optimal Trial in Myocardial Infarction with Angiotensin II Antagonist Losartan. Lancet 2002;360:752–760.


16. Kim G, Kim J, Lim YL, Kim MY, Baik SK. Renin-angiotensin system inhibitors and fibrosis in chronic liver disease: a systematic review. Hepatol Int 2016;10:819–828.



17. Lim YL, Choi E, Jang YO, et al. Clinical implications of the serum apelin level on portal hypertension and prognosis of liver cirrhosis. Gut Liver 2016;10:109–116.




18. Kim G, Eom YW, Baik SK, et al. Therapeutic effects of mesenchymal stem cells for patients with chronic liver diseases: systematic review and meta-analysis. J Korean Med Sci 2015;30:1405–1415.



19. Lubel JS, Herath CB, Burrell LM, Angus PW. Liver disease and the renin-angiotensin system: recent discoveries and clinical implications. J Gastroenterol Hepatol 2008;23:1327–1338.


20. Suzuki Y, Ruiz-Ortega M, Lorenzo O, Ruperez M, Esteban V, Egido J. Inflammation and angiotensin II. Int J Biochem Cell Biol 2003;35:881–900.


21. Ruiz-Ortega M, Lorenzo O, Suzuki Y, Ruperez M, Egido J. Proinflammatory actions of angiotensins. Curr Opin Nephrol Hypertens 2001;10:321–329.


22. Mezzano SA, Ruiz-Ortega M, Egido J. Angiotensin II and renal fibrosis. Hypertension 2001;38(3 Pt 2):635–638.


23. Marrero MB, Schieffer B, Paxton WG, et al. Direct stimulation of Jak/STAT pathway by the angiotensin II AT1 receptor. Nature 1995;375:247–250.



24. Kagami S, Border WA, Miller DE, Noble NA. Angiotensin II stimulates extracellular matrix protein synthesis through induction of transforming growth factor-beta expression in rat glomerular mesangial cells. J Clin Invest 1994;93:2431–2437.



25. Hunyady L, Catt KJ. Pleiotropic AT1 receptor signaling pathways mediating physiological and pathogenic actions of angiotensin II. Mol Endocrinol 2006;20:953–970.



26. Mehta PK, Griendling KK. Angiotensin II cell signaling: physiological and pathological effects in the cardiovascular system. Am J Physiol Cell Physiol 2007;292:C82–C97.


27. de Gasparo M, Catt KJ, Inagami T, Wright JW, Unger T. International union of pharmacology. XXIII. The angiotensin II receptors. Pharmacol Rev 2000;52:415–472.

28. Dinh DT, Frauman AG, Johnston CI, Fabiani ME. Angiotensin receptors: distribution, signalling and function. Clin Sci (Lond) 2001;100:481–492.


30. Schieffer B, Paxton WG, Marrero MB, Bernstein KE. Importance of tyrosine phosphorylation in angiotensin II type 1 receptor signaling. Hypertension 1996;27(3 Pt 2):476–480.


31. Nguyen G, Delarue F, Burckle C, Bouzhir L, Giller T, Sraer JD. Pivotal role of the renin/prorenin receptor in angiotensin II production and cellular responses to renin. J Clin Invest 2002;109:1417–1427.



32. Feldt S, Batenburg WW, Mazak I, et al. Prorenin and renin-induced extracellular signal-regulated kinase 1/2 activation in monocytes is not blocked by aliskiren or the handle-region peptide. Hypertension 2008;51:682–688.


33. Crackower MA, Sarao R, Oudit GY, et al. Angiotensin-converting enzyme 2 is an essential regulator of heart function. Nature 2002;417:822–828.



34. Donoghue M, Hsieh F, Baronas E, et al. A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1-9. Circ Res 2000;87:E1–E9.


35. Yoshiji H, Noguchi R, Ikenaka Y, et al. Losartan, an angiotensin-II type 1 receptor blocker, attenuates the liver fibrosis development of non-alcoholic steatohepatitis in the rat. BMC Res Notes 2009;2:70.



36. Vickers C, Hales P, Kaushik V, et al. Hydrolysis of biological peptides by human angiotensin-converting enzyme-related carboxypeptidase. J Biol Chem 2002;277:14838–14843.


37. Villela DC, Passos-Silva DG, Santos RA. Alamandine: a new member of the angiotensin family. Curr Opin Nephrol Hypertens 2014;23:130–134.


38. Santos RA, Simoes e Silva AC, Maric C, et al. Angiotensin-(1-7) is an endogenous ligand for the G protein-coupled receptor Mas. Proc Natl Acad Sci U S A 2003;100:8258–8263.



39. Lima CV, Paula RD, Resende FL, Khosla MC, Santos RA. Potentiation of the hypotensive effect of bradykinin by short-term infusion of angiotensin-(1-7) in normotensive and hypertensive rats. Hypertension 1997;30(3 Pt 2):542–548.


40. Tallant EA, Clark MA. Molecular mechanisms of inhibition of vascular growth by angiotensin-(1-7). Hypertension 2003;42:574–579.


41. Henrion D, Kubis N, Levy BI. Physiological and pathophysiological functions of the AT(2) subtype receptor of angiotensin II: from large arteries to the microcirculation. Hypertension 2001;38:1150–1157.


42. Touyz RM, Schiffrin EL. Signal transduction mechanisms mediating the physiological and pathophysiological actions of angiotensin II in vascular smooth muscle cells. Pharmacol Rev 2000;52:639–672.

44. Kim G, Shim KY, Baik SK. Diagnostic accuracy of hepatic vein arrival time performed with contrast-enhanced ultrasonography for cirrhosis: a systematic review and meta-analysis. Gut Liver 2017;11:93–101.




45. Bosch J, Abraldes JG, Fernandez M, Garcia-Pagan JC. Hepatic endothelial dysfunction and abnormal angiogenesis: new targets in the treatment of portal hypertension. J Hepatol 2010;53:558–567.


46. Kim MY, Baik SK. Hyperdynamic circulation in patients with liver cirrhosis and portal hypertension. Korean J Gastroenterol 2009;54:143–148.


47. Lichtinghagen R, Michels D, Haberkorn CI, et al. Matrix metalloproteinase (MMP)-2, MMP-7, and tissue inhibitor of metalloproteinase-1 are closely related to the fibropro-liferative process in the liver during chronic hepatitis C. J Hepatol 2001;34:239–247.


48. Baik SK, Jo HS, Suk KT, et al. Inhibitory effect of angiotensin II receptor antagonist on the contraction and growth of hepatic stellate cells. Korean J Gastroenterol 2003;42:134–141.

49. Bataller R, Gines P, Nicolas JM, et al. Angiotensin II induces contraction and proliferation of human hepatic stellate cells. Gastroenterology 2000;118:1149–1156.


50. Bataller R, Schwabe RF, Choi YH, et al. NADPH oxidase signal transduces angiotensin II in hepatic stellate cells and is critical in hepatic fibrosis. J Clin Invest 2003;112:1383–1394.



51. Jang YO, Cho MY, Yun CO, et al. Effect of function-enhanced mesenchymal stem cells infected with decorin-expressing adenovirus on hepatic fibrosis. Stem Cells Transl Med 2016;5:1247–1256.



52. Moreno-Alvarez P, Sosa-Garrocho M, Briones-Orta MA, et al. Angiotensin II increases mRNA levels of all TGF-beta isoforms in quiescent and activated rat hepatic stellate cells. Cell Biol Int 2010;34:969–978.


53. Lubel JS, Herath CB, Tchongue J, et al. Angiotensin-(1-7), an alternative metabolite of the renin-angiotensin system, is up-regulated in human liver disease and has antifibrotic activity in the bile-duct-ligated rat. Clin Sci (Lond) 2009;117:375–386.


54. Pereira RM, Dos Santos RA, Teixeira MM, et al. The renin-angiotensin system in a rat model of hepatic fibrosis: evidence for a protective role of Angiotensin-(1-7). J Hepatol 2007;46:674–681.


55. Suk KT, Yoon JH, Kim MY, et al. Transplantation with autologous bone marrow-derived mesenchymal stem cells for alcoholic cirrhosis: phase 2 trial. Hepatology 2016;64:2185–2197.


56. Burrell LM, Risvanis J, Kubota E, et al. Myocardial infarction increases ACE2 expression in rat and humans. Eur Heart J 2005;26:369–375.



57. Osterreicher CH, Taura K, De Minicis S, et al. Angiotensin-converting-enzyme 2 inhibits liver fibrosis in mice. Hepatology 2009;50:929–938.



58. Albornoz L, Motta A, Alvarez D, et al. Nitric oxide synthase activity in the splanchnic vasculature of patients with cirrhosis: relationship with hemodynamic disturbances. J Hepatol 2001;35:452–456.


59. Sieber CC, Lopez-Talavera JC, Groszmann RJ. Role of nitric oxide in the in vitro splanchnic vascular hyporeactivity in ascitic cirrhotic rats. Gastroenterology 1993;104:1750–1754.


60. Kim G, Cho YZ, Baik SK. Assessment for risk of bias in systematic reviews and meta-analyses in the field of hepatology. Gut Liver 2015;9:701–706.




61. Tandon P, Abraldes JG, Berzigotti A, Garcia-Pagan JC, Bosch J. Renin-angiotensin-aldosterone inhibitors in the reduction of portal pressure: a systematic review and meta-analysis. J Hepatol 2010;53:273–282.


62. Park DH, Baik SK, Choi YH, et al. Inhibitory effect of angiotensin blockade on hepatic fibrosis in common bile duct-ligated rats. Korean J Hepatol 2007;13:61–69.

63. Angus PW. Role of endothelin in systemic and portal resistance in cirrhosis. Gut 2006;55:1230–1232.



64. Kim G, Huh JH, Lee KJ, Kim MY, Shim KY, Baik SK. Relative adrenal insufficiency in patients with cirrhosis: a systematic review and meta-analysis. Dig Dis Sci 2017;62:1067–1079.



65. Ferrario CM, Trask AJ, Jessup JA. Advances in biochemical and functional roles of angiotensin-converting enzyme 2 and angiotensin-(1-7) in regulation of cardiovascular function. Am J Physiol Heart Circ Physiol 2005;289:H2281–H2290.


66. Lebrec D, Nouel O, Corbic M, Benhamou JP. Propranolol: a medical treatment for portal hypertension? Lancet 1980;2:180–182.


67. Tripathi D, Hayes PC. Beta-blockers in portal hypertension: new developments and controversies. Liver Int 2014;34:655–667.


68. Poynard T, Cales P, Pasta L, et al. Beta-adrenergic-antagonist drugs in the prevention of gastrointestinal bleeding in patients with cirrhosis and esophageal varices: an analysis of data and prognostic factors in 589 patients from four randomized clinical trials. Franco-Italian Multicenter Study Group. N Engl J Med 1991;324:1532–1538.


69. Garcia-Tsao G, Bosch J. Management of varices and variceal hemorrhage in cirrhosis. N Engl J Med 2010;362:823–832.


70. Feu F, Garcia-Pagan JC, Bosch J, et al. Relation between portal pressure response to pharmacotherapy and risk of recurrent variceal haemorrhage in patients with cirrhosis. Lancet 1995;346:1056–1059.


71. Groszmann RJ, Garcia-Tsao G, Bosch J, et al. Beta-blockers to prevent gastroesophageal varices in patients with cirrhosis. N Engl J Med 2005;353:2254–2261.


72. Garcia-Pagan JC, Feu F, Navasa M, et al. Long-term haemodynamic effects of isosorbide 5-mononitrate in patients with cirrhosis and portal hypertension. J Hepatol 1990;11:189–195.


73. Albillos A, Lledo JL, Banares R, et al. Hemodynamic effects of alpha-adrenergic blockade with prazosin in cirrhotic patients with portal hypertension. Hepatology 1994;20:611–617.


74. Esler M, Dudley F, Jennings G, et al. Increased sympathetic nervous activity and the effects of its inhibition with clonidine in alcoholic cirrhosis. Ann Intern Med 1992;116:446–455.


75. Debernardi-Venon W, Martini S, Biasi F, et al. AT1 receptor antagonist Candesartan in selected cirrhotic patients: effect on portal pressure and liver fibrosis markers. J Hepatol 2007;46:1026–1033.


76. Schneider AW, Kalk JF, Klein CP. Effect of losartan, an angiotensin II receptor antagonist, on portal pressure in cirrhosis. Hepatology 1999;29:334–339.


77. Baik SK, Park DH, Kim MY, et al. Captopril reduces portal pressure effectively in portal hypertensive patients with low portal venous velocity. J Gastroenterol 2003;38:1150–1154.


78. Kim MY, Baik SK, Park DH, et al. Angiotensin receptor blockers are superior to angiotensin-converting enzyme inhibitors in the suppression of hepatic fibrosis in a bile duct-ligated rat model. J Gastroenterol 2008;43:889–896.


79. Heim MH, Jacob L, Beglinger C. The angiotensin II receptor antagonist candesartan is not effective in reducing portal hypertension in patients with cirrhosis. Digestion 2007;75:122–123.


80. De BK, Bandyopadhyay K, Das TK, et al. Portal pressure response to losartan compared with propranolol in patients with cirrhosis. Am J Gastroenterol 2003;98:1371–1376.


81. Kim JH, Kim JM, Cho YZ, et al. Effects of candesartan and propranolol combination therapy versus propranolol monotherapy in reducing portal hypertension. Clin Mol Hepatol 2014;20:376–383.



82. Kim MY, Cho MY, Baik SK, et al. Beneficial effects of candesartan, an angiotensin-blocking agent, on compensated alcoholic liver fibrosis: a randomized open-label controlled study. Liver Int 2012;32:977–987.


83. Abecasis R, Kravetz D, Fassio E, et al. Nadolol plus spironolactone in the prophylaxis of first variceal bleed in nonascitic cirrhotic patients: a preliminary study. Hepatology 2003;37:359–365.


84. Sugano S, Kawafune T, Okajima T, Ishii K, Watanabe M, Takamura N. Chronic splanchnic hemodynamic effects of spironolactone with unrestricted sodium diet in patients with compensated cirrhosis. Dig Dis Sci 1998;43:893–897.


85. Schepke M, Werner E, Biecker E, et al. Hemodynamic effects of the angiotensin II receptor antagonist irbesartan in patients with cirrhosis and portal hypertension. Gastroenterology 2001;121:389–395.


86. Yokohama S, Tokusashi Y, Nakamura K, et al. Inhibitory effect of angiotensin II receptor antagonist on hepatic stellate cell activation in non-alcoholic steatohepatitis. World J Gastroenterol 2006;12:322–326.



87. Terui Y, Saito T, Watanabe H, et al. Effect of angiotensin receptor antagonist on liver fibrosis in early stages of chronic hepatitis C. Hepatology 2002;36(4 Pt 1):1022.


88. Sookoian S, Fernandez MA, Castano G. Effects of six months losartan administration on liver fibrosis in chronic hepatitis C patients: a pilot study. World J Gastroenterol 2005;11:7560–7563.



89. Rimola A, Londono MC, Guevara G, et al. Beneficial effect of angiotensin-blocking agents on graft fibrosis in hepatitis C recurrence after liver transplantation. Transplantation 2004;78:686–691.


90. Corey KE, Shah N, Misdraji J, et al. The effect of angiotensin-blocking agents on liver fibrosis in patients with hepatitis C. Liver Int 2009;29:748–753.



91. Colmenero J, Bataller R, Sancho-Bru P, et al. Effects of losartan on hepatic expression of nonphagocytic NADPH oxidase and fibrogenic genes in patients with chronic hepatitis C. Am J Physiol Gastrointest Liver Physiol 2009;297:G726–G734.



92. Abu Dayyeh BK, Yang M, Dienstag JL, Chung RT. The effects of angiotensin blocking agents on the progression of liver fibrosis in the HALT-C trial cohort. Dig Dis Sci 2011;56:564–568.



93. Goh GB, Pagadala MR, Dasarathy J, et al. Renin-angiotensin system and fibrosis in non-alcoholic fatty liver disease. Liver Int 2015;35:979–985.


94. Zhu Q, Li N, Li F, et al. Therapeutic effect of renin angiotensin system inhibitors on liver fibrosis. J Renin Angiotensin Aldosterone Syst 2016;17:1470320316628717.



-
METRICS

- Related articles
-
The renin-angiotensin system and aging in the kidney2014 May;29(3)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print


