Application of Cystatin C Reduction Ratio to High-Flux Hemodialysis as an Alternative Indicator of the Clearance of Middle Molecules
Article information
Abstract
Background/Aims
Although high-flux (HF) dialyzers with enhanced membrane permeability are widely used in current hemodialysis (HD) practice, urea kinetic modeling is still being applied to indicate the adequacy of both low-flux (LF) and HF HD. In comparison with urea (molecular weight, 60 Da) and β2-microglobulin (β2MG, 12 kDa), cystatin C (CyC, 13 kDa) is a larger molecule that has attractive features as a marker for assessing solute clearance. We postulated that CyC might be an alternative for indicating the clearance of middle molecules (MMs), especially with HF HD.
Methods
Eighty-nine patients were divided into LF and HF groups. Using single pool urea kinetic modeling, the urea reduction ratio (URR) and equilibrated Kt/Vurea (eKt/Vurea) were calculated. The serum CyC concentrations were measured using particle-enhanced immunonephelometry. As indices of the middle molecular clearance, the reduction ratios of β2MG and CyC were calculated.
Results
The β2MG reduction ratio (β2MGRR) and CyC reduction ratio (CyCRR) were higher in the HF group compared to the LF group. However, the URR and eKt/Vurea did not differ between the two groups. The CyCRR was significantly correlated with the eKt/Vurea and β2MGRR (r = 0.47 and 0.69, respectively, both p < 0.0001).
Conclusions
Compared to the LF dialyzer, the HF dialyzer removed CyC and β2MG more efficiently. Unlike the β2MGRR, the CyCRR was correlated with the eKt/Vurea and β2MGRR. This study suggests a role for the CyCRR as an alternative indicator of the removal of MMs.
INTRODUCTION
The uremic syndrome is attributable to the progressive retention of a large number of compounds, which are called uremic retention solutes or uremic toxins, when they interact negatively with physiologic function. They include not only small plasma solutes, but also protein-bound solutes and middle molecules (MMs) [1]. Their retention may lead to dialysis morbidity and mortality, which are poorly resolved with conventional hemodialysis (HD).
Since the beginning of the 1980s, urea kinetic modeling has been applied to quantify the removal of small solutes by conventional HD. In current practice, high-flux (HF) dialyzers may allow more efficient removal of larger molecules, which is expected to reduce the risk of HD-related morbidity and mortality substantially. Conversely, no consensus exists concerning the best dialysis dose index and method for calculating it [2,3].
Of the MMs, cystatin C (CyC, molecular weight [MW], 13 kDa) is freely filtered at the level of the glomerulus and virtually all is reabsorbed and metabolized by proximal tubular cells [4-7]. Due to its molecular characteristics, numerous studies have shown that CyC is the most promising endogenous marker of the glomerular filtration rate [8]. In addition, several studies have suggested that CyC is useful as a marker of HD toxin removal, since the plasma CyC has attractive features as a representative MM [9,10].
For these reasons, we hypothesized that the CyC reduction ratio (CyCRR) might reflect the removal of MMs and could be applied to the dialysis dose index in HD, and we designed this single-center, case-control study to examine our hypotheses.
METHODS
Patients
All of the patients underwent maintenance HD at Hanyang University Hospital three times per week. Previously, all 89 patients had chosen low-flux (LF) HD as the dialysis modality. The dialysis modality was chosen according to patient preference, and the dialyzers used were F6HPS for LF and F60S for HF (Fresenius Medical Care, Bad Homburg, Germany). We assumed that no residual renal function was preserved in any patient. Patients were excluded if they were medically unstable, younger than 18 years of age, or mentally disabled.
Measurements
All blood samples were taken before and after the second HD session of the week, according to the guidelines for HD adequacy [11]. Blood samples were collected in tubes without additional anticoagulant and allowed to stand at room temperature for 30 to 60 minutes. Then, the samples were centrifuged to collect serum, which was stored at -70℃ until assayed. Urea nitrogen was measured using a Hitachi 7600 series automatic biochemical analyzer (Hitachi High-Technologies, Tokyo, Japan). Serum β2-microglobulin (β2MG, MW, 12 kDa) concentrations were measured using a chemiluminescent enzyme immunoassay (DPC IMMUNLITE 2000, Diagnostic Products, Los Angeles, CA, USA). CyC was assayed using particle-enhanced immunonephelometry (BN II nephelometer, Dade Behring, Reuil Malmaison, France). All other measurements were made using routine laboratory methods.
We calculated the reduction ratios of solutes for urea, β2MG, and CyC during the treatment. The single-pool urea Kt/V (spKt/Vurea) values were calculated using the second-generation natural logarithmic formula described by Daugirdas [12], and we transformed the spKt/Vurea to the equilibrated Kt/Vurea (eKt/Vurea) [13].
The clearance of β2MG (Kβ2MG) was determined using the formula described by Cheung et al. [14] and then the Kt/Vβ2MG was calculated by multiplying the Kβ2MG by the treatment time and dividing the result by the post-dialysis volume.
Statistical analysis
Baseline characteristics are presented as the mean ± SD or as the proportions of patients in the groups. The Shapiro-Wilk test was used to test for normality. Continuous variables were compared using the Mann-Whitney U test. The chi-square test with Fisher's exact test was applied to evaluate associations between nominal variables. The Spearman correlation coefficient was used to study correlations between variables. All p values were analyzed using two-sided tests, and p values < 0.05 were considered statistically significant. All analyses were performed with SAS version 9.1 (SAS Institute, Cary, NC, USA).
RESULTS
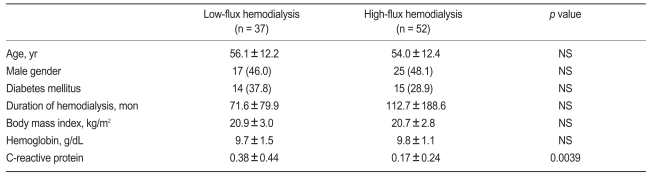
Eighty-nine patients were included and divided into LF (n = 37) and HF (n = 52) groups. Dialysis was performed for 4.0 ± 0.2 hr/session with blood flow rates of 237 ± 23 mL/min and dialysate flow rates of 500 mL/min using bicarbonate dialysate. The dialysis machine, water treatment circuit, and tank were sterilized regularly.
No differences existed between the two groups with respect to the demographic characteristics and the cause of end-stage renal disease (Table 1). The C-reactive protein was higher in the LF HD group (p = 0.0039), but was not correlated with the indices of solute removal in this study (data not shown). The etiology of renal failure in the patients included diabetes mellitus (n = 29, 32.6%), hypertension (n = 18, 20.2%), chronic glomerulonephritis (n = 12, 13.5%), and shrunken kidneys of unknown etiology (n = 23, 25.8%).
The eKt/Vurea in the LF and HF HD groups was 1.19 ± 0.32 and 1.29 ± 0.23, respectively. No differences were observed in the urea reduction ratio (URR) and eKt/Vurea between the two groups (Table 2). However, β2MG and CyC were removed more efficiently by the HF dialyzer (p < 0.0001).
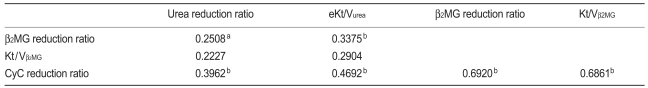
When all patients were considered, the CyCRR was strongly correlated with the β2MG reduction ratio (β2MGRR) and Kt/Vβ2MG and weakly correlated with the URR and eKt/Vurea (Table 3). The pre-dialysis CyC and β2MG levels, and their reduction ratios were not correlated with the albumin, normalized protein catabolic rate, or C-reactive protein (data not shown).
DISCUSSION
We assessed the performance of the CyCRR as representative of the clearance of MMs. As in previous reports [14,15], no difference existed between the two groups in terms of the URR and eKt/Vurea. In addition, the more efficient removal of β2MG in the HF HD group concurred with a past study [14]. However, we found that the CyCRR had a prominent relationship with the β2MGRR and Kt/Vβ2MG in HD.
Most of the small water-soluble uremic retention compounds and MMs that have been shown to exert biologic action have an intradialytic kinetic behavior that is indisputably different from that of urea [16], and their retention is poorly resolved with conventional HD. Therefore, many past studies have emphasized that the removal of small water-soluble uremic retention compounds and MMs using a HF dialyzer may ensure excellent dialysis quality, influence the clinical outcomes of patients in several areas, and reduce the acute and long-term HD-related complications [1,17-20]. These may be particularly important in patients with no residual renal function, who depend completely on dialysis efficiency. Assessing dialysis adequacy using several molecules simultaneously is suitable to reflect small solute and MM clearance [21].
β2MG had been accepted as representative of MMs and is widely recognized as a key component in the development of dialysis-associated amyloidosis [22]. Several studies demonstrated that the use of a HF dialyzer might improve the clearance of β2MG and decrease pre-dialysis plasma levels [23,24]. They expected that HD with a HF dialyzer could reduce the development of amyloidosis and improve patient quality of life. Given the fluctuating β2MG production rate, nonrenal route of elimination, other factors influencing its bloodstream delivery, and intercompartmental transport, β2MG removal is recognized as not being a good indicator [25,26].
After exploring the interrelationship of low-molecular-weight proteins with the glomerular filtration rate, an increasing serum CyC level was found to be related to decreasing renal function and to be associated with some cerebral amyloid angiopathies, tumor progression, and inflammatory processes [27-30]. In addition, some attractive features as a representative MM made us assess the performance of CyC during HD: because CyC should be distributed strictly in extracellular fluid, various kinetic models are not required to describe its kinetics during HD; its production rate is relatively constant or minimally variable; CyC is a free-circulating, unbounded form, and its elimination from the circulation is almost entirely through glomerular filtration; and CyC may be a prognostic biomarker of the risk of death and cardiovascular disease [31,32].
Starting from the hypothesis that the clearance of CyC is representative of the removal of MMs and its reduction ratio is a useful marker of toxin removal in HD, we performed this study to compare the CyCRR with the most widely used methods for measuring HD adequacy. Since CyC has several merits as a representative MM - it is similar to β2MG in molecular weight and clearance during HD, and its reduction ratio has some relationship with the URR and eKt/Vurea - the CyCRR should be useful as an alternative to the β2MGRR and Kt/Vβ2MG for estimating the dialysis clearance of MMs. However, further studies with larger sample sizes should be performed to draw a firm conclusion as to whether the CyCRR is useful indicator of the clearance of MMs.
Notes
No potential conflict of interest relevant to this article was reported.