Cumulative exposure to metabolic syndrome increases thyroid cancer risk in young adults: a population-based cohort study
Article information
Abstract
Background/Aims
A re-increasing trend of thyroid cancer since 2015 has been observed despite a similar examination rate, and the incidence of thyroid cancer among young adults continues to rise.
Methods
This study used data from the Korean National Health Insurance Service. Individuals 20–39 years of age who underwent ≥ 4 health checkups from 2009–2013 were enrolled and followed throughout 2019. To quantify the metabolic burden, groups were divided by the number of diagnoses of metabolic syndrome across four consecutive health examinations.
Results
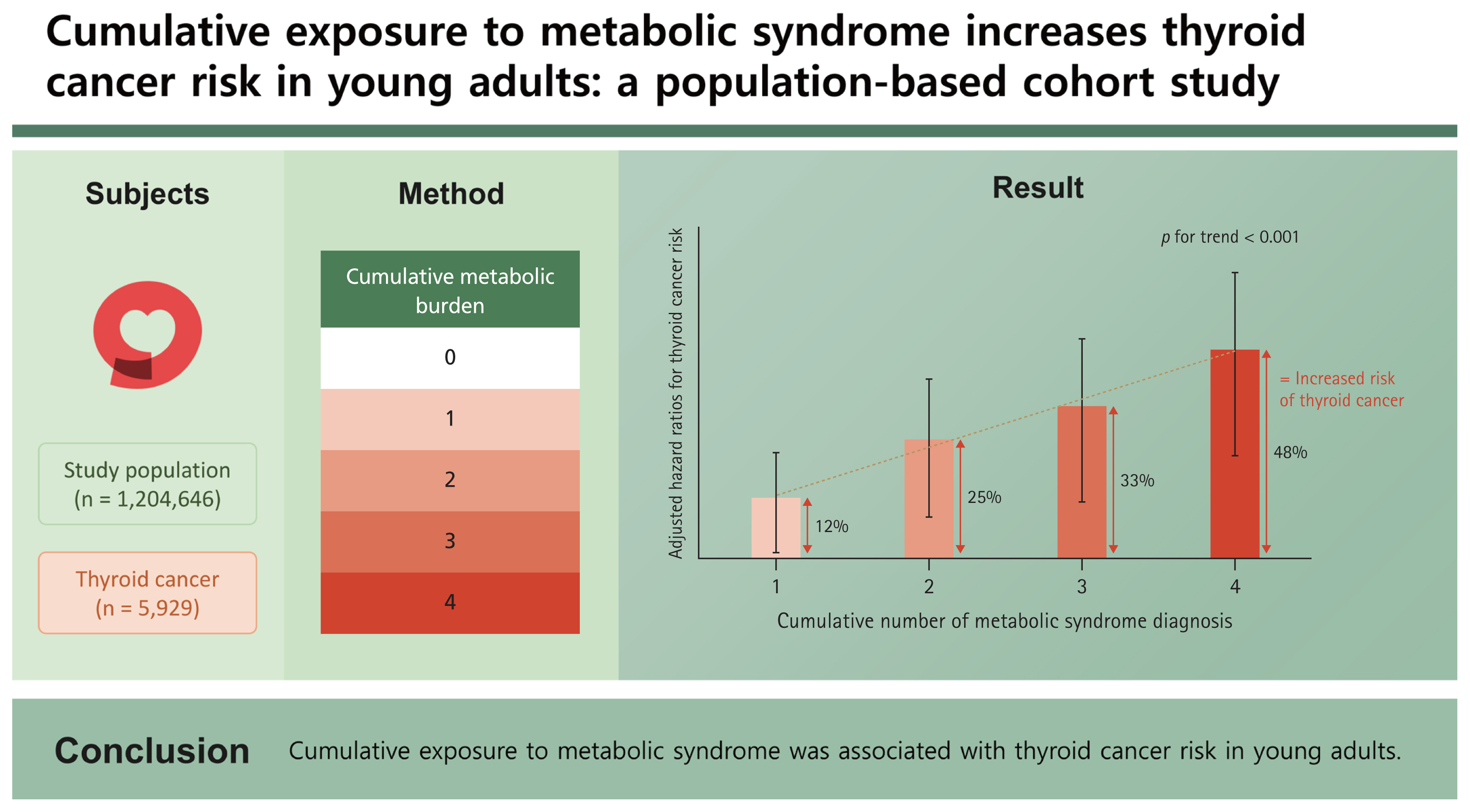
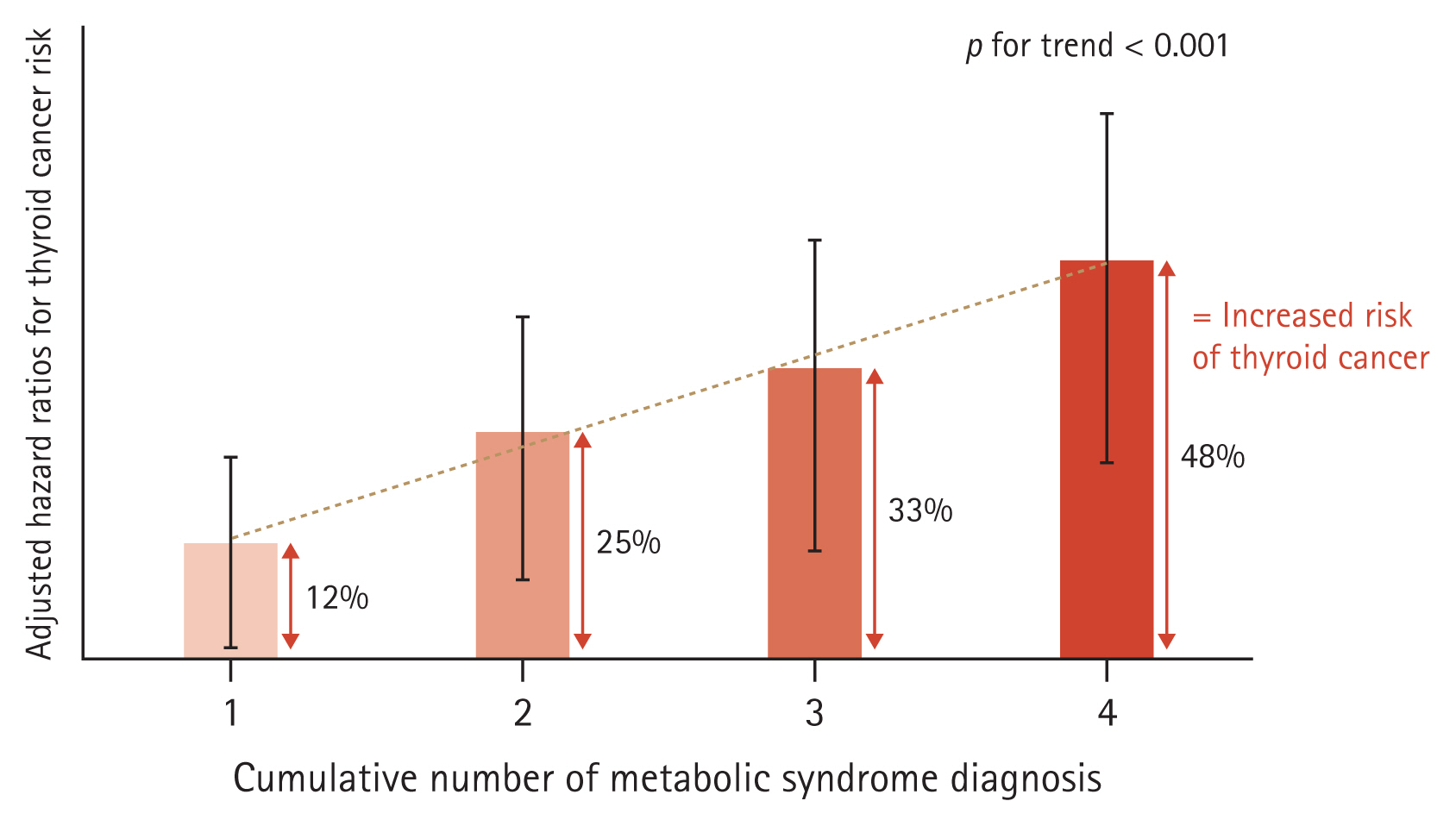
Among the study population (n = 1,204,646), 5,929 (0.5%) were diagnosed with thyroid cancer during a follow-up period of 5 years. The hazard ratio (95% confidence interval) values of thyroid cancer occurrence according to the number (1–4) of diagnoses of metabolic syndrome across the four health examinations compared to the group without met-abolic syndrome were significantly greater, as follows: 1.12 (1.02–1.23), 1.25 (1.10–1.42), 1.33 (1.15–1.55), and 1.48 (1.25–1.75) (p for trend < 0.01), respectively. Each component of metabolic syndrome showed a significant increase in hazard ratio according to the number of diagnoses except for impaired fasting glucose criteria.
Conclusions
Cumulative exposure to metabolic syndrome was associated with thyroid cancer risk in young adults.
INTRODUCTION
Widespread screening for thyroid cancer in Korea started in 1999, and the incidence of thyroid cancer increased by approximately 15-fold from then to 2011 [1]. Because treatment for small, indolent thyroid cancer does not improve survival rates, caution is needed regarding excessive screening and over-treatment of low-risk thyroid cancer [2]. As clinicians focused on addressing over-diagnosis, the incidence of thyroid cancer showed a decrease after peaking in 2015. However, a re-increasing trend of thyroid cancer since 2015 has been observed despite a similar examination rate, and this increase is difficult to explain only based on the diagnosis rate of small, low-risk thyroid cancer [3]. In addition, thyroid cancer incidence has not decreased in the population < 30 years of age since 2010 [4]. This suggests the need for additional studies on young-onset thyroid cancer.
Previous studies have suggested that obesity is a risk factor for thyroid cancer [5,6]. In a meta-analysis, the risk of thyroid cancer increased in a dose-dependent manner with increasing body mass index [7]. Another study suggested that losing weight reduced the risk of thyroid cancer [8]. Metabolic syndrome has also been identified as a risk factor for thyroid cancer [9]. In particular, the risk is greater in metabolically unhealthy individuals than in people with obesity without morbidity [10]. For early-onset cancers, some studies suggest that the probability of influence from hereditary factors is higher than in late-onset cases [11]. However, the incidence of cancer among young adults is increasing across cancer types, and the etiologic role of early-life risk exposure could be an important consideration [12]. Moreover, it is thought that a growing metabolic burden at a young age may negatively affect later health. Therefore, decreasing the exposure of young adults to metabolic abnormalities can be a key concept in the prevention of thyroid cancer.
In the present study, we investigated the risk of thyroid cancer associated with cumulative exposure to metabolic syndrome and its components among young adults in their 20s and 30s.
METHODS
Study population
The present study included adults 20–40 years of age who underwent four or more consecutive health checkups from 2009–2013 provided by the Korean National Health Insurance Service [13]. All patients diagnosed with any type of cancer (C code assigned to cancer patients for medical insurance reimbursement for a serious illness) in 2014 were excluded, and a diagnosis of thyroid cancer (C73 in claims data) during follow-up until 2019 was confirmed (Fig. 1). The protocol of this study was confirmed by the Institutional Review Board (SC22ZESI0025) of Yeouido St. Mary’s Hospital, and it was completed in accordance with the Declaration of Helsinki. The requirement for informed consent was exempted due to the use of anonymized data.
Measurements and definitions
Individuals undergoing a medical examination were advised to fast for 8 hours before blood testing. Height, weight, and blood pressure were measured with an automated electronic device, and waist circumference (below the lowest rib and above the pelvic bone) was measured by an experienced technician.
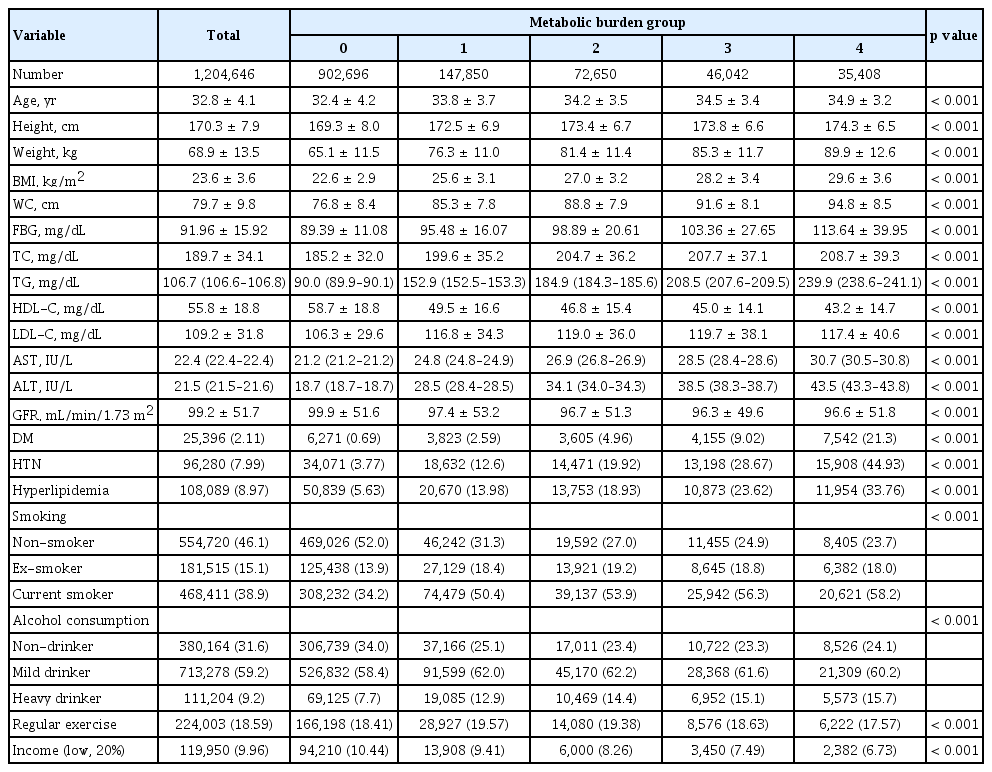
Age, sex, current prescription medications, alcohol consumption, cigarette smoking, regular exercise, and income status were identified using standardized questionnaires. For statistical analysis, categorization was performed according to the amount of smoking, alcohol consumption, and regular exercise. Smoking status was divided into three groups, as follows: non-smoker, ex-smoker, and current smoker. Alcohol consumption was classified as non-drinker, mild drinker, or heavy drinker, with the latter two categories based on an alcohol consumption cutoff of 30 g per day. Regular exercise was defined as light exercise more than five days a week or vigorous exercise that increased heart rate more than 3 days a week. The low-income group was defined as the bottom 20% of income earners or those under medical aid.
Diabetes mellitus was diagnosed by a fasting glucose level of ≥ 126 mg/dL or the use of anti-diabetic pharmaceuticals. Hypertension (HTN) was defined by a systolic blood pressure of ≥ 140 mmHg, diastolic blood pressure of ≥ 90 mmHg, or the use of blood pressure medication. Hyperlipidemia was diagnosed by a total cholesterol level of ≥ 240 mg/day or the use of lipid-lowering medication. Metabolic syndrome was noted according to the definition in Korea [14] and diagnosed when three or more of the following five criteria were met: (a) waist circumference > 90 cm for men or > 80 cm for women, (b) serum triglycerides > 150 mg/dL, (c) high-density lipoprotein cholesterol (HDL-C) < 40 mg/dL in men or < 50 mg/dL in women, (d) blood pressure ≥ 130/85 mmHg or current use of anti-hypertensive drugs, and (e) fasting blood glucose ≥ 100 mg/dL or current use of oral hypoglycemic agents or insulin for diabetes.
Statistical analysis
For descriptive statistics, continuous variables following a normal distribution were expressed as mean and standard deviation values, and continuous variables not following a normal distribution were expressed as median and inter-quartile range values. For categorical variables, median values and ratios were expressed. The incidence rate of thyroid cancer was expressed as an incidence rate of 1,000 person-years, and the hazard ratio (HR) based on the follow-up period was performed using univariate and multivariate Cox proportional regression analyses. Adjusted HRs and their 95% confidence intervals (CIs) were used to express the results. Trend analysis was performed to confirm the increasing trend of HR, and interaction analysis was performed to analyze the difference in HR based on sex and body mass index (BMI) in subgroup analysis. Statistical analysis was performed using SAS version 9.3 (SAS Institute Inc., Cary, NC, USA) and R version 3.2.3 (The R Foundation for Statistical Computing, Vienna, Austria).
RESULTS
Baseline characteristics of the study population
To quantify metabolic burden, individuals were divided based on the cumulative number of metabolic syndrome diagnoses across four consecutive health checkups, and the baseline characteristics for each group are presented in Table 1. Metabolic parameters, including age and comorbid disease, showed significant differences between the four groups. The participants tended to be older and heavier as the cumulative metabolic burden increased. In addition, a higher rate of concomitant diseases, such as diabetes, HTN, and hyperlipidemia, was observed. In terms of lifestyle, the high metabolic burden group smoked more, consumed more alcohol, and exercised less regularly (p < 0.001).
Cumulative metabolic status and the risk of thyroid cancer
Among the 1,204,646 enrolled individuals in their 20s and 30s, 5,929 (0.5%) were diagnosed with thyroid cancer during the five-year follow-up period. As the number of diagnoses of metabolic syndrome increased, baseline metabolic status also tended to deteriorate, so additional correction was performed to confirm the influence of cumulative exposure. The adjusted HRs (95% CIs) of thyroid cancer occurrence based on the number of diagnoses of metabolic syndrome across the four health checkups compared to the group without diagnosis of metabolic syndrome were cumulatively greater: 1.12 (1.02–1.23), 1.25 (1.10–1.42), 1.33 (1.15–1.55), and 1.48 (1.25–1.75) for 1–4 metabolic syndrome diagnoses, respectively (Fig. 2, Supplementary Table 1).
Subgroup analyses
In subgroup analysis, each component of metabolic syndrome except impaired fasting glucose showed a significant increase in adjusted HR according to the number of diagnoses made (Fig. 3, Supplementary Table 1). In addition, statistical significance was confirmed in both sex and BMI (cutoff, 25 kg/m2)-based subgroups, and no difference was observed in the effect of subgroup placement (Supplementary Table 2).

Adjusted (age, sex, smoking, alcohol consumption, regular exercise, low income, and baseline metabolic status of each metabolic syndrome component) hazard ratios for thyroid cancer risk according to the number of diagnoses made with each metabolic syndrome component. 95% confidence intervals for hazard ratios are presented in Supplementary Table 1. MetS, metabolic syndrome; WC, waist circumference; BP, blood pressure; HDL-C, high-density lipoprotein cholesterol; TG, triglycerides; IFG, impaired fasting glucose.
DISCUSSION
In the present study, we suggest that cumulative exposure to metabolic syndrome increases the risk of thyroid cancer among young adults in their 20s and 30s. In a previous large-scale study of the Korean population, metabolic syndrome was found to be a significant risk factor for thyroid cancer [9]. However, metabolic abnormalities are modifiable risk factors, and the study did not include an assessment of changing metabolic status. Once initiated in young adults, metabolic burden is thought to have ongoing effects during the aging process. The improvement of metabolic status in young adults needs to be emphasized in this context given that it may prevent not only the onset of metabolic diseases but also various types of cancer [15].
In this study, we evaluated the change in metabolic status using data from four repeated health examinations, and groups were designed based on the number of repeated diagnoses of metabolic syndrome. As the number of diagnoses of metabolic syndrome increased, patient age and baseline BMI tended to increase. The accompanying rate of metabolic diseases such as HTN, hyperlipidemia, and diabetes increased as well, and the proportions of current smokers and heavy drinkers were also high (Table 1). Even after correcting for these baseline differences, the cumulative effect of metabolic syndrome on thyroid cancer risk was confirmed by an increase in adjusted HR (p for trend < 0.001, Fig. 2) compared to the group without metabolic syndrome in all health examinations as a reference, and this effect was consistently significant in subgroup analysis by sex and obesity status (Supplementary Table 2).
In the analysis based on each component of metabolic syndrome, waist circumference, HTN, serum triglycerides, and HDL-C were significant. However, the cumulative effect was not significant for the classification criteria of impaired fasting glucose (Fig. 3). In previous studies, increases in insulin resistance as well as thyroid volume and nodule number were reported [16,17], and these associations have also been documented in thyroid cancer studies [18,19]. In addition, type 2 diabetes has been suggested to increase the risk of thyroid cancer [20]. However, a significant increase in thyroid cancer was not found in prediabetic participants in this study (Fig. 3, Supplementary Table 2). Several diabetes drugs have been suggested to exert anti-cancer effects [21,22], but additional analysis of drug claims data could not be performed in this study, which was a limitation of our study. Regarding the cumulative effect of impaired fasting glucose on the risk of thyroid cancer, the use of pharmaceuticals, especially anti-diabetic drugs, should be further investigated.
The cumulative effect of low HDL-C as a component of metabolic syndrome increased the risk of thyroid cancer in this study. The association between low HDL-C and thyroid cancer has been reported in several studies [10,23,24]; however, the correlation between low HDL-C and thyroid nodules was not confirmed [16]. Therefore, HDL-C as a protective factor for tumorigenesis is one hypothesis for the association between metabolic syndrome and thyroid cancer. This association may be related to the characteristics of cancer, such as increased expression of HDL-C receptors [25], and further studies are needed to confirm a causal relationship. In addition, an association between statin-type drugs and reduced cancer risk has been reported [23]; this drug effect should be considered in future studies. Despite these limitations, an association between low HDL-C and the risk of several cancers has been reported in a number of studies [24,26], and low HDL-C is being considered a therapeutic target for future use as a biomarker for cancer diagnosis or the development of potential agents [27].
In previous studies, associations between obesity and metabolic syndrome and the occurrence of thyroid cancer were reported and were particularly significant in men [9,28]. From another perspective, the role of estrogen was suggested to play a role in the mechanism of cancer development [29]. However, differences based on sex were not observed in the present study. The results of this study should be interpreted with caution because older men and menopausal women were excluded from our analysis; only young adults were included. Moreover, the diagnostic criteria for metabolic syndrome [30] present differently for men and women, which should be considered when interpreting the effects of metabolic syndrome based on sex.
In addition, although this study was conducted on young adults, the influence of metabolic syndrome on the occurrence of thyroid cancer may be considered important in the middle-aged or older population. In another cross-sectional study of Korean adults aged 40–70 years, the association between metabolic syndrome and thyroid cancer incidence was significant, and the study reported an increase in the number of diagnoses for each component of metabolic syndrome and thyroid cancer risk [31]. The study reported on metabolic burden, including the number of diagnostic criteria as well as the cumulative number of diagnoses. The study also suggests that the correlation between thyroid cancer risk and metabolic syndrome is not limited to young adults. Further research is required to determine the difference in the influence of metabolic syndrome on early-onset thyroid cancer compared to late-onset thyroid cancer.
This study had several limitations. First, thyroid ultrasound is not routine for all individuals in the Korean insurance system. Patients with metabolic syndrome are likely to visit the hospital more frequently due to several related diseases, such as diabetes and cardiovascular events, and the increased number of hospital visits may lead to greater detection of small thyroid cancers. However, the subjects of this study are composed of individuals who received annual checkups, and it is assumed that they have sufficient medical access and resources. Specifically, the incidence rate of thyroid cancer in 1,000 person-years was 0.91 in the current study, and the corresponding incidence rate in adults < 40 years of age in the Korean Cancer Center Registry data was 1.17 [4]. This means that the incidence of thyroid cancer in this study population was similar to those in the general population. Therefore, it is considered that the subject group of this study may have sufficient representation of the entire population. Second, small thyroid nodules are sometimes observed without biopsy, and most thyroid cancers do not grow rapidly [32]. Therefore, a considerable amount of time may be required for the diagnosis of thyroid cancer, and a follow-up time of five years may be rather short. Lastly, pathological classification of thyroid cancer was not possible with the data used in this study. Additional analysis could not be performed on carcinomas in which genetic factors have a significant influence such as medullary thyroid cancer. We also cannot further analyze the association with the diagnosis of localized thyroid cancer due to the absence of thyroid cancer stage information in this data set.
One of the best-understood environmental risk factors for thyroid cancer is exposure to radiation [33]. In addition, exposure to air pollution, heavy metals, and various chemical compounds can increase the risk of thyroid cancer [34]. The influence of obesity and metabolic syndrome may be a comparatively minor issue, but the prevalence of abdominal obesity in Korean adults has rapidly increased from 19% to 24% in the past 10 years [35]. Moreover, metabolic syndrome is a modifiable factor that can be addressed by lifestyle interventions to decrease risk at an individual level.
In conclusion, we found a correlation between cumulative metabolic status and the risk of thyroid cancer among Korean young adults in their 20s and 30s.
KEY MESSAGE
1. Although clinicians continue to pay attention to over-diagnosis, the incidence of thyroid cancer has not decreased among young adults.
2. The incidence of cancer among young adults is increasing across cancer types, and the etiologic role of early-life risk exposure is considered important.
3. The results of this study indicate that cumulative exposure to metabolic syndrome can increase the risk of developing thyroid cancer in young adults.
Notes
Conflict of interest
The authors disclose no conflicts.
CRedit authorship contributions
Jinyoung Kim: funding acquisition, visualization, writing - original draft; Kyungdo Han: data curation, formal analysis, methodology; Mee Kyoung Kim: writing - review & editing; Ki-Hyun Baek: writing - review & editing; Ki-Ho Song: writing - review & editing; Hyuk-Sang Kwon: conceptualization, project administration
Funding
This research was supported by a grant from the Institute of Clinical Medicine Research at Yeouido St. Mary’s Hospital, The Catholic University of Korea.