Effects of Mixed Chimerism and Immune Modulation on GVHD, Disease Recurrence and Survival after HLA-identical Marrow Transplantation for Hematologic Malignancies
Article information
Abstract
Background
The success of allogeneic bone marrow transplantation (allo-BMT) is affected by underlying disease relapse. Although mixed chimerism (MC) is not necessarily a poor prognostic factor, several groups have suggested that MC is associated with an increased risk of disease relapse. There is evidence that patients with MC benefit from additional immunotherapy if the treatment is started in minimal residual disease status (mixed chimerism status), not in frank hematological relapse. The purposes of this study are to evaluate 1) the risk for relapse or graft rejection in correlation to persistent MC status after allo-BMT, and 2) the possibility of preventing relapse by immune modulation treatments (withdrawal or rapid taper-off of post-transplant immuno-suppression, additional interferon treatment, or the administration of donor lymphocytes) in hematologic malignancies.
Patients and Methods
Of 337 allogeneic donor-recipient pairs between March 1996 and August 1998, 12 patients who showed persistent or progressive MC and who received immune modulation treatments were evaluated. Twelve patients, median age 31 years (range 9 to 39 years), received an allo-BMT for: acute myelogenous leukemia (AML, n = 5), chronic myelogenous leukemia (CML, n = 4), acute lymphocytic leukemia (ALL, n = 3). Serial polymerase chain reaction (PCR) analysis of YNZ 22-, 33.6-minisatellites or Y chromosome-specific PCR analysis at short term intervals (pre-and post-transplant 1, 3, 6, 9, … months) was performed. Once MC was detected, immune modulation treatments on the basis of increasing MC in an early phase of recurrence of underlying disease were started.
Results
Nine of 12 patients converted to complete chimerism (CC) (AML 5/5, CML 3/4, ALL 1/3). Four of 9 CC patients developed graft-versus-host disease (GVHD) grade ≤2 during immune modulation. All were treated successfully with steroids. Three patients who were not converted to CC showed relapse of underlying diseases or graft failure.
Conclusion
The results demonstrate that, in patients with hematologic malignancies after allo-BMT, persistent MC is associated with relapse of underlying diseases or graft failure. Furthermore, when patients receive early immune modulation treatment, MC can be changed to complete donor pattern chimerism and ultimately prevent relapse.
INTRODUCTION
Allogeneic bone marrow transplantation (allo-BMT) has become an established procedure for the treatment of hematologic malignancies since it had been first performed in the early 1970’s and its indications and methods have been expanded1–3). The success of this treatment modality is mainly affected by relapse, graft rejection or graft-versus-host disease (GVHD). Factors responsible for relapse or rejection may be insufficient conditioning regimens or a deficient graft-versus-leukemia (GVL) effect eventually due to decreasing amounts of effector cells or to their functional ineffectiveness. Several studies gave evidence that the risk for relapse or graft failure was increased when mixed chimerism (MC) was detectable after allo-BMT1–4). Lawler et al5) reported that detectable MC after BMT in patients with aplastic anemia (AA) had been associated with a high relapse rate and more graft failure than achieving complete chimerism (CC), and Mackimnon et al5) reported similar findings in chronic myelogenous leukemia (CML). However, the prognostic value of predictions relapse after MC detection for patients with acute leukemias remains controversial1,6,7–12).
Various techniques, such as red blood cell (RBC) polymorphism or metaphase cytogenetic analysis, Y-chromosome specific fluorescence in-situ hybridization (FISH) and genetic polymorphism analysis using restriction fragment length polymorphism (RFLP) have been used to document chimerism after allo-BMT4,13–17). Based on these methods, preventive treatment using graft-versus-leukemia effect (GVL effect) was applied in cytogenetic-relapsing patients who showed enhancing recipient type MC after BMT. The methods for inducing GVL included early discontinuation of immunosuppressants18,19), adding the colony-stimulating factors20) or donor lymphocyte infusion (DLI)21–31). These applications are the methods that can detect early cytogenetic relapse and prevent relapse of underlying disease. That might be an influence on BMT success through the methods of detecting residual leukemic cells and immune modulation.
In this study, we introduced an early immune modulation in patients showing MC by polymerase chain reaction (PCR) - RFLP after allo-BMT and then followed up the effects.
PATIENTS AND METHODS
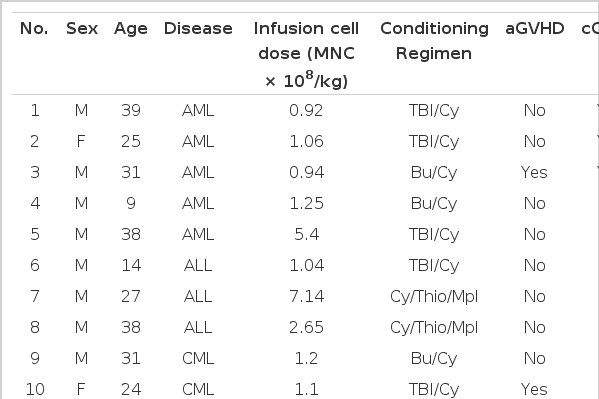
The study included 12 prospectively investigated patients with acute and chronic leukemias following allo-BMT who showed MC by PCR-RFLP and received immune modulation. Disease, age, conditioning regimen, GVHD, outcome of BMT are shown in Table 1. All patients were transplanted with bone marrow from an HLA-identical sibling donor. The transplantation procedures were performed at the Catholic Hematopoietic Stem Cell Transplantation Center (CHSTC), The Catholic University. Informed consent of patients and donors, when appropriate, were obtained according to institutional guidelines.
1. Detection of mixed chimerism
1) Specimens
Peripheral blood or bone marrow samples were collected from the donor and recipient before transplant and from the recipient at regular intervals after engraftment. Genomic DNA was isolated from fresh or frozen blood or marrow samples as described previously32). Isolated DNA was further purified using repeated GNome DNA kit (Bil 101, cat #2010-600) and then stored at 4°C after mixing with 50 μL of 1X TE solution.
2) Primers of PCR
We synthesized an YNZ 22 minisatellite primer and a 33.6 minisatellite primer based on the basic sequences by Ugozzoli et al33) and Y-microsatellite primer bringing to Bioneer company by Lo et al methods34). The sequences are as follows.
-
(1) YNZ 22 minisatellite primer
5′-GGTCGAAGAGTGAAGTGCACAG-3′(5′ primer)
5′-GCCCCATGTATCTTGTGCAGTG-3′(3′ primer)
-
(2) 33.6 minisatellite primer
5′-TGTGAGTAGAGGAGACCTCAC-3′(5′ primer)
5′-AAAGACCACAGAGTGAGGAGC-3′(3′ primer)
-
(3) Y-microsatellite primer
5′-CTACTGAGTTTCTGTTATA-3′(5′ primer)
5′-ATGGCATGTAGTGAG-3′(3′ primer)
3) PCR
After the quantitation of DNA by UV spectrophotometer, the DNA was amplified using YNZ 22 minisatellite primer in the first place. If PCR with YNZ 22 minisatellite could not distinguish the bands because of a same pattern between donor and recipient band, 33.6 minisatellite primer was used for further distinction. If the sex between donor and recipient was different, the presence of Y chromosome was also detected by Y-microsatellite primer. DNA amplification methods are the following.
In brief, 50 ng DNA was mixed with 50 ul of reacting solution containing 25 pmol of 5′ primer and 25 pmol of 3′ primer. The mixtures were denatured at 94°C for 5 minutes and then added 2.5 units of Taq DNA polymerase (BM #1146165). Each sample underwent 5 cycles of denaturation at 94°C for 1 minute, annealing at 63°C for 1 minute, and extension at 72°C for 6 minutes and then underwent further 30 cycles of denaturation at 94°C for 30 seconds, annealing at 60°C for 30 seconds, and extension at 72°C for 1 minutes. Reaction tubes received a final 5 minutes extension at 72°C.
4) Electrophoresis and detection of amplified band
5 μL of the PCR products were electrophoresed on 2% agarose gels (100 v, 20 minutes) and then stained with ethidium-bromide (0.5 ng/ml) for 20 minutes. The length of bands was identified by 100 base pair ladder marker (BM #1721933) and the bands of agarose gel on UV transilluminator were photographed by polaroid camera. Cell separation and DNA extraction and amplification could be performed on the same day and gel electrophoresis, hybridization, and exposure on the day after.
5) Definition of MC and CC
After engraftment, chimeric status was determined by RFLP-PCR at post BMT 1 month. Patients who showed no evidence of recipient cells and had only donor cells after 3 months post BMT were considered to be complete chimerism (CC)35,36). Patients who showed both recipient and donor cells in the peripheral blood and/or bone marrow after 3 months post BMT were defined as mixed chimerism (MC)35,36). When mixed chimerism was detected one time during 1 to 3 months post BMT, it was described as a transient MC35,36).
2. Follow-up and immune modulation
All patients who achieved engraftment were examined for chimeric status at 1 month post BMT. After discharge, the majority of the patient samples were collected and analyzed every 3 months within an observation period of 18 months after allo-BMT. We suggested that MC enhanced to recipient type (residual disease) if the recipient band was progressively amplified, such as a more strong band at 3 months post BMT, and additional laboratory data supported this MC status; first, the quantitative titer of anti-A or anti-B against recipient RBC increased or sustained if the RBC type of donor and recipient was different and its titer could be followed: second, the evidence of predictable hematologic recovery, including platelet count enhancement, was not found or decreased. And if graft-versus-host disease (GVHD) was not developed in addition to the above we introduced an early immune modulation (tapered or discontinued immunosuppressant). If the MC enhanced to recipient type or persisted in suspecting relapse high risk group in spite of an early immune modulation, additional donor lymphocytes were infused. Donor lymphocyte infusion (DLI) consisted of G-CSF mobilized peripheral stem cells, including appropriate lymphocyte counts for overcoming marrow failure due to DLI, or appropriate lymphocytes (5 × 107 CD3+/kg, progressively increment of lymphocytes) alone. In some instances, injection of Interferon-α (3 million units s.c., 3 times/week) developed as an immune modulation. The RFLP-PCR for MC was performed at every 3 month interval.
RESULTS
1. The frequency of MC
From March 1996 to August 1998, of 337 patients who received allo-BMT for various hematologic malignancies at CHSTC, 27 patients revealed consecutive MC more than twice (1 and 3 months post BMT). Twelve of 27 patients who could be followed and who received immune modulation enrolled in this study. The underlying diseases consisted of 5 AML (4 in first complete remission (CR), 1 in early relapse after CR), 4 CML (3 in chronic phase (CP), 1 in second CP), and 3 acute lymphocytic leukemia (ALL) (2 in CR, 1 in reinduction failure). Nine were male and three were female. The conditioning regimens were total body irradiation (TBI, 12 Gy) and cyclophosphamide (Cy, 120 mg/kg), busulfan (Bu, 16 mg/kg) and Cy (120 mg/kg), and Cy (100 mg/kg), thiotepa (500 mg/m3), and melphalan (100 mg/m3) (Table 1). Three recipients were infused with manupulated bone marrow stem cells that were expanded in the donor by G-CSF (10 ug/kg, s.c.) for 3 days prior to BMT. GVHD prophylaxis consisted of cyclosporine 3 mg/kg/day continuous i.v. on days 0 to +20 or until the patient was able to take it orally; and then 2 mg/kg orally every 8 hours until day +100 followed by a 5% decrease per week. Methotrexate was also used 10 mg/m3 i.v. on days +1, +3, +6, and +11. All patients with Bu/Cy regimen received phenytoin (5 mg/kg/day) beginning 24 h before the first dose of Bu and continuing until 24 h after last dose to prevent seizure.
2. The response of immune modulation
Nine of the 12 patients changed MC to CC after immune modulation, including early dose reduction/discontinuation of immunosuppressant. Five of the 9 patients revealed CC at the next follow-up examination after immune modulation and the remainder of the patients progressively changed to donor type, and then ultimately to CC. The diseases that responded to immune modulation and then achieved CC were the following; 5 of 5 AML, 3 of 4 CML and 1 of 3 ALL.
The patients who could not be changed to CC by immune modulation had diseases with high risk, including relapse or rejection; second CP in CML or primary refractory ALL. Those could not respond to immune modulation and their diseases were relapsed or infused grafts were rejected.
3. Development of GVHD
Four patients experienced GVHD during or after an immune modulation but the grade of GVHD was minimal (grade ≤ 2). Acute GVHD (aGVHD) occurred in 2 patients and chronic GVHD (cGVHD) occurred in 3 patients. One of them had transformed GVHD from aGVHD to cGVHD. But all patients with GVHD responded to temporary steroid treatment and achieved CC.
4. Clinical courses
The disease status at transplant, change pattern of MC, immune modulation and its response and the outcomes were the following (Figure 1).
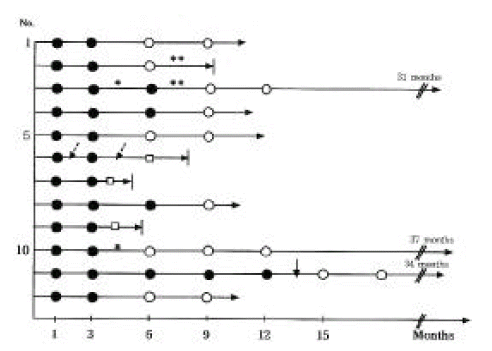
The courses of those patients with mixed chimerism are shown. Black circles indicate state of mixed chimerism. White circles indicate state of complete donor pattern chimerism. The small white boxes indicate state of hematologic relapse. Once mixed chimerism was detected, immunosuppressive therapy (cyclosporine A) was discontinued. No. 11 patient had been mixed chimerism until start of the interferon-α subcutaneous injection (↓). No. 1–5: AML, 6–8: ALL, 9–12: CML, *; acute GVHD,**; chronic GVHD, ↙; PBSCT with donor lymphocyte infusion, →|; death
1) AML
Patients number 1 to 5 were AML. Number 1 patient revealed MC at 1 month post BMT and the MC progressively amplified to recipient type. He was introduced to rapid tapering (reduced 2 times higher than usual rate) and discontinuation of immunosuppressant. He revealed donor type CC at 6 months post BMT examination. He experienced temporary cGVHD in the liver during immune modulation but responded to steroid treatment. He is now well without any relapse evidence in periodic follow-up examinations. Number 2 to 5 patients with MC also achieved CC after immune modulation and they are now well except number 2 patient.
2) ALL
Patients number 6 to 8 were ALL. Number 6 patient, who had a delayed engraftment, received GCSF mobilized peripheral blood stem cells (PBSC) because of hypocellular marrow with MC at 1 month post BMT examination. Due to persisting MC at 3 months post BMT examination, he received second G-CSF mobilized PBSC, including counted lymphocytes for stem cell support and GVL effect. However, hematologic relapse occurred at 6 months post BMT and he died at 8 months post BMT. Patient number 7 was were refractory status at transplant in spite of previous induction chemotherapy. He revealed MC after engraftment and MC enhanced to recipient type after immune modulation. At 5 months post BMT, he had a hematologic relapse. Patient number 8 achieved CC by immune modulation after detecting MC and he is now well without detectable disease evidence.
3) CML
Patients number 9 to 12 were CML. Number 9 patient with second CP in CML was introduced to an early immune modulation just after detecting initial MC. He was recommended for DLI but he refused and for soon relapsed. Patients number 10 and 12 achieved CC by immunosuppressant tapering after detecting MC and they are now well. At 12 months post BMT, patient number 11 maintained stable MC in spite of early discontinuation of immunosuppressants. She received interferon-α (3 million s.c., three times per week) from 13 months post BMT and achieved CC after that. Interferon treatment continued for 6 months and then stopped. She is now well without any disease evidence at 34 months post BMT.
DISCUSSION
Allo-BMT is a curative treatment modality for various hematologic malignancies, including acute and chronic leukemias and AA, but there has not been established a curable treatment modality if relapse occurred after transplantation. DLI that was first introduced in CML and approved its effectiveness is the most effective in cytogenetic relapse21). Unfortunately, DLI has little effect on other diseases, especially a troublesome one such as GVHD. For inducing GVL effect, many methods (immune modulations; control of immunosuppressant, cytokines, growth factors and DLI) have been applied but those revealed their effect in minimal residual disease (MRD) status18–20). Therefore, early detection of MRD suggests an important factor for successful maintenance of transplantation by immune modulations. The clinical significance of MC for the prediction of relapse after allo-BMT has been investigated over the last decade4–15). Some investigators described patients with high levels of residual and/or rapidly increasing amounts of recipient cells to carry a high risk of developing relapse4,36–39), while others found no correlation between MC and relapse1,6,40–41). The associations with immune tolerance and reduction of GVHD were also described41). According to the development of MRD detecting methods and proportional assessment of quantitation between donor and recipient4,38–39,41), relapse prevention has become possible by early immune modulation at detecting the evidence of early relapse. Bader et al36) described that immune modulation, including early discontinuation of immunosuppressant and DLI, prevented relapse, GVHD associated with immune modulation was treatable, and GVL effect and immune tolerance might exist.
Results presented in this study demonstrated that patients who showed MC following engraftment converted to CC and remained in continuous remission by early immune modulation. GVHD which was developed during immune modulation was controllable and might be helpful in converting to CC. The importance of the immunological response after allo-BMT might be substantiated by our finding that no patient with MC developed severe acute or chronic GVHD which is considered to be associated with the GVL effect25). Therefore, we suggested that MC influenced the disease course of recipient and immune tolerance according to proportional changes between donor and recipient, not its presence. On the other hand, early tapering and discontinuation of immunosuppressant and interferon treatment might be helpful in achieving CC in CML. DLI which was known as effective in CML could not find effectiveness in ALL. The pre-BMT disease status was an important factor of BMT success and further immune modulation on the basis of the above results show that early immune modulation was not helpful in refractory ALL and second CP in CML. The change of CC by immune modulation was more effective in acute and chronic myelogenous leukemia which responded well to immune modulation (GVL effect). Infused stem cell dose was not associated with MC formation after BMT and relapse.
On the above results, the detection and quantitation of MC may be important factors for MRD detection in addition to beings possible other markers for assessment of disease status after BMT. Therefore, their follow-up and relapse prevention before hematologic relapse by immune modulation will be needed for successful transplantation. A few patients number and short follow-up period were a limitation in this study for result analysis. Future studies, including many enrolled cases, and longer follow-up periods will be needed for certifying our results.
Acknowledgements
We would like to thank Kwang-Sung Kim, chief nurse & coordinator, and nursing members of the Catholic Hematopoietic Stem Cell Transplantation Center for excellent patient care and monitoring for the study. This work was supported by a grant from the St. Mary’s Hospital Research Fund of the Catholic University, Korea.