 |
 |
| Korean J Intern Med > Volume 41(2); 2026 > Article |
|
Abstract
Syphilis is a sexually transmitted disease caused by the spirochete Treponema pallidum. Recently, there has been a global resurgence of syphilis cases. In South Korea, the reported rate of syphilis steadily increased from 1.93 per 100,000 individuals in 2011 to 3.41 per 100,000 individuals in 2019. Notably, the incidence of the disease has been rising sharply among men in their 20s and 30s since 2016. Diagnosing syphilis based on its stage is complex and time-consuming. The combination of nontreponemal and treponemal tests has long been the cornerstone of syphilis diagnosis. Automated versions of nontreponemal tests and treponemal immunoassays have replaced the traditional manual nontreponemal and treponemal tests. Additionally, traditional direct methods, such as dark-field microscopy and silver staining, have been replaced by polymerase chain reaction testing and immunohistochemistry. Point-of-care tests are promising tools that facilitate the rapid and easy diagnosis of active syphilis. To ensure the proper diagnosis and control of syphilis, a comprehensive surveillance system and a thorough understanding and appropriate interpretation of diagnostic tests are necessary.
Syphilis is a sexually transmitted infection (STI) caused by the bacterium Treponema pallidum subspecies pallidum. The global incident cases of syphilis increased from 8.8 million in 1990 to 14.1 million in 2019 [1]. The World Health Organization (WHO) estimated that 8 million incident cases of active syphilis were acquired by individuals aged 15–49 years in 2022 [2]. Clinical presentation of syphilis varies by stage. Primary syphilis typically presents as a single painless ulcer (chancre) at the site of inoculation. In contrast, secondary syphilis presented systemic features, including skin rash, mucocutaneous lesions, and lymphadenopathy [3]. Tertiary syphilis can present with cardiovascular involvement, gummatous lesions, general paresis, and tabes dorsalis [3]. Asymptomatic latent infections are frequently detected by serological testing during health checkups or preoperative blood tests. Asymptomatic syphilis within one year of infection is referred to as early latent syphilis, whereas infections beyond one year are classified as late latent syphilis [3].
Syphilis is primarily diagnosed based on serological tests. This method has been used for over 100 years since it was first reported by Wassermann et al. in 1906 [4]. Recent updates in diagnostic testing include increased automation, the shift from traditional to reverse algorithms, and the potential for point-of-care (POC) testing [5–7]. Traditional direct detection methods have been replaced by newer technologies. Specifically, dark-field microscopy and silver staining methods have been replaced by polymerase chain reaction (PCR) testing and immunohistochemistry [5–8]. Understanding and correctly interpreting recently used diagnostic tests is crucial for optimal management. Here, we review the changes in the epidemiology of syphilis in South Korea and major developed regions, such as the United States, Japan, and Europe, as well as recent advancements in syphilis diagnostic techniques. The diagnosis of congenital syphilis and neurosyphilis is not discussed here.
In South Korea, the syphilis surveillance system monitors highly contagious primary and secondary syphilis cases, as well as congenital syphilis—a major global cause of fetal and neonatal complications [9]. The system has undergone several changes that complicate the assessment of long-term trends. It operated as a sentinel surveillance system from 2001 to 2010, shifted to mandatory reporting from 2011 to 2019, and reverted to a sentinel system from 2020 to 2023. In 2024, it returned to mandatory reporting because of the substantial psychological and economic burden of syphilis on the public and rising syphilis trends in neighboring countries such as Japan, China, and Taiwan [9]. Additionally, in the post–COVID-19 period, there has been a potential risk of syphilis importation from these countries, underscoring the need for enhanced surveillance. The reporting scope has expanded from three categories—primary, secondary, and congenital syphilis—to five, now including early latent syphilis, which is transmissible, and tertiary syphilis, which can cause severe disease [9].
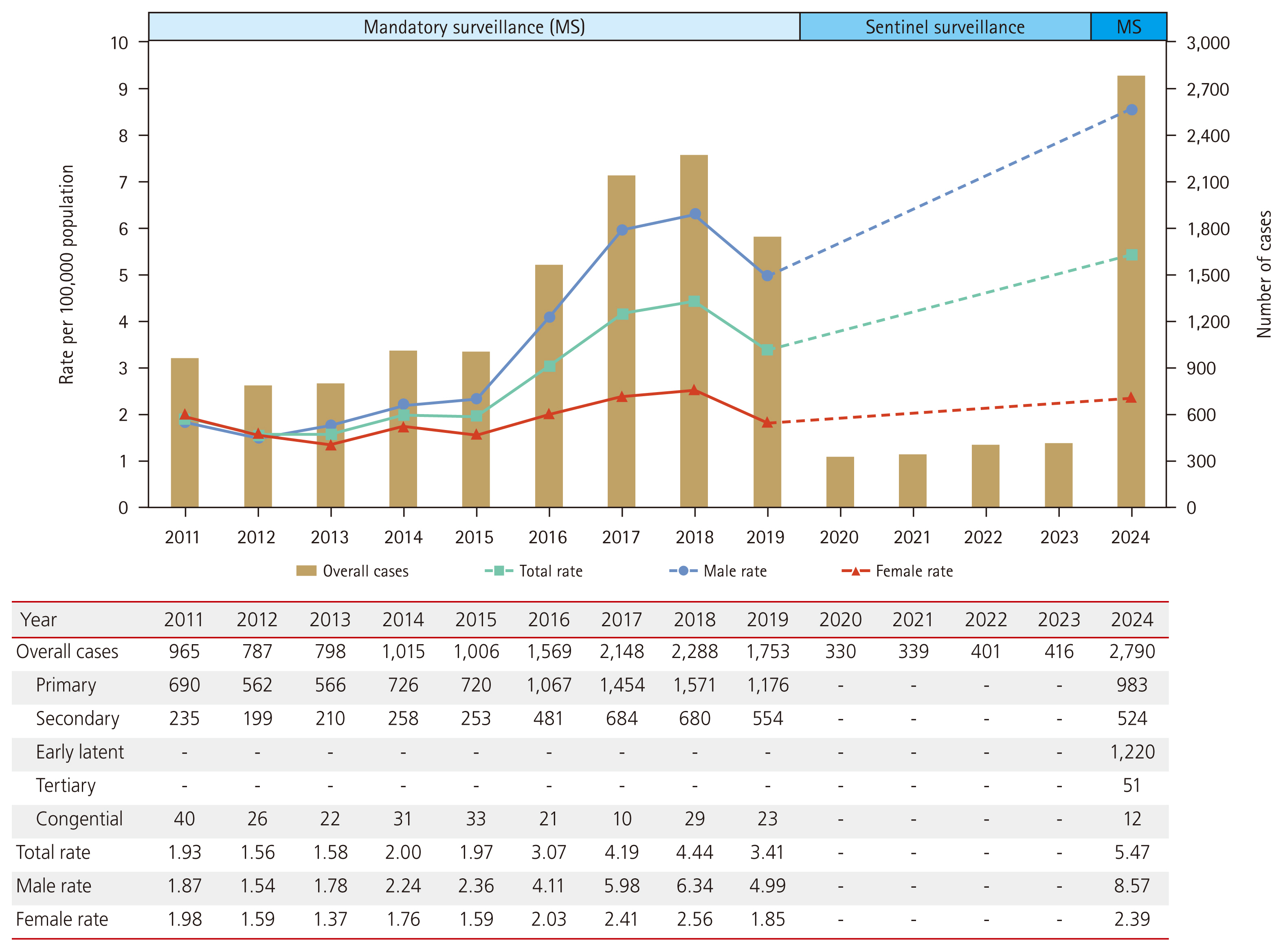
Figure 1 illustrates the reported cases and incidence rates of syphilis between 2011 and 2024. This figure was created using data on reported syphilis cases obtained from the Infectious Diseases Surveillance Yearbook [10–14]. The incidence rate is calculated by dividing the number of reported cases by the mid-year population, which is obtained from the resident registration data provided by the National Statistical Office [15]. Between 2011 and 2019, the overall rate increased from 1.93 to 3.41 cases per 100,000 persons. Notably, since 2016, a sharp increase has been observed among males in their 20s and 30s; however, this trend has not been evident among females (Fig. 2). Individuals with HIV–syphilis coinfection may account for a substantial proportion of syphilis cases. Using the National Health Insurance claims database, one study reported a steady increase in the prevalence of HIV infection from 2008 to 2016. In that cohort, 49.8% (4,278/8,592) of men and 32.2% (258/801) of women living with HIV were co-infected with syphilis [16].
The incidence rates of syphilis from 2020 to 2023 are difficult to compare directly with those of previous periods, owing to changes in the surveillance system. Despite these limitations, the number of reported cases increased steadily during this period, rising from 330 in 2020 to 416 in 2023 (Fig. 1). In 2024, 2,790 cases of syphilis were reported, with 2,177 cases in males (78.0%) and 613 cases in females (22.0%). These 2,790 cases comprised 983 primary, 524 secondary, 51 tertiary, 1,220 early latent, and 12 congenital syphilis cases. The significant increase in 2024 might be influenced by the transition to mandatory surveillance, the expansion of the scope of reportable diseases, and the end of the COVID-19 pandemic. Additionally, all positive test results are now mandatorily reported to public health centers, facilitating the identification of previously missed cases and likely contributing to a reduction in underreporting. Congenital syphilis demonstrated a declining trend from 40 cases in 2011 to 12 cases in 2024.
Japan experienced a substantial increase in the syphilis incidence rate from 2011 to 2023, increasing 18.7-fold from 0.65 to 12.11 cases per 100,000 individuals (Fig. 3A) [17,18]. From 2011 to 2023, syphilis incidence rate surged 15.4-fold in men (from 1.04 to 16.05 per 100,000) and 31.0-fold in women (from 0.27 to 8.37 per 100,000), indicating a steeper rise in women than in men. The increase in female cases was also accompanied by an increase in congenital syphilis cases—from six in 2011 to 37 in 2023 [19,20]. Among male patients, the proportions of men who have sex with men (MSM) and men who have sex with women only (MSW) were similar before 2015. However, MSW surpassed MSM in 2015 and increased steadily, accounting for 82.6% of male cases with known transmission routes in 2023 [19,20].
In the United States, the incidence of primary and secondary syphilis increased from 4.5 to 15.8 per 100,000 individuals between 2011 and 2023 (Fig. 3B) [21]. Similar to the trends in Japan, this increase was observed in men and women, with a more pronounced increase among women. During the same period, the incidence rate surged 2.9-fold in men (from 8.1 to 23.6 per 100,000 individuals) and 9.0-fold in women (from 0.9 to 8.1 per 100,000 individuals) [21]. From 2014 to 2023, the proportion of MSM among primary and secondary syphilis cases decreased from 61.1% to 32.7%, while the proportion of MSW increased from 12.6% to 24.2%, and the proportion of women increased from 9.2% to 26.0% [21]. The sharp increase in female cases coincided with a 9.1-fold increase in congenital syphilis, from 11.6 to 105.8 per 100,000 live births between 2014 and 2023 [21].
According to data from the European Union (EU) and the European Economic Area (EEA), the syphilis incidence rate increased steadily from 4.6 per 100,000 in 2011 to 9.9 per 100,000 in 2023 (Fig. 3C) [22–30]. The decrease in 2020 was most likely due to the impact of the COVID-19 pandemic on high-risk populations and the reduced accessibility of STI services, as medical resources were diverted to the COVID-19 response. Data for the United Kingdom have not been included since 2020 because of its withdrawal from the EU [28]. The overall male-to-female ratio in 2023 was 7.4:1, with rates of 17.6 cases per 100,000 individuals in men and 2.4 cases per 100,000 individuals in women. Unlike in Japan and the United States, most (72%) syphilis cases with known transmission routes were reported among MSM [30].
Nontreponemal serological tests remain the primary screening method in South Korea. These tests are based on the reactivity of serum from infected patients to lipoidal antigens containing cardiolipin, lecithin, and cholesterol, which are released by T. pallidum and damaged host cells [31]. Nontreponemal serological tests include the Venereal Disease Research Laboratory (VDRL) test, rapid plasma reagin (RPR) card test, Unheated Serum Reagin (USR) test, and Toluidine Red Unheated Serum Test (TRUST). Nontreponemal tests have traditionally been used for initial syphilis screening because they are simple, rapid, and inexpensive. They are also used to evaluate the response to treatment because they are the only tests reported as quantitative values, a titer of antibody. An increase or decrease in titer (e.g., from 1:8 to 1:32 or from 1:16 to 1:4) between two serial samples from the same patient is considered clinically significant [32]. Both tests should use the same serological method and manufacturer to ensure comparable results [6]. Titers decrease more rapidly after successful treatment but also gradually wane over time, even without treatment. These tests exhibit low sensitivity for primary and late syphilis. Biological false positives are not common (1% to 2%) but can occur in various situations (Table 1) [7,8].
Hospitals and institutions handling excessive specimens face challenges owing to the labor-intensive nature of manual RPR tests. In South Korea, automated RPR tests have increasingly replaced traditional manual RPR card tests for syphilis screening. For optimal clinical interpretation, non-treponemal tests should be reported as quantitative values or RPR titers. However, certain automated RPR tests have a narrow range of detectable titers (e.g., 1:40–1:64) and cannot provide titers beyond this range [6]. In such cases, manual RPR card tests are required to establish an endpoint titer at the lower or upper bounds. Among automated RPR tests, the latex turbidimetric immunoassay (LTIA) is commonly used in South Korea, with notable examples being Mediace RPR (Sekisui Chemical Co., Ltd., Tokyo, Japan) and Hisens Auto RPR LTIA (HBi Co., Ltd., Anyang, Korea) [33]. Automated RPR tests often produce false-negative results at low RPR titers, and discrepancies can exist between RPR titers obtained from automated and manual tests. In a recent study comparing the manual RPR card test with the automated Mediace RPR test, 48 out of 190 samples (25%) that were positive manually were negative in the automated test [34]. All false negatives in the automated Mediace RPR test occurred within the manual RPR test range of 1:1–1:8 [34]. The probability of the RPR titers from both tests matching within a 2-fold dilution was 54% [34]. Compared to the manual RPR, which has been used for decades, research on the utility of automated RPR tests for assessing treatment responses is relatively limited. In one study on automated RPR tests, the serological response (a 4-fold or greater decrease in RPR titer) after one year of treatment was 77% and 79% for automated and manual tests, respectively [35]. Clinicians should be aware of whether the test results for patients are from manual or automated RPR tests. To evaluate patient treatment, the nontreponemal antibody titer should be determined using the same type of test. Until more data are gathered regarding the utility of automated RPR tests in assessing treatment responses, we suggest using a manual RPR test.
Treponemal tests detect antibodies directed against treponemal antigens and are generally more specific than non-treponemal tests, making them traditionally suitable as confirmatory tests for syphilis when non-treponemal tests are reactive [5]. In the absence of immunosuppression, a nonreactive treponemal test generally indicates no past or present infection. As the antibody titer in treponemal tests does not correlate with disease activity, these tests should not be used to evaluate treatment response or assess reinfection or relapse. Antibodies become detectable 2–4 weeks after exposure, making treponemal tests more sensitive in early infection compared to nontreponemal tests, which may not be reactive at this stage [31]. In immunocompetent patients, the antibodies detected in treponemal tests typically remain reactive for life, even after treatment. However, some individuals treated for primary syphilis may become seronegative after two to three years [36,37].
Manual treponemal tests include the fluorescent antibody- absorption (FTA-ABS) test, T. pallidum hemagglutination assay (TPHA), microhemagglutination assay for T. pallidum (MHA-TP), and T. pallidum particle agglutination (TPPA) test [6,7,31,38]. The FTA-ABS test is an indirect immunofluorescent assay that uses slides fixed with an extract of T. pallidum. The TPHA and MHA-TP are indirect hemagglutination assays that utilize animal erythrocytes coated with T. pallidum antigen, which agglutinate with antitreponemal antibodies [6,7,31,38]. The TPPA test has replaced the MHA-TP and TPHA tests owing to its superior sensitivity and comparable specificity to the FTA-ABS test [31]. Historically, manual treponemal tests have been more labor-intensive and expensive to perform than manual nontreponemal tests. Newer automated treponemal immunoassays, such as enzyme immunoassay (EIA) and chemiluminescence immunoassay (CIA), are less labor-intensive and more efficient, with rapid turnaround times [5]. Consequently, automated treponemal tests have been increasingly used instead of traditional manual treponemal tests. Most studies demonstrate a 91–100% correlation between treponemal immunoassay positivity and treponemal antibody positivity [39–44]. The T. pallidum latex agglutination (TPLA) method, which coats T. pallidum antigens onto latex particles and allows automated analysis, is used extensively in Japan and South Korea [45]. In South Korea, the use of TPPA has been decreasing, whereas that of automation-ready assays (CIA and TPLA) has been increasing [33,46]. In 2023, a survey of 231 institutions using treponemal tests revealed that CIA and TPLA accounted for 26.1% and 20.6%, respectively, whereas TPPA and FTA-ABS accounted for 19.0% and 4.7%, respectively. The immunochromatographic assays are widely used for POC testing and accounted for 27.3% of all treponemal tests [33].
Numerous POC syphilis tests are used globally in clinical and nonclinical environments [47–54]. Most of these tests are based on immunochromatography, allowing for visual confirmation of results within minutes in the field without requiring special equipment. However, they detect treponemal antibodies and cannot distinguish between active and prior infections. Some POC tests can be used to test for syphilis and HIV simultaneously [53,54]. The POC syphilis tests approved by the US FDA are considerably limited, and research on their performance has primarily focused on these approved tests [54,55]. One study showed that the DualPath Platform (DPP) HIV-Syphilis Assay (Chembio Diagnostic Systems, Inc., Medford, NY, USA) demonstrated a sensitivity and specificity of 98.7% and 100%, respectively [54]. In a meta-analysis of the Syphilis Health Check (Trinity Biotech, Bray, Ireland), which includes data from 10 prospective studies, the pooled sensitivity and specificity were 87.7% and 96.7%, respectively [55].
Direct methods can be used to provide a definitive diagnosis of syphilis. As T. pallidum cannot be cultured in the laboratory, the organism must be identified through direct visualization or detection in clinical specimens. Although dark-field microscopy and direct fluorescent antibody testing can be used to detect the organism, they are not commonly used in clinical settings.
PCR tests, which are based on detecting various DNA target sequences, are more commonly used. Most laboratory- developed PCR tests are based on the tp47 (tp074) or polA (tp0105) genes, with varying sensitivities depending on the stage of syphilis and specimen type [6]. The sensitivities have been reported as 72–95% for the exudate of primary syphilis lesions (i.e., chancres) collected by swab, whereas lower sensitivities of 20–86% have been reported for the exudate of secondary syphilis lesions [56]. In general, the sensitivity of PCR tests is higher than that of dark-field microscopy. In a European study comparing the two methods for diagnosing primary syphilis, the sensitivity was 87.5% for PCR and 66.7% for dark-field microscopy [57]. PCR tests show lower sensitivity when performed on blood (15–24%) than on mucosal specimens (82%) [58]. Recently, Aung et al. performed PCR using mucosal specimens among asymptomatic contacts of individuals with syphilis, suggesting that it may be useful for early syphilis screening [59]. Notably, PCR tests amplify living and dead organisms.
The traditional serological testing algorithm involves the use of a nontreponemal test (e.g., VDRL and RPR) for screening and, if reactive, confirmation with a treponemal test (Fig. 3). Confirmation using a treponemal test is necessary because of the possibility of false-positive nontreponemal tests associated with various diseases or conditions (Table 1). A combination of positive nontreponemal and treponemal test results usually indicates a new infection requiring treatment (Table 2). In asymptomatic individuals, additional testing is generally unnecessary if the initial nontreponemal test result is negative. For patients with signs and symptoms of early syphilis (e.g., ulcers and rashes), false-negative reactions should be considered. False-negative tests can occur if testing is performed too early during the antibody formation process. Approximately 20–30% of patients presenting with a chancre have been reported to have a nonreactive nontreponemal test for syphilis [60,61]. False negatives can also occur because of the prozone effect in high-titer situations, such as in secondary syphilis. Prozone reactions occur because of the interference from high concentrations of antibodies in a specimen. If secondary syphilis is suspected, but the nontreponemal test is nonreactive, clinicians should ask the laboratory about the possibility of the prozone effect and confirm the results through sample dilution [6]. Many hospitals in South Korea use automated RPR tests, which can process numerous specimens at a relatively low cost. The traditional algorithm has been validated using the manual qualitative VDRL and RPR tests. Research on the diagnostic performance of initiating this algorithm with the automated RPR, predominantly used in South Korea, remains limited.
The reverse algorithm was first proposed by the United States Centers for Disease Control and Prevention (CDC) in 2008 [62]. This method uses an automated treponemal test (e.g., EIA or CIA) as the initial screening test, followed by a nontreponemal test if the treponemal test is reactive (Fig. 3) [63]. The reverse screening method resulted in more false positives than the traditional approach. However, it can detect syphilis in some additional patients with early, previously treated, and latent syphilis [64–66]. In low-prevalence populations, such as in the United States, where the syphilis seroprevalence was reported to be 0.71% in 2008, the positive predictive value of automated treoponemal tests ranges from 12% to 45%. However, this value can reach 90% as the prevalence of syphilis or the level of clinical suspicion increases in the population being tested [60,67]. In a Korean study evaluating 24,681 samples using the automated RPR test and automated Mediace TPLA test, reverse screening detected more cases of syphilis (0.07% vs. 0.05%) but showed more false-positive results (0.64% vs. 0.13%) [68]. The automated Mediace TPLA test, used in South Korea and Japan, has sensitivity and specificity comparable to or slightly lower than those of the automated treponemal immunoassay [45,69]. In South Korea, automated treponemal tests are substantially more expensive than automated RPR. Therefore, further research is necessary to determine whether implementing the reverse algorithm is cost-effective for syphilis screening and control.
For patients without a history of syphilis, these results usually indicate a new infection requiring treatment (Table 2). Although treponemal and nontreponemal tests can yield false positives, this occurrence is extremely rare (Table 1). For patients with a history of treated syphilis, a positive nontreponemal test result may indicate an evolving response to treatment, reinfection, or relapse [31,32,70]. To differentiate between these possibilities accurately, the current nontreponemal titers should be compared with the titer at the time of treatment using the same test (VDRL or RPR), ideally in the same laboratory. After syphilis treatment, nontreponemal test titers usually decrease at least fourfold and typically revert to nonreactive titers within 12–24 months of the follow-up period. However, some patients may have non-treponemal titers that decline ≥ 4-fold but do not become nonreactive [6,32]. This state is often referred to as the serofast state and is most common in patients with late syphilis and those with multiple episodes of syphilis [6,71,72]. In such situations, retreatment is usually not required unless there is evidence of reinfection or relapse. A fourfold or greater increase in titer following an adequate treatment response suggests reinfection or relapse [31,32,70]. Even without prior titer information, the presence of clinical signs or symptoms consistent with primary or secondary syphilis (e.g., ulcer and rash) should prompt consideration of reinfection.
The most common cause of a combination of positive nontreponemal and negative treponemal test results is a false-positive nontreponemal test (Table 2). Although the rate of false-positive reactions is generally low (1–2%), they can be related to numerous causes (Table 1) [7,8]. Pinta and yaws are chronic tropical skin diseases caused by Treponema carateum and T. pallidum subspecies pertenue, respectively, which can produce false-positive results [73,74]. Acute false-positive reactions last less than six months and can be associated with acute infectious diseases, such as hepatitis, infectious mononucleosis, viral pneumonia, chickenpox, measles, other viral infections, or malaria. They may also occur after recent immunizations and during pregnancy [8]. Chronic false-positive reactions persist for more than six months and may be caused by various factors, such as autoimmune disorders (including systemic lupus erythematosus), immunoglobulin abnormalities, narcotic addiction, aging, leprosy, and malignancies [8]. False positives generally occur at low titers (≤ 1:8) but can also happen at high nontreponemal titers (> 1:8), complicating the differentiation from true positives [8].
A combination of a positive treponemal test and a negative nontreponemal test can occur during initial screening with a treponemal test, as illustrated in the reverse algorithm in Figure 3. In South Korea, these results can be obtained when both tests are performed simultaneously. For patients with a history of treated syphilis, this discordant result reflects an evolving response post-treatment and does not require additional treatment. For patients without a history of syphilis treatment, these results may appear in the early or late stages of syphilis (Table 2). If clinical manifestations suggest primary or secondary syphilis, such as a chancre or rash, the nontreponemal test should be repeated to check for reactivity, and presumptive treatment should be initiated. In the absence of signs or symptoms indicative of syphilis, the possibility of late latent syphilis should be considered. In these cases, a different treponemal test than the one initially performed should be conducted. For example, if a CIA test was initially performed, an FTA-ABS or TPPA test can be conducted (Fig. 3). If the second treponemal test result is positive, treatment for late latent syphilis should be administered. If the second treponemal test is negative, the first treponemal test may have been a false positive (Table 1). In a study conducted in low-prevalence settings, most patients with a positive CIA test followed by negative RPR and TPPA results had an initial treponemal CIA test that was a false positive [40].
The recent resurgence of syphilis in South Korea underscores the need to implement effective diagnostic methods and algorithms tailored to the Korean context. With the increasing incidence of syphilis among sexually active men, more targeted control and behavioral interventions may be essential. This increase could potentially expand to sexually active women, as observed in Japan and the United States, necessitating the proactive development of control strategies. Recent advancements in serological testing, including increased automation, POC testing, and a shift from traditional methods to reverse algorithms, have been significant. Despite these technological advancements, further research is necessary to evaluate the effectiveness of these tests in screening, diagnosing, and controlling the disease in South Korea.
Notes
Figure 2
Age-specific syphilis incidence per 100,000 among males and females in South Korea, 2011–2024.

Figure 3
Total syphilis case counts and incidence rates in Japan (A), the United States (B), and European countries (C), 2011–2023. The total syphilis case count includes primary, secondary, latent, tertiary, and congenital syphilis in Japan; primary and secondary syphilis in the United States; and primary, secondary, and early latent syphilis in Europe.
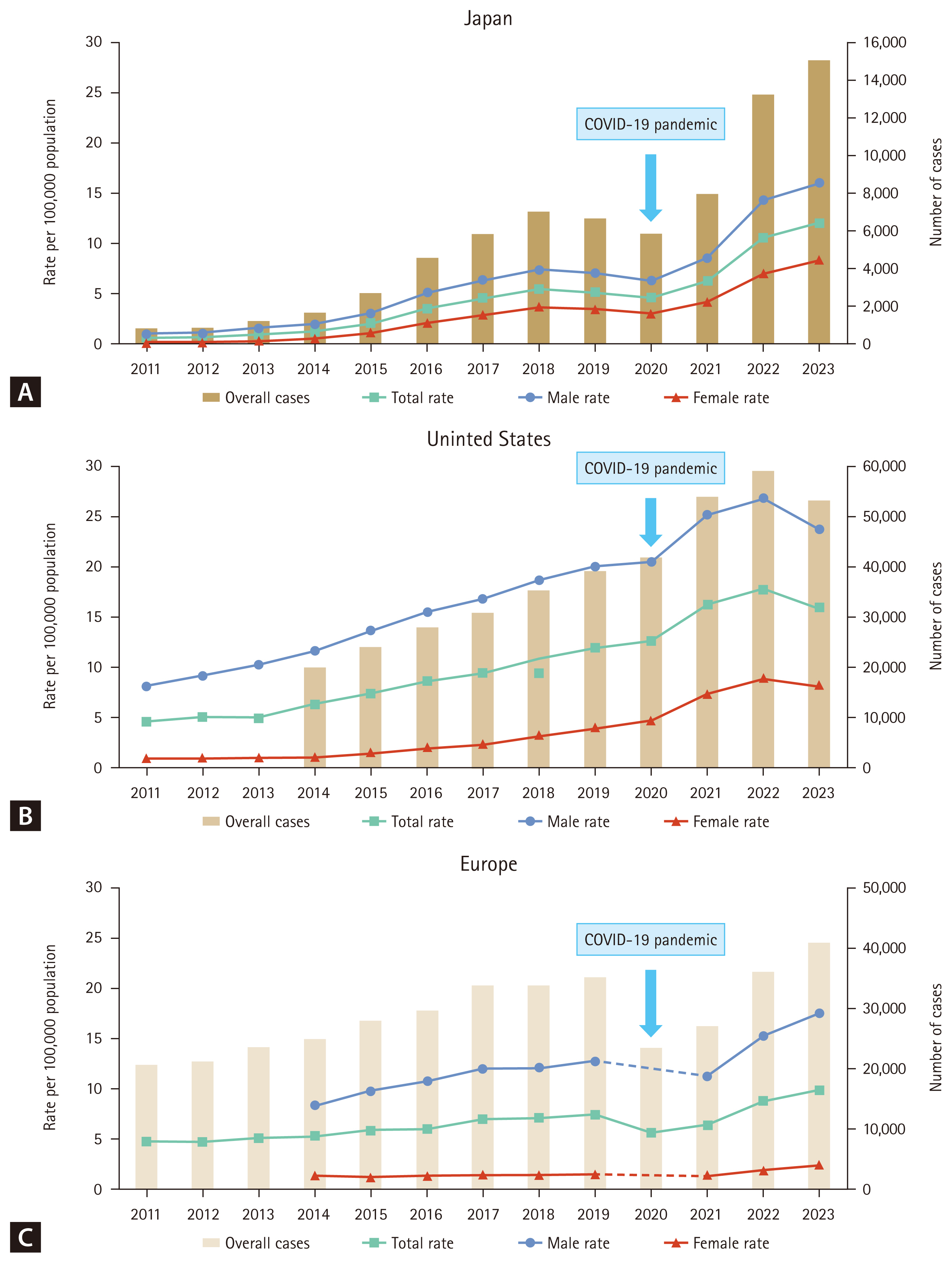
Figure 4
Serological screening algorithms for syphilis, as recommended by the U.S. Centers for Disease Control and Prevention (CDC) [6]. RPR, rapid plasma regain; VDRL, Venereal Disease Research Laboratory; EIA, enzyme immunoassay; CIA, chemiluminescence immunoassay; TPPA, Treponoma pallidum particle agglutination.
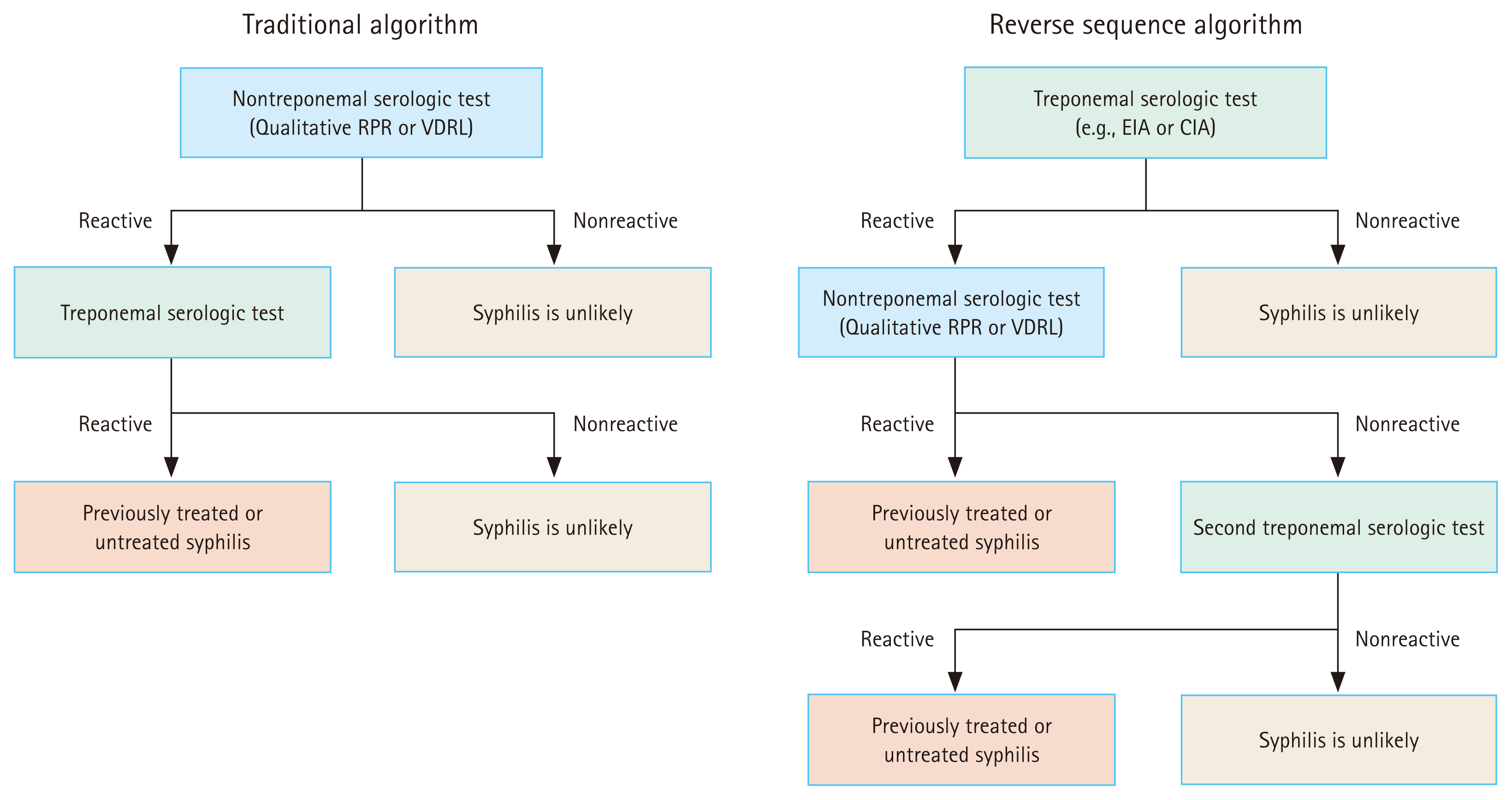
Table 1
| Nontreponemal tests | Treponemal tests |
|---|---|
| Advancing age | Advancing age |
| Autoimmune diseases | Autoimmune diseases |
| Rheumatoid arthritis | Scleroderma |
| Polyarteritis nodosa | Systemic lupus erythematosus |
| Systemic lupus erythematosus | Thyroiditis |
| Thyroiditis | Cirrhosis |
| Ulcerative colitis | Drug addiction |
| Idiopathic thrombocytopenic purpura | Hyperglobulinemia |
| Drug addiction | Immunizations |
| Immunizations | Infections |
| Immunoglobulin abnormalities | Brucellosis |
| Infections | Genital herpes |
| Bacterial endocarditis | Infectious mononucleosis |
| Brucellosis | Leptospirosis |
| Chancroid | Leprosy |
| Chickenpox | Lyme disease |
| Hepatitis | Malaria |
| Infectious mononucleosis | Relapsing fever |
| Leprosy | Yawsa) |
| Lymphogranuloma venereum | Pintaa) |
| Measles | Pregnancy |
| Mumps | |
| Rickettsial disease | |
| Tuberculosis | |
| Pintaa) | |
| Pneumococcal pneumonia | |
| Viral pneumonia | |
| Yawsa) | |
| Malignancy | |
| Pregnancy |
REFERENCES
1. Zheng Y, Yu Q, Lin Y, et al. Global burden and trends of sexually transmitted infections from 1990 to 2019: an observational trend study. Lancet Infect Dis 2022;22:541–551.


2. World Health Organization (WHO). STI: incident cases of syphilis in 15-49 year olds (in thousands) [Internet] Geneva: WHO, 2024. [cited 2025 Jul 3]. Available from: https://www.who.int/data/gho/data/indicators/indicator-details/GHO/incident-cases-of-syphilis-in-individuals-(in-millions).
3. Golden MR, Marra CM, Holmes KK. Update on syphilis: resurgence of an old problem. JAMA 2003;290:1510–1514.


4. Wassermann A, Neisser A, Bruck C. Eine serodiagnostische Reaktion bei Syphilis. Dtsch Med Wochenschr 1906;32:745–746.

5. Sweitzer S, Duncan JA, Seña AC. Update on syphilis diagnostics. Curr Opin Infect Dis 2025;38:44–53.



6. Papp JR, Park IU, Fakile Y, Pereira L, Pillay A, Bolan GA. CDC laboratory recommendations for syphilis testing, United States, 2024. MMWR Recomm Rep 2024;73:1–32.



8. Larsen SA, Steiner BM, Rudolph AH. Laboratory diagnosis and interpretation of tests for syphilis. Clin Microbiol Rev 1995;8:1–21.




9. Wang S, Kim S, Cho S, Kim HS, Min S. Introduction to the transition of mandatory surveillance system in the syphilis monitoring. Public Health Wkly Rep 2023;16:1620–1630.
10. Korea Centers for Disease Control Prevention. Infectious diseases surveillance yearbook 2008 [Internet] Osong: Korea Centers for Disease Control and Prevention, c2009. [cited 2025 Jul 3]. Available from: https://dportal.kdca.go.kr/pot/bbs/BD_selectBbs.do?q_bbsSn=1010&q_bbsDoc-No=20090601372072268&q_clsfNo=1.
11. Korea Centers for Disease Control Prevention. Infectious diseases surveillance yearbook 2010 [Internet] Osong: Korea Centers for Disease Control and Prevention, c2011. [cited 2025 Jul 3]. Available from: https://dportal.kdca.go.kr/pot/bbs/BD_selectBbs.do?q_bbsSn=1010&q_bbsDoc-No=20110601701279713&q_clsfNo=1.
12. Korea Centers for Disease Control Prevention. Infectious diseases surveillance yearbook 2019 [Internet] Osong: Korea Centers for Disease Control and Prevention, c2020. [cited 2025 Jul 3]. Available from: https://dportal.kdca.go.kr/pot/bbs/BD_selectBbs.do?q_bbsSn=1010&q_bbsDocNo=20200501473438871&q_clsfNo=1.
13. Korea Disease Control and Prevention Agency. Annual report on the notified infectious diseases in Korea, 2023 [Internet] Osong: Korea Disease Control and Prevention Agency, c2024. [cited 2025 Jul 3]. Available from: https://dportal.kdca.go.kr/pot/bbs/BD_selectBbs.do?q_bbsSn=1010&q_bbsDoc-No=20240628174016361&q_clsfNo=1.
14. Korea Disease Control and Prevention Agency. Annual report on the notified infectious diseases in Korea, 2024 [Internet] Osong: Korea Disease Control and Prevention Agency, c2025. [cited 2025 Jul 3]. Available from: https://dportal.kdca.go.kr/pot/bbs/BD_selectBbs.do?q_bbsSn=1010&q_bbsDoc-No=20250626112241525&q_clsfNo=1.
15. KOrean Statistical Information Service (KOSIS). Resident registration population statistics [Internet] Daejeon: KOSIS, c2025. [cited 2025 Jul 3]. Available from: https://kosis.kr/statHtml/statHtml.do?sso=ok&returnurl=https%3A%2F%2Fkosis.kr%3A443%2FstatHtml%2FstatHtml.do%3Flist_id%3DA_7%26obj_var_id%3D%26seqNo%3D%26tblId%3DDT_1B040M1_1%26vw_cd%3DMT_ZTITLE%26orgId%3D101%26path%3D%252FstatisticsList%252FstatisticsListIndex.do%26conn_path%3DMT_ZTITLE%26itm_id%3D%26lang_mode%3Dko%26scrId-%3D%26.
16. Lee E, Kim J, Bang JH, Lee JY, Cho SI. Association of HIV-syphilis coinfection with optimal antiretroviral adherence: a nation- wide claims study. AIDS Care 2020;32:651–655.


17. Statistical Bureau of Japan. Overview of population estimates [Internet] Tokyo: Statistical Bureau of Japan, c2025. [cited 2025 Jul 3]. Available from: https://www.stat.go.jp/data/jinsui/2.html#annual.
18. Ministry of Health Labour Welfare. Annual trends in reported numbers of sexually transmitted diseases (STDs) by gender [Internet] Tokyo: Ministry of Health Labour Welfare, c2025. [cited 2025 Jul 3]. Available from: https://www.mhlw.go.jp/topics/2005/04/tp0411-1.html.
19. National Institute of Infectious Diseases (NIID). Infectious Agents surveillance report [Internet] Tokyo: NIID, c2020. [cited 2025 Jul 3]. Available from: https://id-info.jihs.go.jp/idsc/niid/images/idsc/iasr/41/479e.pdf.
20. National Institute of Infectious Diseases (NIID). Summary of syphilis notifications in Japan [Internet] Tokyo (Japan): NIID, c2024. [cited 2025 Jul 3]. Available from: https://id-info.jihs.go.jp/surveillance/idwr/article/syphilis/010/syphilis2024q1.pdf.
21. Centers for Disease Control and Prevention (CDC). STI statististics - sexually transmitted infections surveillance, 2023 [Internet] Atlanta (GA): CDC, c2024. [cited 2025 Jul 3]. Available from: https://www.cdc.gov/sti-statistics/annual/index.html.
22. European Centre for Disease Prevention and Control (ECDC). Syphilis - annual epidemiological report for 2014 [Internet] Stockholm: ECDC, c2016. [cited 2025 Jul 3]. Available from: https://www.ecdc.europa.eu/sites/default/files/documents/Syphilis%20AER_0.pdf.
23. European Centre for Disease Prevention and Control (ECDC). Syphilis - annual epidemiological report for 2015 [Internet] Stockholm: ECDC, c2017. [cited 2025 Jul 3]. Available from: https://www.ecdc.europa.eu/sites/default/files/documents/AER_for_2015-syphilis.pdf.
24. European Centre for Disease Prevention and Control (ECDC). Syphilis - annual epidemiological report for 2016 [Internet] Stockholm: ECDC, c2018. [cited 2025 Jul 3]. Available from: https://www.ecdc.europa.eu/sites/default/files/documents/AER_for_2016-syphilis.pdf.
25. European Centre for Disease Prevention and Control (ECDC). Syphilis - Annual epidemiological report for 2017 [Internet] Stockholm: ECDC, c2019. [cited 2025 Jul 3]. Available from: https://www.ecdc.europa.eu/sites/default/files/documents/syphilis-annual-epidemiological-report-2017.pdf.
26. European Centre for Disease Prevention and Control (ECDC). Syphilis - annual epidemiological report for 2018 [Internet] Stockholm: ECDC, c2020. [cited 2025 Jul 3]. Available from: https://www.ecdc.europa.eu/sites/default/files/documents/syphilis-aer-2018.pdf.
27. European Centre for Disease Prevention and Control (ECDC). Syphilis - annual epidemiological report for 2019 [Internet] Stockholm: ECDC, c2022. [cited 2025 Jul 3]. Available from: https://www.ecdc.europa.eu/sites/default/files/documents/syphilis-annual-epidemiological-report-2019.pdf.
28. European Centre for Disease Prevention and Control (ECDC). Syphilis - annual epidemiological report for 2021 [Internet] Stockholm: ECDC, c2023. [cited 2025 Jul 3]. Available from: https://www.ecdc.europa.eu/sites/default/files/documents/syphilis-annual-epidemiological-report-2021.pdf.
29. European Centre for Disease Prevention and Control (ECDC). Syphilis – annual epidemiological report for 2022 [Internet] Stockholm: ECDC, c2024. [cited 2025 Jul 3]. Available from: https://www.ecdc.europa.eu/sites/default/files/documents/SYPH_AER_2022_Report_0.pdf.
30. European Centre for Disease Prevention and Control (ECDC). Syphilis – annual epidemiological report for 2023 [Internet] Stockholm: ECDC, c2025. [cited 2025 Jul 3]. Available from: https://www.ecdc.europa.eu/sites/default/files/documents/SYPH_AER_2023_Report.pdf.
31. Satyaputra F, Hendry S, Braddick M, Sivabalan P, Norton R. The laboratory diagnosis of syphilis. J Clin Microbiol 2021;59:e0010021.




32. Workowski KA, Bachmann LH, Chan PA, et al. Sexually transmitted infections treatment guidelines, 2021. MMWR Recomm Rep 2021;70:1–187.



33. Cho SH, Choi HW, Choi R, Lee SG, Kee SJ. Annual report on the Korean Association of External Quality Assessment Service on serological tests for syphilis (2022–2023). Lab Med Qual Assur 2025;47:33–42.

34. Leroy AG, Robert M, Carpentier M, et al. Assessment of a fully automated RPR assay (Mediace RPR) for serological diagnosis and follow-up of syphilis: a retrospective study. Diagn Microbiol Infect Dis 2022;104:115767.


35. Tsuboi M, Nishijima T, Aoki T, et al. Usefulness of automated latex turbidimetric rapid plasma reagin test for diagnosis and evaluation of treatment response in syphilis in comparison with manual card test: a prospective cohort study. J Clin Microbiol 2018;56:e01003–18.




36. Romanowski B, Sutherland R, Fick GH, Mooney D, Love EJ. Serologic response to treatment of infectious syphilis. Ann Intern Med 1991;114:1005–1009.


37. Haas JS, Bolan G, Larsen SA, Clement MJ, Bacchetti P, Moss AR. Sensitivity of treponemal tests for detecting prior treated syphilis during human immunodeficiency virus infection. J Infect Dis 1990;162:862–866.


38. Morshed MG, Singh AE. Recent trends in the serologic diagnosis of syphilis. Clin Vaccine Immunol 2015;22:137–147.



39. Loeffelholz MJ, Wen T, Patel JA. Analysis of bioplex syphilis IgG quantitative results in different patient populations. Clin Vaccine Immunol 2011;18:2005–2006.




40. Park IU, Chow JM, Bolan G, Stanley M, Shieh J, Schapiro JM. Screening for syphilis with the treponemal immunoassay: analysis of discordant serology results and implications for clinical management. J Infect Dis 2011;204:1297–1304.


41. Wong EH, Klausner JD, Caguin-Grygiel G, et al. Evaluation of an IgM/IgG sensitive enzyme immunoassay and the utility of index values for the screening of syphilis infection in a high-risk population. Sex Transm Dis 2011;38:528–532.


42. Yen-Lieberman B, Daniel J, Means C, et al. Identification of false-positive syphilis antibody results using a semiquantitative algorithm. Clin Vaccine Immunol 2011;18:1038–1040.



43. Fakile YF, Jost H, Hoover KW, et al. Correlation of treponemal immunoassay signal strength values with reactivity of confirmatory treponemal testing. J Clin Microbiol 2017;56:e01165–17.




44. Prince HE, Marlowe EM, Schwab DA. Relationship between DiaSorin Liaison Treponema pallidum antibody indices and confirmatory assay results in the reverse syphilis testing algorithm. Diagn Microbiol Infect Dis 2021;100:115303.


45. Murai R, Yamada K, Yonezawa H, Yanagihara N, Takahashi S. Evaluation of new algorithm using TPLA as an initial syphilis screening test. J Infect Chemother 2019;25:68–70.


46. Cha YJ, Bae JH, Park Q, Chae SL. Annual report on the external quality assessment scheme of viral markers and serological tests for syphilis in Korea (2014). J Lab Med Qual Assur 2015;37:124–133.

47. Pham MD, Ong JJ, Anderson DA, Drummer HE, Stoové M. Point-of-care diagnostics for diagnosis of active syphilis infection: needs, challenges and the way forward. Int J Environ Res Public Health 2022;19:8172.



48. Bocoum FY, Ouédraogo H, Tarnagda G, et al. Evaluation of the diagnostic performance and operational characteristics of four rapid immunochromatographic syphilis tests in Burkina Faso. Afr Health Sci 2015;15:360–367.



49. Van Den Heuvel A, Smet H, Prat I, et al. Laboratory evaluation of four HIV/syphilis rapid diagnostic tests. BMC Infect Dis 2019;19:1.


50. Jafari Y, Peeling RW, Shivkumar S, Claessens C, Joseph L, Pai NP. Are Treponema pallidum specific rapid and point-of-care tests for syphilis accurate enough for screening in resource limited settings? Evidence from a meta-analysis. PLoS One 2013;8:e54695.



51. Sabidó M, Benzaken AS, de-Andrade-Rodrigues EJ, Mayaud P. Rapid point-of-care diagnostic test for syphilis in high-risk populations, Manaus, Brazil. Emerg Infect Dis 2009;15:647–649.



52. Tsang RS, Shuel M, Hayden K, Van Caeseele P, Stein D. Laboratory evaluation of two point-of-care test kits for the identification of infectious syphilis. Can Commun Dis Rep 2022;48:82–88.



53. Holden J, Goheen J, Jett-Goheen M, Barnes M, Hsieh YH, Gaydos CA. An evaluation of the SD Bioline HIV/syphilis duo test. Int J STD AIDS 2018;29:57–62.




54. Leon SR, Ramos LB, Vargas SK, et al. Laboratory evaluation of a dual-path platform assay for rapid point-of-care HIV and syphilis testing. J Clin Microbiol 2016;54:492–494.




55. Bristow CC, Klausner JD, Tran A. Clinical test performance of a rapid point-of-care syphilis treponemal antibody test: a systematic review and meta-analysis. Clin Infect Dis 2020;71(Suppl 1):S52–S57.




56. Theel ES, Katz SS, Pillay A. Molecular and direct detection tests for treponema pallidum subspecies pallidum: a review of the literature, 1964–2017. Clin Infect Dis 2020;71(Suppl 1):S4–S12.




57. Gayet-Ageron A, Sednaoui P, Lautenschlager S, et al. Use of Treponema pallidum PCR in testing of ulcers for diagnosis of primary syphilis. Emerg Infect Dis 2015;21:127–129.



58. Grange PA, Gressier L, Dion PL, et al. Evaluation of a PCR test for detection of Treponema pallidum in swabs and blood. J Clin Microbiol 2012;50:546–552.




59. Aung ET, Fairley CK, Williamson DA, et al.
Treponema pallidum detection at asymptomatic oral, anal, and vaginal sites in adults reporting sexual contact with persons with syphilis. Emerg Infect Dis 2023;29:2083–2092.



60. Seña AC, White BL, Sparling PF. Novel Treponema pallidum serologic tests: a paradigm shift in syphilis screening for the 21st century. Clin Infect Dis 2010;51:700–708.


61. Hart G. Syphilis tests in diagnostic and therapeutic decision making. Ann Intern Med 1986;104:368–376.



62. Centers for Disease Control and Prevention (CDC). Syphilis testing algorithms using treponemal tests for initial screening--four laboratories, New York City, 2005–2006. MMWR Morb Mortal Wkly Rep 2008;57:872–875.

63. Cantor AG, Pappas M, Daeges M, Nelson HD. Screening for syphilis: updated evidence report and systematic review for the US preventive services task force. JAMA 2016;315:2328–2337.


64. Binnicker MJ, Jespersen DJ, Rollins LO. Direct comparison of the traditional and reverse syphilis screening algorithms in a population with a low prevalence of syphilis. J Clin Microbiol 2012;50:148–150.




65. Mishra S, Boily MC, Ng V, et al. The laboratory impact of changing syphilis screening from the rapid-plasma reagin to a treponemal enzyme immunoassay: a case-study from the Greater Toronto Area. Sex Transm Dis 2011;38:190–196.


66. Centers for Disease Control and Prevention (CDC). Discordant results from reverse sequence syphilis screening--five laboratories, United States, 2006–2010. MMWR Morb Mortal Wkly Rep 2011;60:133–137.


67. Gottlieb SL, Pope V, Sternberg MR, et al. Prevalence of syphilis seroreactivity in the United States: data from the National Health and Nutrition Examination Surveys (NHANES) 2001–2004. Sex Transm Dis 2008;35:507–511.


68. Huh HJ, Chung JW, Park SY, Chae SL. Comparison of automated treponemal and nontreponemal test algorithms as first-line syphilis screening assays. Ann Lab Med 2016;36:23–27.




69. Huh HJ, Lee KK, Kim ES, Chae SL. Analysis of positive results in mediace rapid plasma reagin and Treponema pallidum latex agglutination as the automated syphilis test. Korean J Lab Med 2007;27:324–329Korean.
70. Janier M, Unemo M, Dupin N, Tiplica GS, Potočnik M, Patel R. 2020 European guideline on the management of syphilis. J Eur Acad Dermatol Venereol 2021;35:574–588.



71. Kiołbasa M, Kaminiów K, Pastuszczak M. Serofast state after syphilis treatment: implications and recommendations for clinical practice. Narrative review. Postepy Dermatol Alergol 2025;42:215–220.



72. Tong ML, Lin LR, Liu GL, et al. Factors associated with serological cure and the serofast state of HIV-negative patients with primary, secondary, latent, and tertiary syphilis. PLoS One 2013;8:e70102.



-
METRICS

-
- 0 Crossref
- 0 Scopus
- 208 View
- 79 Download
- Related articles
-
Changes in the epidemiology and burden of community-acquired pneumonia in Korea2014 November;29(6)
Epidemiologic Study of Hepatitis B in Pregnant Korean Women1986 July;1(2)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print


