 |
 |
| Korean J Intern Med > Volume 32(1); 2017 > Article |
|
Abstract
Background/Aims
Methods
Results
Acknowledgments
Supplementary Materials
Supplementary Figure 1.
Figure 1.
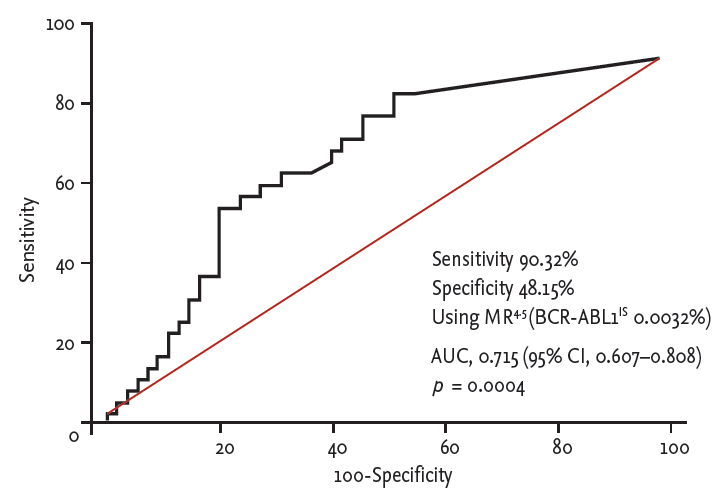
Figure 2.
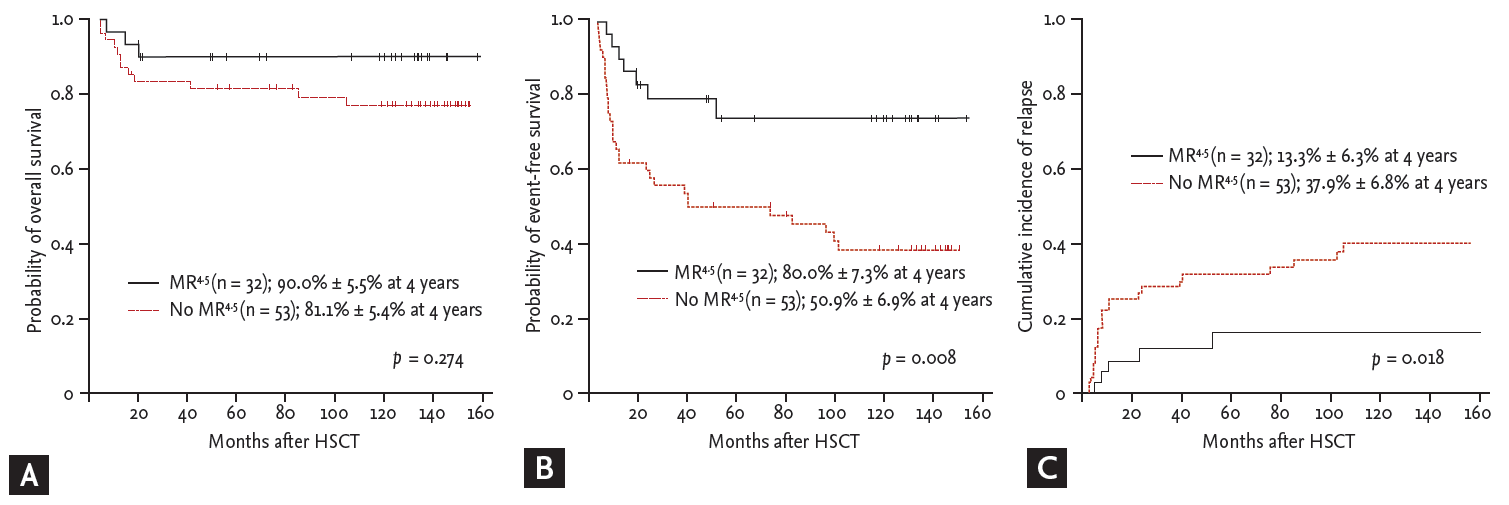
Table 1.
| Parameter | Total (n = 101) |
BCR-ABL1 transcript at post-transplant 3 monthsa |
||
|---|---|---|---|---|
| MR4.5 (n = 32) | No MR4.5 (n = 53) | p value | ||
| Age, yr | 32 (13-54) | 33.5 (20-51) | 32 (14-54) | 0.350 |
| Sex, male/female | 60 (59)/41 (41) | 15 (47)/17 (53) | 34 (64)/19 (36) | 0.118 |
| Disease phase at diagnosis, CP/AP/BP | 79 (78)/17 (17)/5 (5) | 22 (69)/8 (25)/2 (6) | 45 (85)/6 (11)/2 (4) | 0.203 |
| Disease phase at transplant, CP | 101 (100) | 32 (100) | 53 (100) | - |
| Disease status at transplant | ||||
| No MCyR/MCyR | 63 (62)/38 (38) | 17 (53)/15 (47) | 39 (74)/14 (26) | 0.054 |
| > 10% MR/≤ 10% MR | 63 (62)/38 (38) | 17 (53)/15 (47) | 39 (74)/14 (26) | 0.054 |
| No MMR/MMR | 90 (89)/11 (11) | 26 (81)/6 (19) | 49 (93)/4 (7) | 0.167 |
| Previous therapy before SCT | 0.003 | |||
| No prior TKI | 47 (47) | 10 (31) | 34 (64) | |
| Prior TKI(s) | 54 (54)b | 22 (69) | 19 (36) | |
| Interval from diagnosis to transplant, mon | 11.6 (4.0-132.2) | 13.2 (4.0-58.7) | 10.4 (4.8-132.2) | 0.023 |
| Sokal score, low/intermediate/high/NA | 31 (31)/22 (22)/30 (30)/18 (18) | 8 (25)/4 (12)/13 (41)/7 (22) | 19 (36)/16 (30)/11 (21)/7 (13) | 0.069 |
| EBMT score | 0.043 | |||
| 0 and 1 | 23 (23) | 4 (13) | 17 (32) | |
| 2 | 38 (38) | 11 (34) | 22 (42) | |
| 3 and 4 | 38 (38) | 16 (50) | 14 (26) | |
| 5 | 2 (2) | 1 (3) | 0 | |
| Donor type | 0.407 | |||
| Related | 58 (57) | 58 (57) | 58 (57) | |
| Unrelated | 43 (43) | 43 (43) | 43 (43) | |
| Donor/recipient sex match | ||||
| F to M/othersc | 18 (18)/83 (82) | 6 (19)/26 (81) | 10 (19)/43 (81) | 0.407 |
| Source of graft | ||||
| BM/PBSC-basedd | 72 (71)/29 (29) | 18 (56)/14 (44) | 43 (81)/10 (19) | 0.014 |
| CD34+ cells (× 106/kg) | 4.1 (1.2-20.0) | 4.7 (1.3-20.0) | 3.7 (1.2-17.9) | 0.285 |
| CD3+ cells (× 107/kg) | 5.35 (0.2-109.7) | 8.59 (0.3-57.3) | 5.49 (1.7-109.7) | 0.549 |
| Conditioning intensity, standard/RIC | 64 (63)/37 (37) | 22 (69)/10 (31) | 32 (60)/21 (40) | 0.437 |
| GVHD prophylaxis, CsA + MTX/FK506 + MTX | 52 (52)/49 (49) | 14 (44)/18 (56) | 28 (53)/25 (47) | 0.417 |
Values are presented as median (range) or number (%).
MR, molecular response; CP, chronic phase; AP, accelerated phase; BP, blast phase; MCyR, major cytogenetic response; MMR, major molecular response; SCT, stem cell transplantation; TKI, tyrosine kinase inhibitor; NA, not available; EBMT, European Group for Blood and Marrow Transplantation; F, female; M, male; BM, bone marrow; PBSC, peripheral blood stem cell; RIC, reduced-intensity conditioning; GVHD, graft-versus-host disease; CsA, cyclosporine; MTX, methotrexate.
a 85 patients had available quantitative reverse transcriptase polymerase chain reaction records at 3 months after SCT.
Table 2.
| Parameter | Total (n = 101) |
BCR-ABL1 transcript at post-transplant 3 monthsa |
||
|---|---|---|---|---|
| MR4.5 (n = 32) | No MR4.5 (n = 53) | p value | ||
| Hematologic reconstitution and engraftment | ||||
| Primary graft failure | 1 | - | - | - |
| Secondary graft failure | 0 | - | - | - |
| Engraftment kineticsb | ||||
| Days to ANC > 0.5 × 109/L | 13 (0-38) | 12 (7-25) | 14 (0-25) | 0.164 |
| Days to PLT > 20 × 109/L | 19 (0-53) | 15 (0-28) | 22 (0-53) | 0.165 |
| Acute GVHD | 0.616 | |||
| Grade I | 7 (7) | 1 (3) | 5 (9) | |
| Grade II | 17 (17) | 7 (22) | 7 (13) | |
| Grade III | 4 (4) | 1 (3) | 2 (4) | |
| Grade IV | 2 (2) | 0 | 1 (2) | |
| CI (≥ grade II) at 100 days | 22.8 ± 4.2 | 25.3 ± 7.9 | 18.9 ± 5.4 | 0.475 |
| Chronic GVHD, E/N | 0.385 | |||
| Limited | 19/93 (20) | 6/31 (19) | 13/53 (25) | |
| Extensive | 35/93 (38) | 14/31 (45) | 16/53 (30) | |
| CI (extensive) at 4 years | 35.0 ± 7.8 | 45.3 ± 9.3 | 30.2 ± 6.4 | 0.199 |
| Cumulative incidence of relapse | ||||
| 3 mon | 2.0 ± 1.4 | 0 | 0 | - |
| 1 yr | 23.4 ± 4.3 | 10.0 ± 5.6 | 30.2 ± 6.4 | 0.034 |
| 4 yr | 29.6 ± 4.6 | 13.3 ± 6.3 | 37.9 ± 6.8 | 0.018 |
| Cumulative incidence of TRM | ||||
| 3 mon | 2.0 ± 1.4 | 0 | 0 | - |
| 1 yr | 8.1 ± 2.7 | 0 | 7.5 ± 3.7 | 0.121 |
| 4 yr | 12.2 ± 3.3 | 6.7 ± 4.6 | 9.5 ± 4.1 | 0.628 |
| Event-free survival | ||||
| 3 mon | 95.0 ± 2.2 | 100 | 100 | - |
| 1 yr | 68.6 ± 4.7 | 90.0 ± 5.5 | 62.3 ± 6.7 | 0.007 |
| 4 yr | 57.3 ± 5.0 | 80.0 ± 7.3 | 50.9 ± 6.9 | 0.008 |
| Overall survival | ||||
| 3 mon | 97.0 ± 1.7 | 100 | 100 | - |
| 1 yr | 86.8 ± 3.4 | 96.7 ± 3.3 | 86.8 ± 4.7 | 0.145 |
| 4 yr | 80.6 ± 4.0 | 90.0 ± 5.5 | 81.1 ± 5.4 | 0.274 |
Values are presented as median (range), number (%), or mean ± SE.
MR, molecular response; ANC, absolute neutrophil count; PLT, platelet; GVHD, graft-versus-host disease; E/N, number of events/number of evaluable patients; CI, cumulative incidence; TRM, transplant-related mortality.
Table 3.
| Variable |
OS at 4 years |
EFS at 4 years |
Relapse at 4 years |
TRM at 4 years |
||||
|---|---|---|---|---|---|---|---|---|
| RR (95% CI) | p value | RR (95% CI) | p value | RR (95% CI) | p value | RR (95% CI) | p value | |
| Increasing age, yr | 1.03 (0.98-1.08) | 0.210 | 1.00 (0.96-1.03) | 0.777 | 0.96 (0.92-1.00) | 0.041 | 1.07 (1.02-1.12) | 0.006 |
| Age of patient, yr | ||||||||
| ≤ 35 | 1 | 1 | 1 | 1 | ||||
| > 35 | 2.01 (0.81-4.94) | 0.130 | 0.78 (0.41-1.48) | 0.446 | 0.22 (0.08-0.64) | 0.005 | 5.51 (1.51-20.10) | 0.010 |
| Patient sex | ||||||||
| Male | 1 | 1 | 1 | 1 | ||||
| Female | 0.66 (0.25-1.74) | 0.400 | 0.96 (0.52-1.78) | 0.902 | 0.99 (0.45-2.05) | 0.980 | 1.04 (0.33-3.24) | 0.950 |
| Disease phase at diagnosis | ||||||||
| Chronic phase | 1 | 1 | 1 | 1 | ||||
| Accelerated phase | 1.45 (0.47-4.44) | 0.518 | 0.84 (0.35-1.99) | 0.685 | 0.80 (0.27-2.35) | 0.680 | 1.02 (0.22-4.66) | 0.980 |
| Blast phase | 3.85 (0.87-17.12) | 0.077 | 3.26 (0.99-10.70) | 0.052 | 2.19 (0.46-10.39) | 0.320 | 2.48 (0.27-22.73) | 0.420 |
| Disease status at transplant | ||||||||
| MCyR | ||||||||
| No | 1 | 1 | 1 | 1 | ||||
| Yes | 1.33 (0.50-3.49) | 0.566 | 0.86 (0.47-1.60) | 0.637 | 0.57 (0.28-1.16) | 0.120 | 3.14 (0.71-13.90) | 0.130 |
| MR, % | ||||||||
| > 10 | 1 | 1 | 1 | 1 | ||||
| ≤ 10 | 1.37 (0.52-3.61) | 0.520 | 1.00 (0.54-1.87) | 0.993 | 0.68 (0.33-1.41) | 0.300 | 3.14 (0.71-13.90) | 0.130 |
| MMR | ||||||||
| No | 1 | 1 | 1 | 1 | ||||
| Yes | 1.15 (0.27-4.97) | 0.854 | 1.43 (0.51-4.01) | 0.495 | 1.24 (0.42-3.66) | 0.690 | 1.44 (0.196-10.50) | 0.720 |
| Treatment prior to SCT | ||||||||
| No TKI | 1 | 1 | 1 | 1 | ||||
| IM | 1.17 (0.45-3.04) | 0.744 | 0.98 (0.50-1.92) | 0.958 | 0.93 (0..43-2.05) | 0.860 | 1.12 (0.35-3.59) | 0.850 |
| IM and 2G-TKI(s)a | 0.67 (0.15-3.11) | 0.609 | 1.10 (0.46-2.59) | 0.835 | 1.14 (0.40-3.31) | 0.800 | 0.47 (0.06-4.00) | 0.490 |
| Interval from diagnosis to transplant, yr | 1.00 (0.97-1.03) | 0.810 | 1.01 (1.00-1.03) | 0.198 | 1.01 (0.98-1.03) | 0.700 | 1.00 (0.97-1.03) | 0.860 |
| EBMT score | ||||||||
| 0-2 | 1 | 1 | 1 | 1 | ||||
| ≥ 3 | 2.01 (0.82-4.95) | 0.130 | 1.36 (0.74-2.51) | 0.321 | 0.72 (0.33-1.57) | 0.400 | 3.56 (1.09-11.60) | 0.035 |
| Sokal score | ||||||||
| Low | 1 | 1 | 1 | 1 | ||||
| Intermediate | 1.03 (0.33-3.26) | 0.956 | 0.94 (0.42-2.10) | 0.884 | 1.02 (0.42-2.47) | 0.970 | 0.71 (0.14-3.72) | 0.680 |
| High | 0.49 (0.13-1.91) | 0.307 | 0.74 (0.33-1.64) | 0.452 | 0.69 (0.26-1.80) | 0.440 | 0.57 (0.10-3.06) | 0.510 |
| NA | 1.12 (0.33-3.84) | 0.854 | 0.81 (0.33-1.98) | 0.638 | 0.42 (0.12-1.39) | 0.150 | 1.97 (0.51-7.60) | 0.320 |
| Year of transplant | ||||||||
| 2001-2004 | 1 | 1 | 1 | 1 | ||||
| 2005-2008 | 0.61 (0.08-4.58) | 0.628 | 1.20 (0.42-3.40) | 0.732 | 1.39 (0.49-3.93) | 0.530 | 1.02 (0.15-6.99) | 0.990 |
| 2009-2012 | 0.59 (0.14-2.58) | 0.485 | 0.91 (0.38-2.17) | 0.828 | 0.92 (0.29-2.87) | 0.880 | 0.44 (0.06-3.51) | 0.440 |
| Donor type | ||||||||
| Related | 1 | 1 | 1 | 1 | ||||
| Unrelated | 1.59 (0.64-3.90) | 0.316 | 1.51 (0.83-2.77) | 0.180 | 1.12 (0.55-2.31) | 0.750 | 2.04 (0.66-6.33) | 0.220 |
| Donor/recipient sex match | ||||||||
| F to M | 1 | 1 | 1 | 1 | ||||
| Others | 0.82 (0.27-2.47) | 0.721 | 1.11 (0.49-2.50) | 0.805 | 1.42 (0.48-4.17) | 0.520 | 0.65 (0.18-2.36) | 0.510 |
| Source of graft | ||||||||
| BM | 1 | 1 | 1 | 1 | ||||
| PBSC-based | 0.65 (0.22-1.95) | 0.441 | 0.34 (0.14-0.81) | 0.014 | 0.08 (0.01-0.55) | 0.011 | 1.29 (0.40-4.21) | 0.670 |
| Conditioning intensity | ||||||||
| Standard | 1 | 1 | 1 | 1 | ||||
| RIC | 0.69 (0.26-1.82) | 0.455 | 0.69 (0.36-1.32) | 0.262 | 0.93 (0.45-1.91) | 0.830 | 0.52 (0.15-1.85) | 0.310 |
| GVHD prophylaxis | ||||||||
| CsA-based | 1 | 1 | 1 | 1 | ||||
| FK506-based | 1.15 (0.47-2.84) | 0.785 | 1.52 (0.83-2.80) | 0.179 | 1.39 (0.67-2.87) | 0.380 | 1.51 (0.48-4.74) | 0.480 |
OS, overall survival; EFS, event-free survival; TRM, transplant-related mortality; RR, risk ratio; CI, confidence interval; MCyR, major cytogenetic response; MR, molecular response; MMR, major molecular response; SCT, stem cell transplantation; TKI, tyrosine kinase inhibitor; IM, imatinib mesylate; 2G-TKI, second generation tyrosine kinase inhibitor; EBMT, European Group for Blood and Marrow Transplantation; NA, not available; BM, bone marrow; PBSC, peripheral blood stem cell; RIC, reduced-intensity conditioning; GVHD, graft-versus-host disease; CsA, cyclosporine.
Table 4.
| Variablea |
Patients with post-SCT qRT-PCR at 3 months (n = 85) |
||
|---|---|---|---|
| No. | RR (95% CI) | p value | |
| Relapse | |||
| Age, yr | |||
| ≤ 35 | 54 | 1 | |
| > 35 | 31 | 0.26 (0.08-0.86) | 0.028 |
| Source of graft | |||
| PBSC-based | 24 | 1 | |
| BM | 61 | 7.04 (0.94-52.44) | 0.057 |
| MR4.5 at 3 months | |||
| Yes | 32 | 1 | |
| No | 53 | 2.46 (0.87-6.95) | 0.089 |
| Transplant-related mortality | |||
| Age, yr | |||
| ≤ 35 | 54 | 1 | |
| > 35 | 31 | 8.05 (0.66-97.80) | 0.100 |
| EBMT score | |||
| 0-2 | 54 | 1 | |
| > 3 | 31 | 3.34 (0.47-23.60) | 0.230 |
| MR4.5 at 3 months | |||
| Yes | 32 | 1 | |
| No | 53 | 2.46 (0.46-13.30) | 0.290 |
| Event-free survival | |||
| Source of graft | |||
| PBSC based | 24 | 1 | |
| BM | 61 | 2.71 (0.94-7.79) | 0.064 |
| MR4.5 at 3 months | |||
| Yes | 32 | 1 | |
| No | 53 | 2.73 (1.12-6.68) | 0.028 |
| Overall survival | |||
| Disease phase at diagnosis | |||
| Chronic phase | 67 | 1 | |
| Accelerated phase | 14 | 1.04 (0.22-4.86) | 0.961 |
| Blast phase | 4 | 2.52 (0.32-19.79) | 0.379 |
| MR4.5 at 3 months | |||
| Yes | 32 | 1 | |
| No | 53 | 2.04 (0.55-7.58) | 0.285 |
REFERENCES
- TOOLS



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Supplement 1
Supplement 1 Print
Print



