 |
 |
| Korean J Intern Med > Volume 38(3); 2023 > Article |
|
Abstract
Sarcopenia is a condition characterized by a loss of muscle mass and function. In chronic kidney disease (CKD), where a chronic catabolic state exists, sarcopenia commonly occurs through various mechanisms, resulting in muscle wasting and decreased muscle endurance. Sarcopenic patients with CKD have high morbidity and mortality rates. Indeed, the prevention and treatment of sarcopenia are mandatory. An imbalance between protein synthesis and degradation in muscle and increased oxidative stress and inflammation persist in CKD and induce muscle wasting. In addition, uremic toxins negatively affect muscle maintenance. A variety of potential therapeutic drugs targeting these muscle-wasting mechanisms in CKD have been investigated, but most of the trials focused on aged patients without CKD, and none of these drugs have been approved for the treatment of sarcopenia so far. Further studies on the molecular mechanisms of sarcopenia in CKD and targets for potential therapeutics are needed to improve the outcomes of sarcopenic patients with CKD.
Sarcopenia is a condition characterized by a loss of muscle mass and function (either muscle strength or physical performance) and generally develops with age [1]. When first used, the term sarcopenia was used to describe an age-related loss of muscle mass only [2]. The definition of sarcopenia has been updated and now includes muscle function. The most widely used definition of sarcopenia nowadays is proposed by the European Working Group on Sarcopenia in Older People (EWGSOP) 2. The EWGSOP2 considers low muscle mass as a key characteristic of sarcopenia, low muscle quantity and quality to confirm the diagnosis and poor physical performance to indicate the severe sarcopenia [3]. The operational definition of sarcopenia proposed by EWGSOP2 is presented in Table 1. Sarcopenia is associated with poor health-related quality of life, including organ dysfunction [4,5] and is a significant risk factor for some cancers [6].
However, in addition to aging, several underlying conditions, including malnutrition, low physical activity, specific drugs and diseases can also cause sarcopenia. In patients with chronic kidney disease (CKD), where a chronic catabolic state exists, muscle wasting and decreased muscle endurance occur, and sarcopenia commonly occurs [2]. Compared to the aging-related sarcopenia where protein degradation is not changed, CKD-related sarcopenia is related to the increased muscle protein degradation and protein energy wasting (PEW), cachexia are usually present in patients with CKD [7,8]. The prevalence of sarcopenia using different definitions (mostly EWGSOP1 [9] and EWGSOP2 [3] criteria) ranged from 5.9% [10] to 14% [11] in CKD patients without kidney replacement therapy, 13.7% [12] to 42.2% [13] in patients on hemodialysis (HD), and 4% [14] to 15.5% [15] in patients on peritoneal dialysis. Indeed, sarcopenic patients with CKD have significantly worse physical performance [11,16], a higher risk of disability [13], an increased risk of intradialytic hypotension during HD [17], increased mortality [10,18-21], and cardiovascular events [22,23]. Therefore, the diagnosis and interventions to treat sarcopenia in patients with CKD are important to improve outcomes.
Risk factors for sarcopenia in patients with CKD include both modifiable factors (malnutrition [11,12,18,19,22,24-26] and low body mass index [16,19,27,28]) and non-modifiable factors (age [19,24,26,27,29], male sex [27,30], diabetes mellitus [12,19,27,29], longer dialysis duration [28] and dialysis modality [29]). Because of the differences between the aging-related and CKD-related sarcopenia as described above, the treatment goals are also different. In patients with aging-related sarcopenia, restoring mobility and quality of life is the main goals. In patients with CKD-related sarcopenia where muscle wasting and PEW are more prominent, recovering nutritional status to improve the response to the treatment of the CKD is more important [7]. Aerobic and resistance exercises show inconsistent positive effects on sarcopenia but still play a major role as interventions for the prevention and treatment of sarcopenia in patients with CKD [31-34]. Additionally, nutritional interventions [35-39], optimized dialysis, and correction of acidosis [40,41] are important strategies [42]. However, there is no consensus on the unified methods of both exercise and nutritional interventions, which limits the practical approach to sarcopenia in patients with CKD. Therefore, understanding the pathophysiology and molecular mechanisms of sarcopenia in CKD and developing potential therapeutic agents are important. In this review, we aimed to address pharmacologic and nutritional interventions in which the targets were derived from the molecular mechanisms of sarcopenia in CKD.
Chronic catabolic conditions persist in CKD and induce an imbalance between protein synthesis and degradation, resulting in muscle wasting [43,44]. Chronic inflammation and uremic toxin-induced muscle wasting are other important factors associated with sarcopenia in CKD. This review involves elucidating the mechanisms of sarcopenia associated with CKD in terms of protein synthesis and degradation of muscle, oxidative stress with inflammation, and the effect of uremic toxins, and describing the available drugs or nutritional supplements for each mechanism (Fig. 1). The ongoing clinical trials designed to investigate the drug’s effect on muscle were researched on October 2022 through ClinicalTrials.gov and are presented in Table 2.
Alterations in protein synthesis have been consistently observed in animal models and some patients with CKD [45-50]. Through the mammalian target of ramamycin (mTOR) and inactivation of the Forkhead box protein O (FoxO), the insulin or insulin-like growth factor (IGF)-1-phosphatidylinositol 3-kinase-Akt pathway increases protein synthesis in muscle [51-55]. Impaired insulin tolerance induces muscle atrophy [56] and IGF-1 treatment suppresses FoxO expression [57]. In addition, testosterone binds to the androgen receptor (AR) and regulates myogenic gene expression by stimulating Akt/mTOR complex 1 (mTORC1) and suppressing FoxO-targeted gene expression. As a result, it promotes protein synthesis and inhibits protein degradation [58,59]. Drugs relative to the mechanisms of protein synthesis include metformin, testosterone, selective androgen receptor modulators (SARM), and mTOR inhibitors. Also, the supplementation of the amino acid is another treatment option to stimulate the muscle protein synthesis.
Diabetic nephropathy is the leading cause of CKD in Korea and the United States [60-62]. Diabetes mellitus is a major risk factor for sarcopenia in patients with both CKD and non-CKD [12,19,27,29,63] and interestingly, sarcopenia is an independent risk factor for diabetic nephropathy in type 2 diabetes [64]. Hyperglycemia contributes to the loss of both muscle mass and function by increasing insulin resistance, inflammatory cytokines, and accumulation of glycation end-products [65-67]. Several classes of anti-diabetic agents have shown a positive effect on improving energy metabolism in muscles in vivo [68-72] and this review is focused on the most extensively investigated drug, metformin. Metformin is a commonly prescribed drug for type 2 diabetes, and by activating AMP-activated protein kinase (AMPK), it induces the expression of muscle hexokinase and glucose transporters, mimicking the effects of extensive exercise training [73]. In addition, metformin is known to increase the follicular fluid IGF-1 levels in patients with polycystic ovary syndrome [74] and this finding suggests the promotion of protein synthesis in muscles by metformin. However, metformin treatment did not increase muscle mass or longevity in either the sedentary or exercise mouse groups [75]. Metformin also blunted increases in mTORC1 signaling in response to progressive resistance exercise training and negatively impacted the hypertrophic responses to exercise in healthy older adults [76]. In line with these findings, a meta-analysis showed that metformin did not affect exercise capacity [77]. Although the previous disappointing results of the studies, several clinical trials are recruiting the patients to investigate the effect of metformin on muscle mass and function (Table 2).
Testosterone plays an important role in the maintenance of muscle mass and function via the aforementioned molecular mechanisms. More than 60% of men with CKD have testosterone deficiency and uremic hypogonadism [78-80]. In mice, testosterone improved skeletal muscle regeneration and prevented muscle atrophy [81,82]. Several trials involving testosterone have consistently shown a positive effect of the drug in increasing muscle mass and function in older male patients [83-86]. However, various side effects of testosterone, including cardiovascular events, prostate hyperplasia, and lower urinary tract symptoms, occurred during the trials [83,87]. Clinical trials using testosterone undecanoate, which showed no risk of prostate cancer or cardiovascular disease [88] are recruiting patients and one trial (NCT05249634) is recruiting the patients with CKD (Table 2). SARMs function as anoints/antagonists of AR, and by their selective action, SARM therapy has fewer off-target side effects [89]. Several SARMs, such as MK-0773, GTx-024, and GSK2881078, increased lean body mass in healthy older individuals, but MK-0773 and GTx-024 failed to improve physical performance [90-93].
mTORC1 promotes protein synthesis by phosphorylating two key effectors, p70S6 kinase 1 and eIF4E binding protein [94]. Acute activation of mTORC1 signaling in vivo promotes muscle hypertrophy [95], but in the chronic state, both inhibition and hyperactivation of mTORC1 result in muscle atrophy. Long-term inhibition of mTOR by rapamycin induced insulin resistance of muscle in rats [96]. In a retrospective study in which the patients received mTOR inhibitors for at least 6 months, the drugs significantly decreased the skeletal muscle area and lean body mass [97]. Moreover, mTOR hyperactivation has been observed in both aged rodent and human muscles [98]. In a mouse model of muscle dystrophy, hyperactive mTORC1 signaling was observed, and the mTORC1 inhibitor rapamycin improved skeletal muscle function [99]. Although evidence of hyperactivation of mTORC1 in muscles is scarce in CKD, acute and chronic kidney injury constitutively activates mTOR signaling in kidney fibroblasts, leading to kidney damage [100]. Therefore, mTOR inhibitors are potential drugs for the treatment of sarcopenia in CKD. Based on these findings, two clinical trials investigating the impact of mTOR inhibition on muscle are currently recruiting patients (Table 2).
Amino acids are classified to the essential amino acid (EAA) and non-EAA. Among the EAAs, the branched-chain amino acids (BCAA) [101,102] and leucine [103-105] are known to induce stimulation of muscle protein synthesis. In CKD, various combined conditions (inflammation, catabolic illnesses [106], acidosis [43,107,108], nutritional loss to dialysate [109-111], endocrine disorders such as resistance to insulin [112], growth hormone [113], and IGF-1 [114], hyperparathyroidism [115]) can lead to PEW where body stores of protein and energy fuels are decreased [116]. Therefore, the plasma and cellular levels of the BCAA and leucine are commonly low in CKD [115]. Since protein restriction is essential to minimize uremic toxicity and delay progression of the kidney disease, BCAA and leucine supplements are effective to improve the sarcopenia while reducing the total amount of the protein intake in CKD patients [117]. Both in rat and elderly patients, administration of BCAA and leucine are effective to improve muscle protein synthesis [118-121]. In HD patients with malnutrition, the EAA supplements improved appetite, increased plasma albumin levels and enhanced muscle strength [36,37,122]. However, β-hydroxy-β-methylbutyrate, a metabolite of leucine showed no benefit on body composition in HD patients [123]. Currently, several clinical trials are recruiting patients with chronic liver disease to investigate the effect of amino acid supplementation on sarcopenia (Table 2).
In CKD, protein degradation increases via increased expression of atrophy-inducing genes (atrogen) and atrophy-related biomarkers [51,124-127]. Myostatin (growth differentiation factor 8) is an autocrine inhibitor of muscle growth and is mainly produced in skeletal muscles [128,129]. It binds to activin type 2 receptors on the muscle fiber membrane and subsequently recruits and activates activin type 1 receptor B and transforming growth factor β to phosphorylate Smad 2/3 [130]. In addition, it reduces Akt signaling, regulates the Akt/mTOR pathways, and suppresses the FoxO pathway [131]. Through this pathway, myostatin promotes protein degradation in the muscle and functions as a negative regulator of muscle mass [132]. The myostatin maturation process and extracellular binding proteins, such as follistatin, also regulate the myostatin pathway, and follistatin functions as a myostatin antagonist [133]. Myostatin increases in patients with CKD, and several factors, including low physical activity, oxidative stress and inflammation, uremic toxins, angiotensin II (Ang II), metabolic acidosis, and glucocorticoids, maybe the contributors [51,134-136]. Drugs that target the myostatin pathway include myostatin inhibitors, activin receptor antagonists, and follistatin-based drugs. Most trials investigating the effects of drugs on sarcopenia have recruited patients without CKD.
The upregulation of muscle myostatin was observed both in a CKD rodent model and in patients with CKD [135,137]. In CKD mice, an anti-myostatin peptibody suppressed circulating inflammatory cytokines and reversed the loss of body weight and muscle mass [135]. Landogrozumab (LY-2495655), a humanized monoclonal antibody for myostatin, increased lean mass and showed a tendency to improve functional measures of muscle power in older patients [138]. Another monoclonal anti-myostatin antibody, trevogrumab (REGN-1033), showed a tendency toward increased muscle size in only a few patients with muscular dystrophy and demonstrated good safety and tolerability [139].
In a mouse model, bimagrumab, a specific monoclonal antibody that binds to the activin type 2A/2B receptor, significantly promoted skeletal muscle hypertrophy [140,141]. A phase II clinical trial with bimagrumab showed the effect of the drug on increasing thigh muscle mass and grip strength and improving mobility in patients with sarcopenia [142]. A subsequent phase II/III trial was completed in 2018, and data analysis is ongoing [143]. In patients with type 2 diabetes and obesity, bimagrumab resulted in the loss of fat mass, gain of lean mass, and metabolic improvements [144]. Ramatercept, a soluble form of the activin type 2 receptor, significantly increased the cross-sectional area of type I and II muscle fibers in a mouse model [145], but in its phase II trial, serious non-muscle-related adverse events of the drug were observed, and the trial was terminated [146].
Intramuscular injection of FST288-Fc, a follistatin fusion protein, induced the growth of targeted muscles [147] and systemic administration of monovalent follistatin-like 3-Fc-fusion protein induced muscle fiber hypertrophy and increased muscle mass in a mouse model [148]. Another follistatin-based fusion protein, ACE-083, also induced localized skeletal muscle hypertrophy and increased focal force generation in a mouse model [149]. However, in a phase II trial of ACE-083, treatment increased muscle mass but did not improve functional outcomes [150]. Associated virus (AAV) serotype 1. Follistatin, which acts as a natural myostatin antagonist, significantly increases muscle mass and strength in a mouse model of muscular dystrophy [151]. FS344, an isoform of follistatin by AAV 1, improved ambulation in patients with Becker muscular dystrophy or sporadic inclusion body myositis [152-154].
Oxidative stress and inflammation are features of CKD [155] and they also induce muscle wasting. Through the nuclear factor kappa-light-chain-enhancer of activated B cells pathway, reactive oxygen species (ROS)-induced tumor necrosis factor (TNF)-α activates myostatin expression [137] and increased inflammatory cytokines, such as TNF-α and interleukin (IL)-6, causing muscle atrophy in patients with CKD [156-158]. Owing to their antioxidant effects, angiotensin-converting enzyme (ACE) inhibitor, Ang II type I receptor blocker (ARB), vitamins, resveratrol, and its anti-inflammatory effect, ghrelin are potential therapeutic drugs and supplements for sarcopenia in CKD.
The RAAS plays a role in systemic physiology and is responsible for blood pressure control, maintenance of fluid homeostasis, and electrolyte balance [159]. Along with the major contributions of RAAS to these mechanisms, aberrant signaling through RAAS in CKD also influences muscle wasting [160]. The protease renin cleaves angiotensinogen and forms angiotensin I (Ang I), and ACE cleaves Ang I to produce Ang II. The three most investigated membrane receptors for RAAS hormone peptides are the Ang II type 1 receptor (AT1R), Ang II type 2 receptor, and the mitochondrial assembly receptor (MASR). These receptors are expressed in various tissues, including smooth muscle and skeletal muscle fibers [161,162]. When Ang II binds to AT1R on the cell membrane, the classical RAAS signaling pathways are activated. By transducing the signals to downstream secondary messengers, AT1R signaling produces ROS and contributes to muscle wasting [160]. Angiotensin-(1-7), the principal hormone in the non-classical RAAS pathway, activates MASR and inhibits AT1R activation [163]. Therefore, disrupting the classical RAAS pathway through inhibition of ACE or blocking of AT1R and promoting MASR are potential therapeutic targets to reduce muscle wasting.
ACE inhibitors and ARB are the most commonly prescribed antihypertensive drugs in patients with CKD because of their effect on slowing the decline in kidney function, decreasing urine protein excretion, and adverse cardiovascular outcomes [164-167]. Along with renoprotective effects, these drugs are expected to inhibit muscle atrophy by blocking Ang II production. Treatment of hypertension with ACE inhibitors showed a slower decline in muscle strength and mobility [168] and higher muscle mass of the lower limb than with other antihypertensive drugs [169]. It also increased the IGF-1 levels in older patients [170]. However, in a recently published randomized controlled trial to determine the efficacy of leucine and/or perindopril in improving physical performance or muscle mass in older patients with sarcopenia, neither leucine nor perindopril showed this effect [171]. In the case of ARB, losartan improved muscle remodeling, protected against disuse atrophy [172], improved mobility, and reduced inflammation and oxidative stress in sarcopenic mice [173]. However, losartan showed no effect in preventing mobility loss in older adults [174]. In addition, there was no significant effect in preventing muscle strength loss in pre-frail older patients with losartan treatment (NCT01989793, completed in 2016). One clinical trial (NCT03295734) is recruiting older patients to investigate the effect of perindopril on muscle function compared to losartan or hydrochlorothiazide while all patients will participate in a structured aerobic exercise intervention (Table 2).
MASR agonists attenuate muscle atrophy by activating MASR and inhibiting the downstream signaling of the AT1 receptor. AVE 0991, an MASR agonist, showed multiple attenuated muscles wasting in mice with cancer cachexia [175]. A phase II trial investigating the safety and tolerability of the MASR agonist BIO101 measured gait speed, several body mass indicators, and power in older patients (NCT03452488). The study was completed but no significant results were reported.
Vitamin B is a cofactor with methyl donors regulating the level of homocysteine. Uremia-induced hyperhomocysteinemia occurs in patients with CKD and is associated with poorer outcomes [176-178]. Several studies demonstrated that high homocysteine levels are also linked to impaired muscle strength [179,180] and physical performance [181,182] in older patients. A 2-year randomized controlled trial of vitamin B12 and folic acid supplementation showed a positive effect on gait speed, but not on muscle strength [183]. Two clinical trials investigating the vitamin B3 are ongoing (Table 2). Vitamin C is a potential water-soluble antioxidant, and its effects have been demonstrated in many in vitro experiments [184-188]. Plasma vitamin C concentration declines with kidney function [189] and additional loss of this component into the dialysate occurs in patients on HD [190]. Higher vitamin C intake was associated with higher skeletal muscle mass and power in free-living women [185]. Therefore, clinical trials investigating the effect of vitamin C supplementation on muscle in patients with CKD seem promising though not conducted yet.
Because vitamin D has a structure homologous to cholesterol, it may be regarded as an antioxidant, and this effect has been suggested to have a non-calcemic role [158]. Vitamin D deficiency in CKD is common and is associated with low bone formation rate, bone mineral density, and muscle atrophy [191]. In a mouse model, vitamin D3 reduced the extent of lipid peroxidation and induced superoxide dismutase activity; these effects were similar to those of vitamin E [158]. In older patients, vitamin D supplementation increased muscle mass and strength [192,193]. The effect of vitamin D was enhanced in older patients with vitamin D deficiency [194]. Several clinical trials are actively recruiting patients to investigate the effect of vitamin D on muscle and two trials are recruiting the patients on maintenance dialysis (Table 2). Vitamin K acts as a cofactor of γ-carboxylation of some proteins [195,196] and one of the γ-carboxylated proteins, AR has an important role in protein synthesis of the muscle as described above [197]. CKD patients have subclinical vitamin K deficiency [198,199]. Several clinical studies demonstrated that high vitamin K was associated with better physical performance, suggesting the beneficial effect of vitamin K in muscle quality [200-203]. To investigate the benefit of vitamin K supplementation on muscle, two clinical studies are designed and one study is currently recruiting the patients (Table 2).
Resveratrol is a natural phenolic compound found in many foods, such as grapes, blueberries, and peanuts. Resveratrol has been shown to have antioxidant and anti-inflammatory properties [204,205]. Resveratrol improved skeletal muscle mass and function and prevented sarcopenia in a rat model [206-208]. In addition, resveratrol improved exercise performance and physical endurance in a mouse model [209,210]. However, in a trial of humans, resveratrol impaired exercise training-induced improvements by reducing oxidative stress and inflammation markers in skeletal muscles [211]. Two clinical trials evaluating the clinical efficacy of resveratrol in improving skeletal muscle in patients with chronic heart failure or peripheral artery disease are being conducted (Table 2).
Ghrelin is an acylated peptide that stimulates growth hormones and subsequently stimulates feeding [212]. In addition to its effects on appetite regulation, ghrelin has been shown to exert anti-inflammatory effects. There are major forms of circulating ghrelin, acylated and des-acyl ghrelin. Acylated ghrelin increases food intake and des-acyl ghrelin induces negative energy balance [213,214] The levels of des-acyl were elevated in CKD patients [215]. Ghrelin and a synthetic ghrelin receptor agonist significantly decreased the expression of IL-1 receptor-I transcript in the brain and thus improved lean body mass retention in a rat model of cancer cachexia [216]. In CKD rats, treatment with ghrelin and two ghrelin receptor agonists (BIM-28125 and BIM-28131) resulted in decreased muscle protein degradation and circulatory inflammatory cytokines, thus increasing food intake and improving lean body mass [217]. Ghrelin and the ghrelin agonist anamorelin have been shown to increase food intake and muscle mass in cancer patients [218-221]. One clinical trial investigating the effect of ghrelin is recruiting patients with peripheral artery disease (NCT04377126). Capromorelin, a ghrelin receptor agonist, increased lean mass and physical performance in sarcopenic elderly patients [222]. A phase II trial designed to determine the effect of MK-0677, a growth hormone secretagogue, on lean body mass in CKD stage 4/5 patients was withdrawn because the investigators could not obtain drug supply from the manufacturer (NCT01343641).
Uremic toxins increase in serum along with a decline in kidney function, and the negative effect of accumulated uremic toxins on muscle wasting is a specific mechanism of CKD [223]. Protein-bound uremic toxins, including p-Cresyl sulfate and indoxyl sulfate (IS), have been investigated for their effects on muscle wasting. These toxins are taken up by cells through an organic anion transporter [224,225]. In mice, p-Cresyl sulfate alters the insulin signaling pathway by suppressing insulin-induced phosphorylation of Akt, resulting in insulin resistance [226]. IS induces metabolic alterations via nuclear factor (erythroid-2-related factor)-2 in skeletal muscle [227]. IS also enhances the production of muscle atrophy-related genes like myostatin and atrogin-1 and increases ROS and inflammatory cytokines [228,229]. In addition, it acts as an aryl hydrocarbon receptor (AHR) ligand, and AHR works as a component of the ubiquitin ligase complex [230,231]. Through these mechanisms, IS induces skeletal muscle wasting and is a potential therapeutic drug target [229,232]. AST-120, an absorbent capsule used to remove circulating IS, significantly reversed the negative changes in the skeletal muscle by reducing circulating IS in CKD mice [232]. In a phase IV trial, AST-120 showed modest benefits in gait speed change and quality of life, but the changes were not significant in patients with CKD [233].
This review elucidated the brief molecular mechanisms of sarcopenia associated with CKD and the potential therapeutic drugs and nutritional supplements for sarcopenia categorized by each mechanism. Various mechanisms, including an imbalance in protein synthesis and degradation, increased oxidative stress and inflammation, and uremic toxins, contribute to muscle wasting and result in sarcopenia in CKD. Some potential therapeutic drugs have been investigated, and promising drugs are under ongoing clinical trials. Further clinical trials testing the effects of drugs in patients with CKD and more studies unveiling the potential molecular treatment targets for sarcopenia are needed to improve the outcomes of sarcopenic patients with CKD.
Notes
Figure 1.
Molecular mechanisms of muscle wasting in chronic kidney disease and targeted drugs or supplements. CKD, chronic kidney disease; ROS, reactive oxygen species; RAAS, renin-angiotensin-aldosterone system; IL, interleukin; TNF, tumor necrosis factor; IGF, insulin- like growth factor; SARM, selective androgen receptor modulator; IS, indoxyl sulfate; Rc, receptor; ActRIIB, activin receptor type IIB; OAT, organic anion transport; PI3K, phosphatidylinositol 3-kinase; FoxO, forkhead box protein O; mTOR, mammalian target of ramamycin; mTORi, mammalian target of ramamycin inhibitor; MuRF1, muscle RING-finger protein-1; AHR, aryl hydrocarbon receptor; NADPH, nicotinamide adenine dinucleotide phosphate.
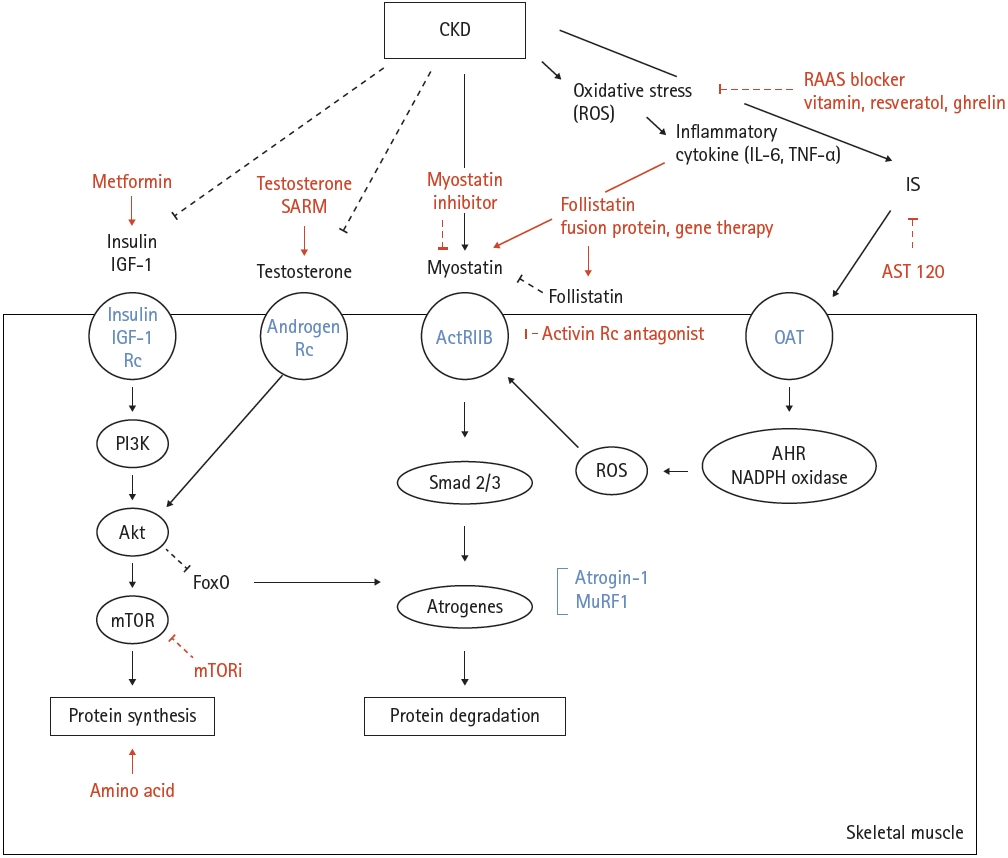
Table 1.
Operational definition of sarcopenia proposed by the European Working Group on Sarcopenia in Older People 2 [3]
| Cut-off points | Diagnosis | ||
|---|---|---|---|
| Find cases | SARC-F or clinical suspicion | ||
| Assess | Muscle strength | Sarcopenia probablea | |
| Grip strength: men < 27 kg, women < 16 kg, ch air stand > 15 seconds for five rises | |||
| Confirm | Muscle massb (quantity or quality) | Sarcopenia confirmed | |
| Appendicular muscle mass: men < 20 kg, women < 15 kg | |||
| Appendicular muscle mass/height2: men < 7.0 kg/m2, women < 5.5 kg/m2 | |||
| Severity | Physical performance | Sarcopenia severe | |
| Gait speed ≤ 0.8 m/s | |||
| Short physical performance battery ≤ 8 score, timed up and go test ≥ 20 seconds, 400 m walk: non-completion or ≥ 6 minutes for completion | |||
Table 2.
The ongoing clinical trials investigating the effect of drugs and nutritional supplements on muscle
AMPK, AMP-activated protein kinase; NA, not applicable; CKD, chronic kidney disease; mTOR, mammalian target of ramamycin; EAA, essential amino acid; BAA, balanced amino acid; HMB, β-hydroxy-β-methylbutyrate; ACE, angiotensin-converting enzyme; Ang II, angiotensin II; HD, hemodialysis; PD, peritoneal dialysis.
REFERENCES
3. Cruz-Jentoft AJ, Bahat G, Bauer J, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 2019;48:601.



4. Park H, Jang IY, Han M, et al. Sarcopenia is associated with severe erectile dysfunction in older adults: a population-based cohort study. Korean J Intern Med 2020;35:1245–1253.




5. Sun S, Lee H, Yim HW, Won HS, Ko YH. The impact of sarcopenia on health-related quality of life in elderly people: Korean National Health and Nutrition Examination Survey. Korean J Intern Med 2019;34:877–884.




6. Kim MC, Kim KO, Kang MK. Prevalence and associated risk of advanced colorectal neoplasia in adults with sarcopenia. Korean J Intern Med 2022;37:294–303.




7. Sabatino A, Cuppari L, Stenvinkel P, Lindholm B, Avesani CM. Sarcopenia in chronic kidney disease: what have we learned so far? J Nephrol 2021;34:1347–1372.




8. Carrero JJ, Thomas F, Nagy K, et al. Global prevalence of protein-energy wasting in kidney disease: a meta-analysis of contemporary observational studies from the international society of renal nutrition and metabolism. J Ren Nutr 2018;28:380–392.


9. Cruz-Jentoft AJ, Baeyens JP, Bauer JM, et al. Sarcopenia: European consensus on definition and diagnosis: report of the European Working Group on sarcopenia in older people. Age Ageing 2010;39:412–423.


10. Pereira RA, Cordeiro AC, Avesani CM, et al. Sarcopenia in chronic kidney disease on conservative therapy: prevalence and association with mortality. Nephrol Dial Transplant 2015;30:1718–1725.


11. Vettoretti S, Caldiroli L, Armelloni S, Ferrari C, Cesari M, Messa P. Sarcopenia is associated with malnutrition but not with systemic inflammation in older persons with advanced CKD. Nutrients 2019;11:1378.



12. Ren H, Gong D, Jia F, Xu B, Liu Z. Sarcopenia in patients undergoing maintenance hemodialysis: incidence rate, risk factors and its effect on survival risk. Ren Fail 2016;38:364–371.


13. Hotta C, Hiraki K, Wakamiya A, et al. Relation of physical function and physical activity to sarcopenia in hemodialysis patients: a preliminary study. Int J Cardiol 2015;191:198–200.


14. da Silva MZC, Vogt BP, Reis NSDC, Caramori JCT. Update of the European consensus on sarcopenia: what has changed in diagnosis and prevalence in peritoneal dialysis? Eur J Clin Nutr 2019;73:1209–1211.



15. Abro A, Delicata LA, Vongsanim S, Davenport A. Differences in the prevalence of sarcopenia in peritoneal dialysis patients using hand grip strength and appendicular lean mass: depends upon guideline definitions. Eur J Clin Nutr 2018;72:993–999.



16. Souza VA, Oliveira D, Barbosa SR, et al. Sarcopenia in patients with chronic kidney disease not yet on dialysis: analysis of the prevalence and associated factors. PLoS One 2017;12:e0176230.



17. Son HE, Ryu JY, Lee K, et al. The importance of muscle mass in predicting intradialytic hypotension in patients undergoing maintenance hemodialysis. Kidney Res Clin Pract 2022;41:611–622.




18. Kamijo Y, Kanda E, Ishibashi Y, Yoshida M. Sarcopenia and frailty in PD: impact on mortality, malnutrition, and inflammation. Perit Dial Int 2018;38:447–454.



19. Mori K, Nishide K, Okuno S, et al. Impact of diabetes on sarcopenia and mortality in patients undergoing hemodialysis. BMC Nephrol 2019;20:105.




20. Kittiskulnam P, Chertow GM, Carrero JJ, Delgado C, Kaysen GA, Johansen KL. Sarcopenia and its individual criteria are associated, in part, with mortality among patients on hemodialysis. Kidney Int 2017;92:238–247.



21. Kang SH, Kim AY, Do JY. Association of sarcopenia and its components with clinical outcomes in patients undergoing peritoneal dialysis. Kidney Res Clin Pract 2022;41:741–752.




22. Kim JK, Kim SG, Oh JE, et al. Impact of sarcopenia on long-term mortality and cardiovascular events in patients undergoing hemodialysis. Korean J Intern Med 2019;34:599–607.




23. Tabibi H, As’habi A, Najafi I, Hedayati M. Prevalence of dynapenic obesity and sarcopenic obesity and their associations with cardiovascular disease risk factors in peritoneal dialysis patients. Kidney Res Clin Pract 2018;37:404–413.



24. Isoyama N, Qureshi AR, Avesani CM, et al. Comparative associations of muscle mass and muscle strength with mortality in dialysis patients. Clin J Am Soc Nephrol 2014;9:1720–1728.



25. Kim JK, Choi SR, Choi MJ, et al. Prevalence of and factors associated with sarcopenia in elderly patients with end-stage renal disease. Clin Nutr 2014;33:64–68.


26. Lin YL, Liou HH, Lai YH, et al. Decreased serum fatty acid binding protein 4 concentrations are associated with sarcopenia in chronic hemodialysis patients. Clin Chim Acta 2018;485:113–118.


27. Ishikawa S, Naito S, Iimori S, et al. Loop diuretics are associated with greater risk of sarcopenia in patients with non-dialysis-dependent chronic kidney disease. PLoS One 2018;13:e0192990.



28. Bataille S, Serveaux M, Carreno E, Pedinielli N, Darmon P, Robert A. The diagnosis of sarcopenia is mainly driven by muscle mass in hemodialysis patients. Clin Nutr 2017;36:1654–1660.


29. Silva MZC, Antonio KJ, Reis JMS, Alves LS, Caramori JCT, Vogt BP. Age, diabetes mellitus, and dialysis modality are associated with risk of poor muscle strength and physical function in hemodialysis and peritoneal dialysis patients. Kidney Res Clin Pract 2021;40:294–303.




30. As’habi A, Najafi I, Tabibi H, Hedayati M. Prevalence of sarcopenia and dynapenia and their determinants in Iranian peritoneal dialysis patients. Iran J Kidney Dis 2018;12:53–60.

31. Lopes LCC, Mota JF, Prestes J, et al. Intradialytic resistance training improves functional capacity and lean mass gain in individuals on hemodialysis: a randomized pilot trial. Arch Phys Med Rehabil 2019;100:2151–2158.


32. Dong ZJ, Zhang HL, Yin LX. Effects of intradialytic resistance exercise on systemic inflammation in maintenance hemodialysis patients with sarcopenia: a randomized controlled trial. Int Urol Nephrol 2019;51:1415–1424.




33. Zhou Y, Hellberg M, Hellmark T, Höglund P, Clyne N. Muscle mass and plasma myostatin after exercise training: a substudy of Renal Exercise (RENEXC)-a randomized controlled trial. Nephrol Dial Transplant 2021;36:95–103.




34. Koh KP, Fassett RG, Sharman JE, Coombes JS, Williams AD. Effect of intradialytic versus home-based aerobic exercise training on physical function and vascular parameters in hemodialysis patients: a randomized pilot study. Am J Kidney Dis 2010;55:88–99.


35. Allman MA, Stewart PM, Tiller DJ, Horvath JS, Duggin GG, Truswell AS. Energy supplementation and the nutritional status of hemodialysis patients. Am J Clin Nutr 1990;51:558–562.


36. Eustace JA, Coresh J, Kutchey C, et al. Randomized double-blind trial of oral essential amino acids for dialysis-associated hypoalbuminemia. Kidney Int 2000;57:2527–2538.


37. Hiroshige K, Sonta T, Suda T, Kanegae K, Ohtani A. Oral supplementation of branched-chain amino acid improves nutritional status in elderly patients on chronic haemodialysis. Nephrol Dial Transplant 2001;16:1856–1862.


38. Zilles M, Betz C, Jung O, et al. How to prevent renal cachexia? A clinical randomized pilot study testing oral supplemental nutrition in hemodialysis patients with and without human immunodeficiency virus infection. J Ren Nutr 2018;28:37–44.


39. Pupim LB, Majchrzak KM, Flakoll PJ, Ikizler TA. Intradialytic oral nutrition improves protein homeostasis in chronic hemodialysis patients with deranged nutritional status. J Am Soc Nephrol 2006;17:3149–3157.


40. de Brito-Ashurst I, Varagunam M, Raftery MJ, Yaqoob MM. Bicarbonate supplementation slows progression of CKD and improves nutritional status. J Am Soc Nephrol 2009;20:2075–2084.



41. Stein A, Moorhouse J, Iles-Smith H, et al. Role of an improvement in acid-base status and nutrition in CAPD patients. Kidney Int 1997;52:1089–1095.


42. Stenvinkel P, Carrero JJ, von Walden F, Ikizler TA, Nader GA. Muscle wasting in end-stage renal disease promulgates premature death: established, emerging and potential novel treatment strategies. Nephrol Dial Transplant 2016;31:1070–1077.


43. Bailey JL, Wang X, England BK, Price SR, Ding X, Mitch WE. The acidosis of chronic renal failure activates muscle proteolysis in rats by augmenting transcription of genes encoding proteins of the ATP-dependent ubiquitin-proteasome pathway. J Clin Invest 1996;97:1447–1453.



44. Wang XH, Du J, Klein JD, Bailey JL, Mitch WE. Exercise ameliorates chronic kidney disease-induced defects in muscle protein metabolism and progenitor cell function. Kidney Int 2009;76:751–759.



45. Adey D, Kumar R, McCarthy JT, Nair KS. Reduced synthesis of muscle proteins in chronic renal failure. Am J Physiol Endocrinol Metab 2000;278:E219–E225.


46. Deger SM, Hung AM, Gamboa JL, et al. Systemic inflammation is associated with exaggerated skeletal muscle protein catabolism in maintenance hemodialysis patients. JCI Insight 2017;2:e95185.



47. Garibotto G, Sofia A, Russo R, et al. Insulin sensitivity of muscle protein metabolism is altered in patients with chronic kidney disease and metabolic acidosis. Kidney Int 2015;88:1419–1426.



48. Raj DS, Dominic EA, Wolfe R, et al. Coordinated increase in albumin, fibrinogen, and muscle protein synthesis during hemodialysis: role of cytokines. Am J Physiol Endocrinol Metab 2004;286:E658–E664.


49. van Vliet S, Skinner SK, Beals JW, et al. Dysregulated handling of dietary protein and muscle protein synthesis after mixed-meal ingestion in maintenance hemodialysis patients. Kidney Int Rep 2018;3:1403–1415.



50. May RC, Kelly RA, Mitch WE. Mechanisms for defects in muscle protein metabolism in rats with chronic uremia. Influence of metabolic acidosis. J Clin Invest 1987;79:1099–1103.



51. Wang XH, Mitch WE. Mechanisms of muscle wasting in chronic kidney disease. Nat Rev Nephrol 2014;10:504–516.




52. Bodine SC, Latres E, Baumhueter S, et al. Identification of ubiquitin ligases required for skeletal muscle atrophy. Science 2001;294:1704–1708.


53. Stitt TN, Drujan D, Clarke BA, et al. The IGF-1/PI3K/Akt pathway prevents expression of muscle atrophy-induced ubiquitin ligases by inhibiting FOXO transcription factors. Mol Cell 2004;14:395–403.


54. Lai KM, Gonzalez M, Poueymirou WT, et al. Conditional activation of akt in adult skeletal muscle induces rapid hypertrophy. Mol Cell Biol 2004;24:9295–9304.




55. Bonaldo P, Sandri M. Cellular and molecular mechanisms of muscle atrophy. Dis Model Mech 2013;6:25–39.




56. Lee SW, Dai G, Hu Z, Wang X, Du J, Mitch WE. Regulation of muscle protein degradation: coordinated control of apoptotic and ubiquitin-proteasome systems by phosphatidylinositol 3 kinase. J Am Soc Nephrol 2004;15:1537–1545.

57. Sandri M, Sandri C, Gilbert A, et al. Foxo transcription factors induce the atrophy-related ubiquitin ligase atrogin-1 and cause skeletal muscle atrophy. Cell 2004;117:399–412.



59. White JP, Gao S, Puppa MJ, Sato S, Welle SL, Carson JA. Testosterone regulation of Akt/mTORC1/FoxO3a signaling in skeletal muscle. Mol Cell Endocrinol 2013;365:174–186.



60. Johansen KL, Chertow GM, Foley RN, et al. US renal data system 2020 annual data report: epidemiology of kidney disease in the United States. Am J Kidney Dis 2021;77(4 Suppl 1):A7–A8.


61. Hong YA, Ban TH, Kang CY, et al. Trends in epidemiologic characteristics of end-stage renal disease from 2019 Korean Renal Data System (KORDS). Kidney Res Clin Pract 2021;40:52–61.




62. Jin DC, Yun SR, Lee SW, et al. Current characteristics of dialysis therapy in Korea: 2016 registry data focusing on diabetic patients. Kidney Res Clin Pract 2018;37:20–29.



63. Kim TN, Park MS, Yang SJ, et al. Prevalence and determinant factors of sarcopenia in patients with type 2 diabetes: the Korean Sarcopenic Obesity Study (KSOS). Diabetes Care 2010;33:1497–1499.


64. Huang YM, Chen WM, Chen M, Shia BC, Wu SY. Sarcopenia is an independent risk factor for severe diabetic nephropathy in type 2 diabetes: a long-term follow-up propensity score-matched diabetes cohort study. J Clin Med 2022;11:2992.



65. Abbatecola AM, Ferrucci L, Ceda G, et al. Insulin resistance and muscle strength in older persons. J Gerontol A Biol Sci Med Sci 2005;60:1278–1282.


66. Abbatecola AM, Paolisso G. Is there a relationship between insulin resistance and frailty syndrome? Curr Pharm Des 2008;14:405–410.


67. Mori H, Kuroda A, Ishizu M, et al. Association of accumulated advanced glycation end-products with a high prevalence of sarcopenia and dynapenia in patients with type 2 diabetes. J Diabetes Investig 2019;10:1332–1340.




68. Yokota T, Kinugawa S, Hirabayashi K, et al. Pioglitazone improves whole-body aerobic capacity and skeletal muscle energy metabolism in patients with metabolic syndrome. J Diabetes Investig 2017;8:535–541.




69. Sato H, Kubota N, Kubota T, et al. Anagliptin increases insulin-induced skeletal muscle glucose uptake via an NO-dependent mechanism in mice. Diabetologia 2016;59:2426–2434.



70. Skov V, Glintborg D, Knudsen S, et al. Pioglitazone enhances mitochondrial biogenesis and ribosomal protein biosynthesis in skeletal muscle in polycystic ovary syndrome. PLoS One 2008;3:e2466.



71. Chai W, Dong Z, Wang N, et al. Glucagon-like peptide 1 recruits microvasculature and increases glucose use in muscle via a nitric oxide-dependent mechanism. Diabetes 2012;61:888–896.




72. Giannocco G, Oliveira KC, Crajoinas RO, et al. Dipeptidyl peptidase IV inhibition upregulates GLUT4 translocation and expression in heart and skeletal muscle of spontaneously hypertensive rats. Eur J Pharmacol 2013;698:74–86.


73. Holmes BF, Kurth-Kraczek EJ, Winder WW. Chronic activation of 5’-AMP-activated protein kinase increases GLUT-4, hexokinase, and glycogen in muscle. J Appl Physiol (1985) 1999;87:1990–1995.


74. Pawelczyk L, Spaczynski RZ, Banaszewska B, Duleba AJ. Metformin therapy increases insulin-like growth factor binding protein-1 in hyperinsulinemic women with polycystic ovary syndrome. Eur J Obstet Gynecol Reprod Biol 2004;113:209–213.


75. Hernández-Álvarez D, Mena-Montes B, Toledo-Pérez R, et al. Long-term moderate exercise combined with metformin treatment induces an hormetic response that prevents strength and muscle mass loss in old female wistar rats. Oxid Med Cell Longev 2019;2019:3428543.


76. Walton RG, Dungan CM, Long DE, et al. Metformin blunts muscle hypertrophy in response to progressive resistance exercise training in older adults: a randomized, double-blind, placebo-controlled, multicenter trial: the MASTERS trial. Aging Cell 2019;18:e13039.




77. Das S, Behera SK, Srinivasan A, et al. Effect of metformin on exercise capacity: a meta-analysis. Diabetes Res Clin Pract 2018;144:270–278.


78. Dousdampanis P, Trigka K, Fourtounas C, Bargman JM. Role of testosterone in the pathogenesis, progression, prognosis and comorbidity of men with chronic kidney disease. Ther Apher Dial 2014;18:220–230.


79. Leavey SF, Weitzel WF. Endocrine abnormalities in chronic renal failure. Endocrinol Metab Clin North Am 2002;31:107–119.


81. Serra C, Tangherlini F, Rudy S, et al. Testosterone improves the regeneration of old and young mouse skeletal muscle. J Gerontol A Biol Sci Med Sci 2013;68:17–26.



82. Axell AM, MacLean HE, Plant DR, et al. Continuous testosterone administration prevents skeletal muscle atrophy and enhances resistance to fatigue in orchidectomized male mice. Am J Physiol Endocrinol Metab 2006;291:E506–E516.


83. LeBrasseur NK, Lajevardi N, Miciek R, Mazer N, Storer TW, Bhasin S. Effects of testosterone therapy on muscle performance and physical function in older men with mobility limitations (the TOM trial): design and methods. Contemp Clin Trials 2009;30:133–140.



84. Bhasin S, Ellenberg SS, Storer TW, et al. Effect of testosterone replacement on measures of mobility in older men with mobility limitation and low testosterone concentrations: secondary analyses of the testosterone trials. Lancet Diabetes Endocrinol 2018;6:879–890.



85. Dias JP, Veldhuis JD, Carlson O, et al. Effects of transdermal testosterone gel or an aromatase inhibitor on serum concentration and pulsatility of growth hormone in older men with age-related low testosterone. Metabolism 2017;69:143–147.



86. O’Connell MD, Roberts SA, Srinivas-Shankar U, et al. Do the effects of testosterone on muscle strength, physical function, body composition, and quality of life persist six months after treatment in intermediate-frail and frail elderly men? J Clin Endocrinol Metab 2011;96:454–458.



87. Dias JP, Melvin D, Shardell M, et al. Effects of transdermal testosterone gel or an aromatase inhibitor on prostate volume in older men. J Clin Endocrinol Metab 2016;101:1865–1871.




88. Corona G, Maseroli E, Maggi M. Injectable testosterone undecanoate for the treatment of hypogonadism. Expert Opin Pharmacother 2014;15:1903–1926.


89. Christiansen AR, Lipshultz LI, Hotaling JM, Pastuszak AW. Selective androgen receptor modulators: the future of androgen therapy? Transl Androl Urol 2020;9(Suppl 2):S135–S148.



90. Papanicolaou DA, Ather SN, Zhu H, et al. A phase IIA randomized, placebo-controlled clinical trial to study the efficacy and safety of the selective androgen receptor modulator (SARM), MK-0773 in female participants with sarcopenia. J Nutr Health Aging 2013;17:533–543.



91. Dalton JT, Barnette KG, Bohl CE, et al. The selective androgen receptor modulator GTx-024 (enobosarm) improves lean body mass and physical function in healthy elderly men and postmenopausal women: results of a double-blind, placebo-controlled phase II trial. J Cachexia Sarcopenia Muscle 2011;2:153–161.




92. Crawford J, Prado CM, Johnston MA, et al. Study design and rationale for the phase 3 clinical development program of enobosarm, a selective androgen receptor modulator, for the prevention and treatment of muscle wasting in cancer patients (POWER trials). Curr Oncol Rep 2016;18:37.




93. Dalton JT. The long and winding road for selective androgen receptor modulators. Br J Clin Pharmacol 2017;83:2131–2133.




94. Saxton RA, Sabatini DM. mTOR signaling in growth, metabolism, and disease. Cell 2017;168:960–976.



95. Bodine SC, Stitt TN, Gonzalez M, et al. Akt/mTOR pathway is a crucial regulator of skeletal muscle hypertrophy and can prevent muscle atrophy in vivo. Nat Cell Biol 2001;3:1014–1019.



96. Deblon N, Bourgoin L, Veyrat-Durebex C, et al. Chronic mTOR inhibition by rapamycin induces muscle insulin resistance despite weight loss in rats. Br J Pharmacol 2012;165:2325–2340.



97. Gyawali B, Shimokata T, Honda K, et al. Muscle wasting associated with the long-term use of mTOR inhibitors. Mol Clin Oncol 2016;5:641–646.



98. Chrienova Z, Nepovimova E, Kuca K. The role of mTOR in age-related diseases. J Enzyme Inhib Med Chem 2021;36:1678–1692.



99. Ramos FJ, Chen SC, Garelick MG, et al. Rapamycin reverses elevated mTORC1 signaling in lamin A/C-deficient mice, rescues cardiac and skeletal muscle function, and extends survival. Sci Transl Med 2012;4:144ra103.



101. Garlick PJ, Grant I. Amino acid infusion increases the sensitivity of muscle protein synthesis in vivo to insulin. Effect of branched-chain amino acids. Biochem J 1988;254:579–584.




102. Li JB, Jefferson LS. Influence of amino acid availability on protein turnover in perfused skeletal muscle. Biochim Biophys Acta 1978;544:351–359.


103. Buse MG, Reid SS. Leucine. A possible regulator of protein turnover in muscle. J Clin Invest 1975;56:1250–1261.



104. Anthony JC, Yoshizawa F, Anthony TG, Vary TC, Jefferson LS, Kimball SR. Leucine stimulates translation initiation in skeletal muscle of postabsorptive rats via a rapamycin-sensitive pathway. J Nutr 2000;130:2413–2419.


105. Anthony JC, Reiter AK, Anthony TG, et al. Orally administered leucine enhances protein synthesis in skeletal muscle of diabetic rats in the absence of increases in 4E-BP1 or S6K1 phosphorylation. Diabetes 2002;51:928–936.



106. Grodstein GP, Blumenkrantz MJ, Kopple JD. Nutritional and metabolic response to catabolic stress in uremia. Am J Clin Nutr 1980;33:1411–1416.


107. Kalantar-Zadeh K, Mehrotra R, Fouque D, Kopple JD. Poor nutritional status and inflammation: metabolic acidosis and malnutrition‐inflammation complex syndrome in chronic renal failure. Semin Dial 2004;17:455–465.


108. Pickering WP, Price SR, Bircher G, Marinovic AC, Mitch WE, Walls J. Nutrition in CAPD: serum bicarbonate and the ubiquitin-proteasome system in muscle. Kidney Int 2002;61:1286–1292.


109. Wolfson M, Jones MR, Kopple JD. Amino acid losses during hemodialysis with infusion of amino acids and glucose. Kidney Int 1982;21:500–506.


110. Ikizler TA, Flakoll PJ, Parker RA, Hakim RM. Amino acid and albumin losses during hemodialysis. Kidney Int 1994;46:830–837.


111. Chazot C, Shahmir E, Matias B, Laidlaw S, Kopple JD. Dialytic nutrition: provision of amino acids in dialysate during hemodialysis. Kidney Int 1997;52:1663–1670.


112. Mak RH. Insulin resistance but IGF-I sensitivity in chronic renal failure. Am J Physiol 1996;271(1 Pt 2):F114–F119.


113. Gupta V, Lee M. Growth hormone in chronic renal disease. Indian J Endocrinol Metab 2012;16:195–203.



114. Fouque D, Peng SC, Shamir E, Kopple JD. Recombinant human insulin-like growth factor-1 induces an anabolic response in malnourished CAPD patients. Kidney Int 2000;57:646–654.


115. Kopple JD, Cianciaruso B, Massry SG. Does parathyroid hormone cause protein wasting? Contrib Nephrol 1980;20:138–148.


116. Fouque D, Kalantar-Zadeh K, Kopple J, et al. A proposed nomenclature and diagnostic criteria for protein-energy wasting in acute and chronic kidney disease. Kidney Int 2008;73:391–398.


117. Ikizler TA, Burrowes JD, Byham-Gray LD, et al. KDOQI clinical practice guideline for nutrition in CKD: 2020 update. Am J Kidney Dis 2020;76(3 Suppl 1):S1–S107.


118. Haba Y, Fujimura T, Oyama K, et al. Effect of oral branched-chain amino acids and glutamine supplementation on skeletal muscle atrophy after total gastrectomy in rat model. J Surg Res 2019;243:281–288.


119. Baptista IL, Silva WJ, Artioli GG, et al. Leucine and HMB differentially modulate proteasome system in skeletal muscle under different sarcopenic conditions. PLoS One 2013;8:e76752.



120. Bai GH, Tsai MC, Tsai HW, Chang CC, Hou WH. Effects of branched-chain amino acid-rich supplementation on EWGSOP2 criteria for sarcopenia in older adults: a systematic review and meta-analysis. Eur J Nutr 2022;61:637–651.



121. Gielen E, Beckwée D, Delaere A, et al. Nutritional interventions to improve muscle mass, muscle strength, and physical performance in older people: an umbrella review of systematic reviews and meta-analyses. Nutr Rev 2021;79:121–147.



122. Acchiardo S, Moore L, Cockrell S. Effect of essential amino acids (EAA) on chronic hemodialysis (CHD) patients (PTS). Trans Am Soc Artif Intern Organs 1982;28:608–614.

123. Wu H, Xia Y, Jiang J, et al. Effect of beta-hydroxy-beta-methylbutyrate supplementation on muscle loss in older adults: a systematic review and meta-analysis. Arch Gerontol Geriatr 2015;61:168–175.


125. Cohen S, Nathan JA, Goldberg AL. Muscle wasting in disease: molecular mechanisms and promising therapies. Nat Rev Drug Discov 2015;14:58–74.



126. Du J, Wang X, Miereles C, et al. Activation of caspase-3 is an initial step triggering accelerated muscle proteolysis in catabolic conditions. J Clin Invest 2004;113:115–123.



127. Workeneh BT, Rondon-Berrios H, Zhang L, et al. Development of a diagnostic method for detecting increased muscle protein degradation in patients with catabolic conditions. J Am Soc Nephrol 2006;17:3233–3239.


128. Elkasrawy MN, Hamrick MW. Myostatin (GDF-8) as a key factor linking muscle mass and bone structure. J Musculoskelet Neuronal Interact 2010;10:56–63.


129. Han HQ, Zhou X, Mitch WE, Goldberg AL. Myostatin/activin pathway antagonism: molecular basis and therapeutic potential. Int J Biochem Cell Biol 2013;45:2333–2347.


130. Walker RG, Poggioli T, Katsimpardi L, et al. Biochemistry and biology of GDF11 and myostatin: similarities, differences, and questions for future investigation. Circ Res 2016;118:1125–1142discussion 1142.



131. Trendelenburg AU, Meyer A, Rohner D, Boyle J, Hatakeyama S, Glass DJ. Myostatin reduces Akt/TORC1/p70S6K signaling, inhibiting myoblast differentiation and myotube size. Am J Physiol Cell Physiol 2009;296:C1258–C1270.


132. Argilés JM, Orpí M, Busquets S, López-Soriano FJ. Myostatin: more than just a regulator of muscle mass. Drug Discov Today 2012;17:702–709.


133. Feike Y, Zhijie L, Wei C. Advances in research on pharmacotherapy of sarcopenia. Aging Med (Milton) 2021;4:221–233.




134. Yano S, Nagai A, Isomura M, et al. Relationship between blood myostatin levels and kidney function:shimane CoHRE study. PLoS One 2015;10:e0141035.



135. Verzola D, Procopio V, Sofia A, et al. Apoptosis and myostatin mRNA are upregulated in the skeletal muscle of patients with chronic kidney disease. Kidney Int 2011;79:773–782.


136. Price SR, Gooch JL, Donaldson SK, Roberts-Wilson TK. Muscle atrophy in chronic kidney disease results from abnormalities in insulin signaling. J Ren Nutr 2010;20(5 Suppl):S24–S28.



137. Zhang L, Pan J, Dong Y, et al. Stat3 activation links a C/EBPδ to myostatin pathway to stimulate loss of muscle mass. Cell Metab 2013;18:368–379.



138. Becker C, Lord SR, Studenski SA, et al. Myostatin antibody (LY2495655) in older weak fallers: a proof-of-concept, randomised, phase 2 trial. Lancet Diabetes Endocrinol 2015;3:948–957.


139. Wagner KR, Fleckenstein JL, Amato AA, et al. A phase I/IItrial of MYO-029 in adult subjects with muscular dystrophy. Ann Neurol 2008;63:561–571.


140. Morvan F, Rondeau JM, Zou C, et al. Blockade of activin type II receptors with a dual anti-ActRIIA/IIB antibody is critical to promote maximal skeletal muscle hypertrophy. Proc Natl Acad Sci U S A 2017;114:12448–12453.



141. Lach-Trifilieff E, Minetti GC, Sheppard K, et al. An antibody blocking activin type II receptors induces strong skeletal muscle hypertrophy and protects from atrophy. Mol Cell Biol 2014;34:606–618.




142. Rooks D, Praestgaard J, Hariry S, et al. Treatment of sarcopenia with bimagrumab: results from a phase II, randomized, controlled, proof-of-concept study. J Am Geriatr Soc 2017;65:1988–1995.



143. Rooks D, Swan T, Goswami B, et al. Bimagrumab vs optimized standard of care for treatment of sarcopenia in community-dwelling older adults: a randomized clinical trial. JAMA Netw Open 2020;3:e2020836.



144. Heymsfield SB, Coleman LA, Miller R, et al. Effect of bimagrumab vs placebo on body fat mass among adults with type 2 diabetes and obesity: a phase 2 randomized clinical trial. JAMA Netw Open 2021;4:e2033457.



145. Cadena SM, Tomkinson KN, Monnell TE, et al. Administration of a soluble activin type IIB receptor promotes skeletal muscle growth independent of fiber type. J Appl Physiol (1985) 2010;109:635–642.



146. Campbell C, McMillan HJ, Mah JK, et al. Myostatin inhibitor ACE-031 treatment of ambulatory boys with Duchenne muscular dystrophy: results of a randomized, placebo-controlled clinical trial. Muscle Nerve 2017;55:458–464.



147. Castonguay R, Lachey J, Wallner S, et al. Follistatin-288-Fc fusion protein promotes localized growth of skeletal muscle. J Pharmacol Exp Ther 2019;368:435–445.


148. Ozawa T, Morikawa M, Morishita Y, et al. Systemic administration of monovalent follistatin-like 3-Fc-fusion protein increases muscle mass in mice. iScience 2021;24:102488.



149. Pearsall RS, Davies MV, Cannell M, et al. Follistatin-based ligand trap ACE-083 induces localized hypertrophy of skeletal muscle with functional improvement in models of neuromuscular disease. Sci Rep 2019;9:11392.




150. Statland JM, Campbell C, Desai U, et al. Randomized phase 2 study of ACE-083, a muscle-promoting agent, in facioscapulohumeral muscular dystrophy. Muscle Nerve 2022;66:50–62.


151. Giesige CR, Wallace LM, Heller KN, et al. AAV-mediated follistatin gene therapy improves functional outcomes in the TIC-DUX4 mouse model of FSHD. JCI Insight 2018;3:e123538.



152. Mendell JR, Sahenk Z, Al-Zaidy S, et al. Follistatin gene therapy for sporadic inclusion body myositis improves functional outcomes. Mol Ther 2017;25:870–879.



153. Al-Zaidy SA, Sahenk Z, Rodino-Klapac LR, Kaspar B, Mendell JR. Follistatin gene therapy improves ambulation in Becker muscular dystrophy. J Neuromuscul Dis 2015;2:185–192.



154. Mendell JR, Sahenk Z, Malik V, et al. A phase 1/2a follistatin gene therapy trial for becker muscular dystrophy. Mol Ther 2015;23:192–201.



155. Ravarotto V, Bertoldi G, Stefanelli LF, Nalesso F, Calò LA. Pathomechanism of oxidative stress in cardiovascularrenal remodeling and therapeutic strategies. Kidney Res Clin Pract 2022;41:533–544.




156. Mak RH, Ikizler AT, Kovesdy CP, Raj DS, Stenvinkel P, Kalantar-Zadeh K. Wasting in chronic kidney disease. J Cachexia Sarcopenia Muscle 2011;2:9–25.



157. Axelsson J, Heimbürger O, Stenvinkel P. Adipose tissue and inflammation in chronic kidney disease. Contrib Nephrol 2006;151:165–174.


158. Cheung WW, Paik KH, Mak RH. Inflammation and cachexia in chronic kidney disease. Pediatr Nephrol 2010;25:711–724.



159. Ramalingam L, Menikdiwela K, LeMieux M, et al. The renin angiotensin system, oxidative stress and mitochondrial function in obesity and insulin resistance. Biochim Biophys Acta Mol Basis Dis 2017;1863:1106–1114.


160. Powers SK, Morton AB, Hyatt H, Hinkley MJ. The renin-angiotensin system and skeletal muscle. Exerc Sport Sci Rev 2018;46:205–214.



161. Allen AM, Zhuo J, Mendelsohn FA. Localization and function of angiotensin AT1 receptors. Am J Hypertens 2000;13(1 Pt 2):31S–38S.


162. Linderman JR, Greene AS. Distribution of angiotensin II receptor expression in the microcirculation of striated muscle. Microcirculation 2001;8:275–281.


163. Santos RAS, Sampaio WO, Alzamora AC, et al. The ACE2/angiotensin-(1-7)/MAS axis of the renin-angiotensin system: focus on angiotensin-(1-7). Physiol Rev 2018;98:505–553.



164. Wright JT Jr, Bakris G, Greene T, et al. Effect of blood pressure lowering and antihypertensive drug class on progression of hypertensive kidney disease: results from the AASK trial. JAMA 2002;288:2421–2431.


165. Lewis EJ, Hunsicker LG, Clarke WR, et al. Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med 2001;345:851–860.


166. Jafar TH, Schmid CH, Landa M, et al. Angiotensin-converting enzyme inhibitors and progression of nondiabetic renal disease. A meta-analysis of patient-level data. Ann Intern Med 2001;135:73–87.


167. Mann JF, Gerstein HC, Pogue J, Bosch J, Yusuf S. Renal insufficiency as a predictor of cardiovascular outcomes and the impact of ramipril: the HOPE randomized trial. Ann Intern Med 2001;134:629–636.


168. Onder G, Penninx BW, Balkrishnan R, et al. Relation between use of angiotensin-converting enzyme inhibitors and muscle strength and physical function in older women: an observational study. Lancet 2002;359:926–930.


169. Hutcheon SD, Gillespie ND, Crombie IK, Struthers AD, McMurdo ME. Perindopril improves six minute walking distance in older patients with left ventricular systolic dysfunction: a randomised double blind placebo controlled trial. Heart 2002;88:373–377.



170. Maggio M, Ceda GP, Lauretani F, et al. Relation of angiotensin-converting enzyme inhibitor treatment to insulin-like growth factor-1 serum levels in subjects >65 years of age (the InCHIANTI study). Am J Cardiol 2006;97:1525–1529.



171. LACE study group; Achison M, et al.; Adamson S. Effect of perindopril or leucine on physical performance in older people with sarcopenia: the LACE randomized controlled trial. J Cachexia Sarcopenia Muscle 2022;13:858–871.




172. Burks TN, Andres-Mateos E, Marx R, et al. Losartan restores skeletal muscle remodeling and protects against disuse atrophy in sarcopenia. Sci Transl Med 2011;3:82ra37.



173. Lin CH, Yang H, Xue QL, et al. Losartan improves measures of activity, inflammation, and oxidative stress in older mice. Exp Gerontol 2014;58:174–178.



174. Pahor M, Anton SD, Beavers DP, et al. Effect of losartan and fish oil on plasma IL-6 and mobility in older persons. The ENRGISE pilot randomized clinical trial. J Gerontol A Biol Sci Med Sci 2019;74:1612–1619.




175. Murphy KT, Hossain MI, Swiderski K, et al. Mas receptor activation slows tumor growth and attenuates muscle wasting in cancer. Cancer Res 2019;79:706–719.




176. Levi A, Cohen E, Levi M, Goldberg E, Garty M, Krause I. Elevated serum homocysteine is a predictor of accelerated decline in renal function and chronic kidney disease: a historical prospective study. Eur J Intern Med 2014;25:951–955.


177. Bostom AG, Shemin D, Verhoef P, et al. Elevated fasting total plasma homocysteine levels and cardiovascular disease outcomes in maintenance dialysis patients. A prospective study. Arterioscler Thromb Vasc Biol 1997;17:2554–2558.

178. Robinson K, Gupta A, Dennis V, et al. Hyperhomocysteinemia confers an independent increased risk of atherosclerosis in end-stage renal disease and is closely linked to plasma folate and pyridoxine concentrations. Circulation 1996;94:2743–2748.


179. Swart KM, van Schoor NM, Heymans MW, Schaap LA, den Heijer M, Lips P. Elevated homocysteine levels are associated with low muscle strength and functional limitations in older persons. J Nutr Health Aging 2013;17:578–584.



180. Vidoni ML, Pettee Gabriel K, Luo ST, Simonsick EM, Day RS. Relationship between homocysteine and muscle strength decline: the baltimore longitudinal study of aging. J Gerontol A Biol Sci Med Sci 2018;73:546–551.



181. Ng TP, Aung KC, Feng L, Scherer SC, Yap KB. Homocysteine, folate, vitamin B-12, and physical function in older adults: cross-sectional findings from the Singapore Longitudinal Ageing Study. Am J Clin Nutr 2012;96:1362–1368.


182. van Schoor NM, Swart KM, Pluijm SM, et al. Cross-sectional and longitudinal association between homocysteine, vitamin B12 and physical performance in older persons. Eur J Clin Nutr 2012;66:174–181.



183. Swart KM, Ham AC, van Wijngaarden JP, et al. A randomized controlled trial to examine the effect of 2-year vitamin B12 and folic acid supplementation on physical performance, strength, and falling: additional findings from the B-PROOF study. Calcif Tissue Int 2016;98:18–27.




184. Padayatty SJ, Katz A, Wang Y, et al. Vitamin C as an antioxidant: evaluation of its role in disease prevention. J Am Coll Nutr 2003;22:18–35.


185. Welch AA, Jennings A, Kelaiditi E, Skinner J, Steves CJ. Cross-sectional associations between dietary antioxidant vitamins C, E and carotenoid intakes and sarcopenic indices in women aged 18-79 years. Calcif Tissue Int 2020;106:331–342.




186. Konopacka M, Rzeszowska-Wolny J. Antioxidant vitamins C, E and beta-carotene reduce DNA damage before as well as after gamma-ray irradiation of human lymphocytes in vitro. Mutat Res 2001;491:1–7.

187. Poljšak B, Raspor P. The antioxidant and pro-oxidant activity of vitamin C and trolox in vitro: a comparative study. J Appl Toxicol 2008;28:183–188.


188. Liu X, Wang P, Zou YX, Luo ZG, Tamer TM. Co-encapsulation of vitamin C and β-carotene in liposomes: storage stability, antioxidant activity, and in vitro gastrointestinal digestion. Food Res Int 2020;136:109587.


189. Takahashi N, Morimoto S, Okigaki M, et al. Decreased plasma level of vitamin C in chronic kidney disease: comparison between diabetic and non-diabetic patients. Nephrol Dial Transplant 2011;26:1252–1257.


190. Deicher R, Hörl WH. Vitamin C in chronic kidney disease and hemodialysis patients. Kidney Blood Press Res 2003;26:100–106.



191. Molina P, Carrero JJ, Bover J, et al. Vitamin D, a modulator of musculoskeletal health in chronic kidney disease. J Cachexia Sarcopenia Muscle 2017;8:686–701.




192. Bauer JM, Verlaan S, Bautmans I, et al. Effects of a vitamin D and leucine-enriched whey protein nutritional supplement on measures of sarcopenia in older adults, the PROVIDE study: a randomized, double-blind, placebo-controlled trial. J Am Med Dir Assoc 2015;16:740–747.


193. Beaudart C, Buckinx F, Rabenda V, et al. The effects of vitamin D on skeletal muscle strength, muscle mass, and muscle power: a systematic review and meta-analysis of randomized controlled trials. J Clin Endocrinol Metab 2014;99:4336–4345.


194. Murad MH, Elamin KB, Abu Elnour NO, et al. The effect of vitamin D on falls: a systematic review and meta-analysis. J Clin Endocrinol Metab 2011;96:2997–3006.


195. Nelsestuen GL, Zytkovicz TH, Howard JB. The mode of action of vitamin K. Identification of gamma-carboxyglutamic acid as a component of prothrombin. J Biol Chem 1974;249:6347–6350.

196. Stenflo J, Fernlund P, Egan W, Roepstorff P. Vitamin K dependent modifications of glutamic acid residues in prothrombin. Proc Natl Acad Sci U S A 1974;71:2730–2733.



197. Tew BY, Hong TB, Otto-Duessel M, et al. Vitamin K epoxide reductase regulation of androgen receptor activity. Oncotarget 2017;8:13818–13831.



198. Holden RM, Morton AR, Garland JS, Pavlov A, Day AG, Booth SL. Vitamins K and D status in stages 3-5 chronic kidney disease. Clin J Am Soc Nephrol 2010;5:590–597.



199. Cranenburg EC, Schurgers LJ, Uiterwijk HH, et al. Vitamin K intake and status are low in hemodialysis patients. Kidney Int 2012;82:605–610.


200. Shea MK, Loeser RF, Hsu FC, et al. Vitamin K status and lower extremity function in older adults: the health aging and body composition study. J Gerontol A Biol Sci Med Sci 2016;71:1348–1355.



201. van Ballegooijen AJ, van Putten SR, Visser M, Beulens JW, Hoogendijk EO. Vitamin K status and physical decline in older adults-The Longitudinal Aging Study Amsterdam. Maturitas 2018;113:73–79.


202. Shea MK, Dawson-Hughes B, Gundberg CM, Booth SL. Reducing undercarboxylated osteocalcin with vitamin K supplementation does not promote lean tissue loss or fat gain over 3 years in older women and men: a randomized controlled trial. J Bone Miner Res 2017;32:243–249.




203. Fulton RL, McMurdo ME, Hill A, et al. Effect of vitamin K on vascular health and physical function in older people with vascular disease--a randomised controlled trial. J Nutr Health Aging 2016;20:325–333.



204. Meng X, Zhou J, Zhao CN, Gan RY, Li HB. Health benefits and molecular mechanisms of resveratrol: a narrative review. Foods 2020;9:340.



205. Harikumar KB, Aggarwal BB. Resveratrol: a multitargeted agent for age-associated chronic diseases. Cell Cycle 2008;7:1020–1035.


206. Joseph AM, Malamo AG, Silvestre J, et al. Short-term caloric restriction, resveratrol, or combined treatment regimens initiated in late-life alter mitochondrial protein expression profiles in a fiber-type specific manner in aged animals. Exp Gerontol 2013;48:858–868.



207. Bennett BT, Mohamed JS, Alway SE. Effects of resveratrol on the recovery of muscle mass following disuse in the plantaris muscle of aged rats. PLoS One 2013;8:e83518.



208. Liao ZY, Chen JL, Xiao MH, et al. The effect of exercise, resveratrol or their combination on Sarcopenia in aged rats via regulation of AMPK/Sirt1 pathway. Exp Gerontol 2017;98:177–183.


209. Kan NW, Ho CS, Chiu YS, et al. Effects of resveratrol supplementation and exercise training on exercise performance in middle-aged mice. Molecules 2016;21:661.



210. Muhammad MH, Allam MM. Resveratrol and/or exercise training counteract aging-associated decline of physical endurance in aged mice; targeting mitochondrial biogenesis and function. J Physiol Sci 2018;68:681–688.



211. Olesen J, Gliemann L, Biensø R, Schmidt J, Hellsten Y, Pilegaard H. Exercise training, but not resveratrol, improves metabolic and inflammatory status in skeletal muscle of aged men. J Physiol 2014;592:1873–1886.



212. Kojima M, Hosoda H, Date Y, Nakazato M, Matsuo H, Kangawa K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 1999;402:656–660.



213. Hosoda H, Kojima M, Matsuo H, Kangawa K. Purification and characterization of rat des-Gln14-Ghrelin, a second endogenous ligand for the growth hormone secretagogue receptor. J Biol Chem 2000;275:21995–22000.


214. Hosoda H, Kojima M, Matsuo H, Kangawa K. Ghrelin and des-acyl ghrelin: two major forms of rat ghrelin peptide in gastrointestinal tissue. Biochem Biophys Res Commun 2000;279:909–913.


215. Rodriguez Ayala E, Pecoits-Filho R, Heimbürger O, Lindholm B, Nordfors L, Stenvinkel P. Associations between plasma ghrelin levels and body composition in end-stage renal disease: a longitudinal study. Nephrol Dial Transplant 2004;19:421–426.


216. DeBoer MD, Zhu XX, Levasseur P, et al. Ghrelin treatment causes increased food intake and retention of lean body mass in a rat model of cancer cachexia. Endocrinology 2007;148:3004–3012.


217. DeBoer MD, Zhu X, Levasseur PR, et al. Ghrelin treatment of chronic kidney disease: improvements in lean body mass and cytokine profile. Endocrinology 2008;149:827–835.




218. Miyazaki T, Tanaka N, Hirai H, et al. Ghrelin level and body weight loss after esophagectomy for esophageal cancer. J Surg Res 2012;176:74–78.


219. Argilés JM, Stemmler B. The potential of ghrelin in the treatment of cancer cachexia. Expert Opin Biol Ther 2013;13:67–76.


220. Garcia JM, Boccia RV, Graham CD, et al. Anamorelin for patients with cancer cachexia: an integrated analysis of two phase 2, randomised, placebo-controlled, double-blind trials. Lancet Oncol 2015;16:108–116.


221. Temel J, Bondarde S, Jain M, Yan Y, Duus E, Allen S. Efficacy and safety results from a phase II study of anamorelin HCl, a ghrelin receptor agonist. NSCLC patients. J Cachexia Sarcopenia Muscle 2013;4:295–343.


222. White HK, Petrie CD, Landschulz W, et al. Effects of an oral growth hormone secretagogue in older adults. J Clin Endocrinol Metab 2009;94:1198–1206.


223. Alcalde-Estévez E, Sosa P, Asenjo-Bueno A, et al. Uraemic toxins impair skeletal muscle regeneration by inhibiting myoblast proliferation, reducing myogenic differentiation, and promoting muscular fibrosis. Sci Rep 2021;11:512.




224. Ohtsuki S, Asaba H, Takanaga H, et al. Role of blood-brain barrier organic anion transporter 3 (OAT3) in the efflux of indoxyl sulfate, a uremic toxin: its involvement in neurotransmitter metabolite clearance from the brain. J Neurochem 2002;83:57–66.


225. Deguchi T, Ohtsuki S, Otagiri M, et al. Major role of organic anion transporter 3 in the transport of indoxyl sulfate in the kidney. Kidney Int 2002;61:1760–1768.


226. Koppe L, Pillon NJ, Vella RE, et al. p-Cresyl sulfate promotes insulin resistance associated with CKD. J Am Soc Nephrol 2013;24:88–99.



227. Sato E, Mori T, Mishima E, et al. Metabolic alterations by indoxyl sulfate in skeletal muscle induce uremic sarcopenia in chronic kidney disease. Sci Rep 2016;6:36618.




228. Niwa T. Role of indoxyl sulfate in the progression of chronic kidney disease and cardiovascular disease: experimental and clinical effects of oral sorbent AST-120. Ther Apher Dial 2011;15:120–124.


229. Enoki Y, Watanabe H, Arake R, et al. Indoxyl sulfate potentiates skeletal muscle atrophy by inducing the oxidative stress-mediated expression of myostatin and atrogin-1. Sci Rep 2016;6:32084.




230. Sallée M, Dou L, Cerini C, Poitevin S, Brunet P, Burtey S. The aryl hydrocarbon receptor-activating effect of uremic toxins from tryptophan metabolism: a new concept to understand cardiovascular complications of chronic kidney disease. Toxins (Basel) 2014;6:934–949.



231. Watanabe I, Tatebe J, Namba S, Koizumi M, Yamazaki J, Morita T. Activation of aryl hydrocarbon receptor mediates indoxyl sulfate-induced monocyte chemoattractant protein-1 expression in human umbilical vein endothelial cells. Circ J 2013;77:224–230.


-
METRICS

- Related articles
-
The role of roxadustat in chronic kidney disease patients complicated with anemia2023 March;38(2)
Emotional and cognitive changes in chronic kidney disease2022 May;37(3)
Hepatitis C virus infection in chronic kidney disease: paradigm shift in management2018 July;33(4)
Treatment of diabetic kidney disease: current and future targets2017 July;32(4)



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print


